Solubility Solubility is the property of a substance

Solubility

Solubility is the property of a substance (solute) to dissolve in another substance (solvent); solutions are characterized by their capacity to do so Soluble the solutes ability to dissolve in a solvent Insoluble the solute is not able to dissolve in a solvent aqueous solutions of KMn. O 4 Cd. S Pb. S Ni(OH)2 Al(OH)3

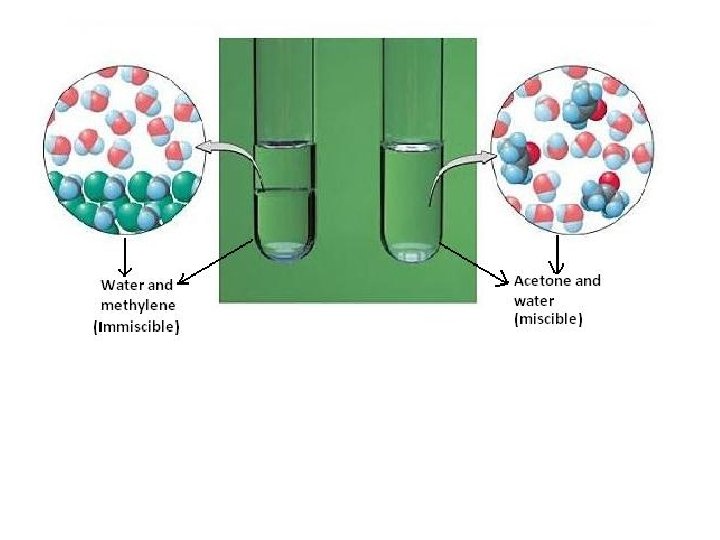
Solubility is the property of a substance (solute) to dissolve in another substance (solvent). • Measured in terms of the maximum amount of solute dissolved in a solvent at a specific temperature • The resulting solution is called a saturated solution • Certain substances are soluble in all proportions with a given solvent, this property is known as miscibility. • Solubility ranges widely from infinitely soluble to poorly soluble; in a relevant sense scientists refer to substances as being soluble, slightly soluble, or insoluble • Insoluble is often applied to poorly soluble compounds, though strictly speaking there are very few cases where there is absolutely no material dissolved 3

Example Predict the relative solubility in the following cases: (a) Bromine in benzene and in water (b) KCl in carbon tetrachloride and in liquid ammonia (c) formaldehyde (CH 2 O) in carbon disulfide and in water

Solubility Units
7
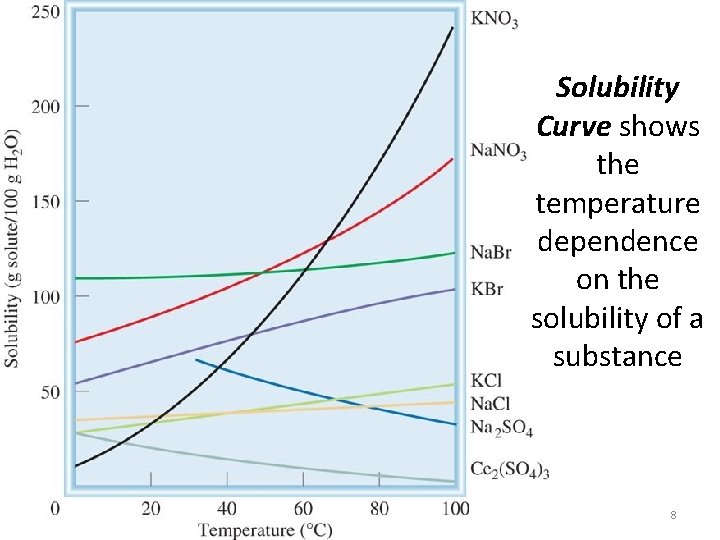
Solubility Curve shows the temperature dependence on the solubility of a substance 8
Are the solutions the same? How can we describe them?

Concentration is the amount of solute present for a given amount of solvent or solution; relative or absolute Concentrated contains greater amount of solute per volume of solution Dilute contains relatively small amounts of solute Add solvent per volume of solution

Concentration Units M = molarity = moles of solute liters of solution ppm = parts per million = mg of solute liters of solution 11

Examples 1. Calculate the molarity of a 1. 75 L solution containing 3. 45 moles of calcium chloride. 1. Calculate the molarity of an 855 m. L solution containing 2. 75 moles of sodium nitrate.

3. A 100. 5 m. L IV solution contains 5. 10 g of glucose. What is the molarity of this solution? 3. How many moles of sodium nitrate are in 655 m. L of a 3. 45 M solution?

