SOLUBILITY Solubility how many grams of solute that

SOLUBILITY

Solubility how many grams of solute that will dissolve in 100 g of solvent to make it saturated at a given temperature

Dissolution- the rate at which a substance dissolves

FACTORS AFFECTING SOLUBILITY

Factors Affecting Solubility Surface Area Stirring Temperature Pressure Polarity

AGITATION Stirring or Agitation increases the rate of solubility of solid solute particles in a solvent. It speeds up the contact between the surface of the solute and the solvent particles.

Effect of Temperature on Solubility of Solids and Liquids • Generally, increasing the temperature will increase solubility of solids and liquids. • Increase temperature increases kinetic energy of the molecules (i. e. molecules move faster) • Solute and solvent and come in contact more frequently thereby increasing rate of solution
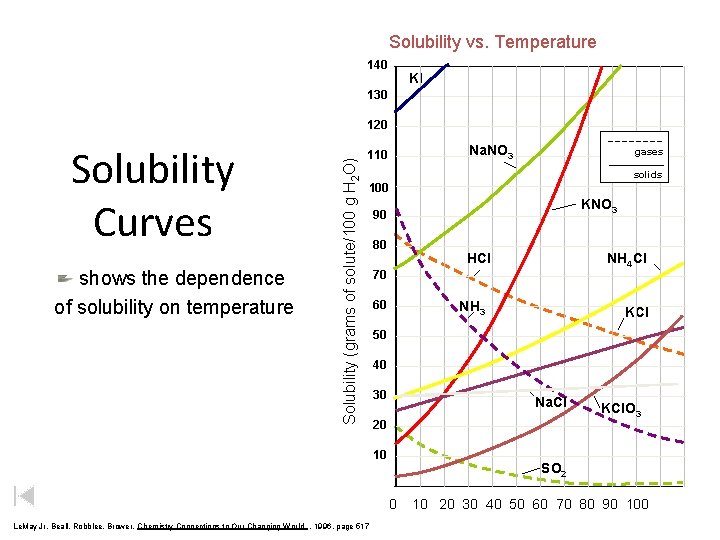
Solubility vs. Temperature 140 KI 130 Solubility Curves shows the dependence of solubility on temperature Solubility (grams of solute/100 g H 2 O) 120 Na. NO 3 110 gases solids 100 KNO 3 90 80 HCl 70 60 NH 3 KCl 50 40 30 Na. Cl 20 10 KCl. O 3 SO 2 0 Le. May Jr, Beall, Robblee, Brower, Chemistry Connections to Our Changing World , 1996, page 517 NH 4 Cl 10 20 30 40 50 60 70 80 90 100

Effect of Temperature on Solubility of Solids and Liquids But increasing temperature will lower the solubility of gases (the gas will have enough K. E. to overcome the attraction of the solvent and escape from solution).
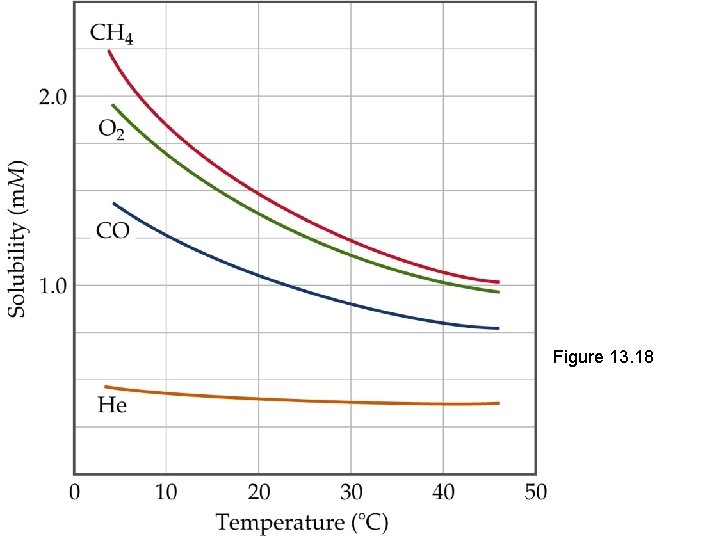
Figure 13. 18

Making Connections • Thermal pollution: if lakes get too warm, CO 2 and O 2 become less soluble and are not available for plants or animals. • Warm Pop

SURFACE AREA Particle size – area of solute particles exposed to the action of the solvent particles. Increase in surface area “windows of opportunity for solute to contact solvent” will increase solubility Example: fine table salt dissolves faster than rock salt http: //www. youtube. com/watch? v=y. Rw 4 ZRqmx. Oc

The Effect of Pressure on the Solubility of Gases • Pressure has no effect on the solubility of solids and liquids but has a strong effect on the solubility of gases. • The solubility of gases increases when the pressure above the gas is increased. In other words, more gas will dissolve when pressure is increased. This is known as HENRY’S LAW (William Henry, English chemist).

Figure 13. 14: Factors Affecting Solubility Pressure Effects

Making Connections • Nitrogen narcosis, the “bends” • Soda http: //www. youtube. com/watch? v=Lf. COn. GHheok&feature=fvst


Types of Solvent How does a solid dissolve into a liquid? What ‘drives’ the dissolution process