5. 150 m. L of an aqueous sodium chloride solution contains 0. 0045 g of sodium chloride. Calculate the concentration of sodium chloride in parts per million. 6. If 25 grams of a chemical is dissolved in 75 grams of water, what is the concentration of the chemical in parts per million?
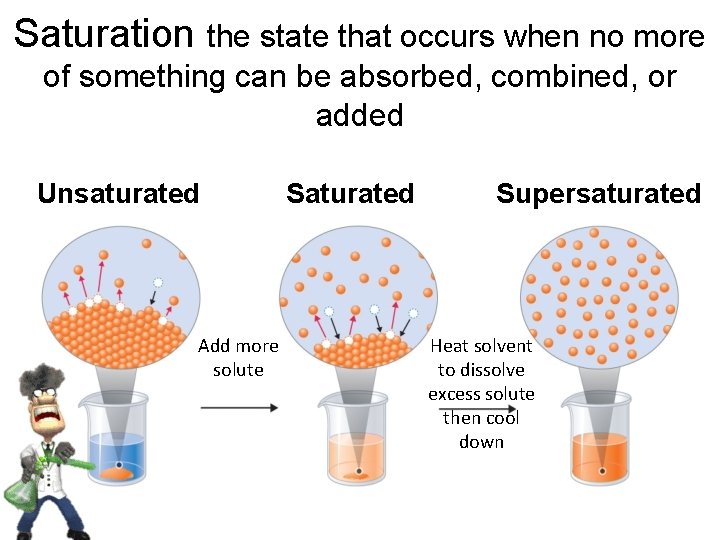
Saturation the state that occurs when no more of something can be absorbed, combined, or added Unsaturated Add more solute Saturated Supersaturated Heat solvent to dissolve excess solute then cool down

A saturated solution contains the maximum amount of a solute that will dissolve in a given solvent at a specific temperature. An unsaturated solution contains less solute than the solvent has the capacity to dissolve at a specific temperature. A supersaturated solution contains more solute than is present in a saturated solution at a specific temperature. 16

Crystallization is the process in which dissolved solute comes out of solution and forms crystals; describes the separation of excess solid substance from a supersaturated solution. Sodium acetate crystals rapidly form when a seed crystal is added to a supersaturated solution of sodium acetate. 17

Factors that Affect Solubility 1. Temperature a. Solids b. Gases 2. Pressure (gasses only)

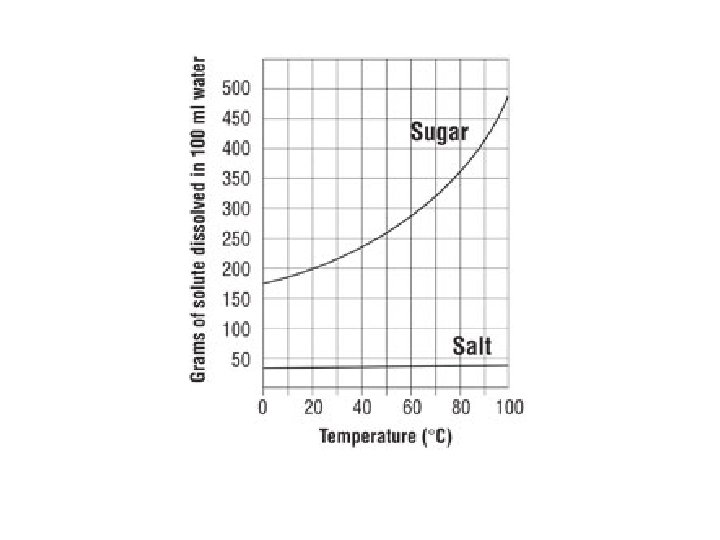
Solid Solubility & Temp Salt Sugar
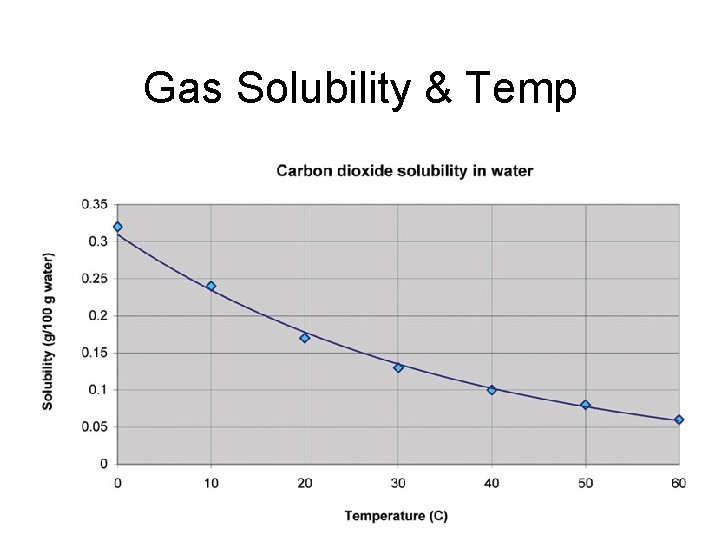
Gas Solubility & Temp
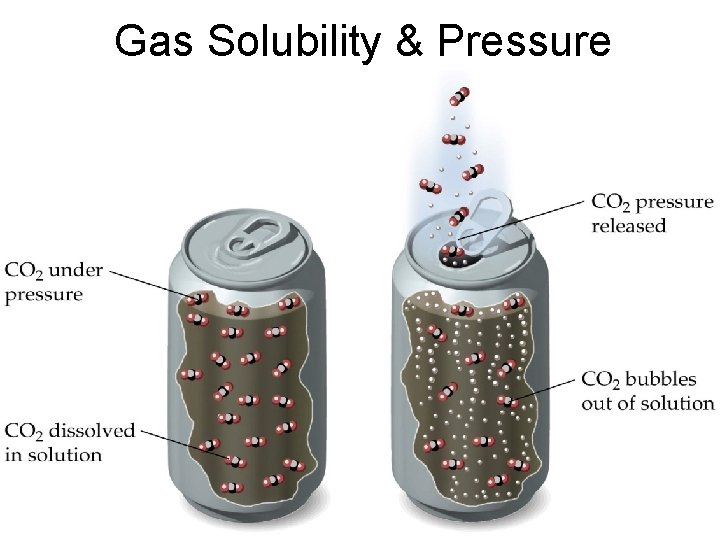
Gas Solubility & Pressure
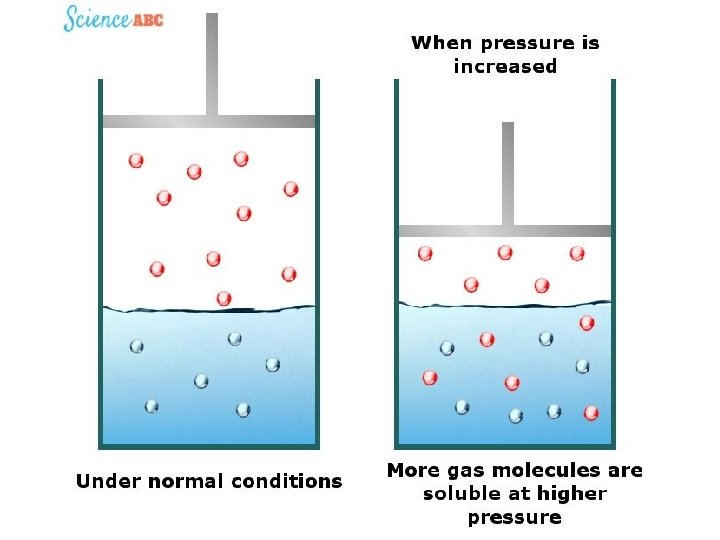
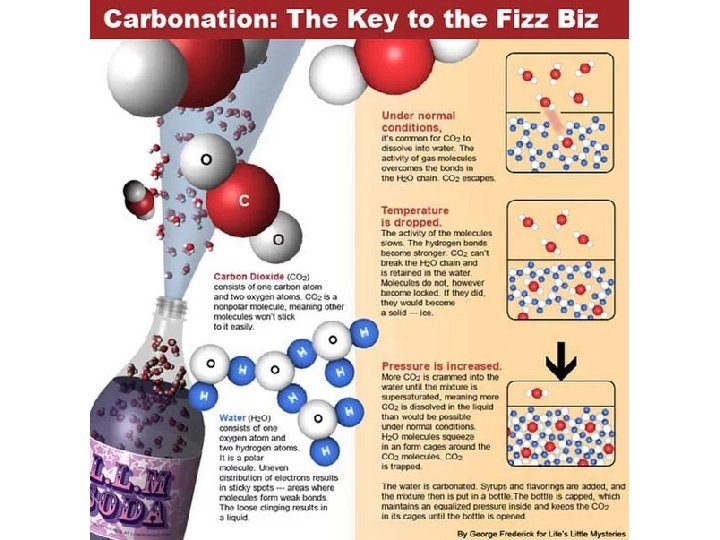

Henry’s Law The solubility of a gas in a liquid is proportional to the pressure of the gas over the solution S is the solubility of the substance in solution P is the pressure of the gas over the solution low P high P low c high c 25

Example The solubility of a gas is 0. 35 g/L at 25. 0 k. Pa of pressure. What is the solubility when the pressure is increased to 115 k. Pa?
- Slides: 26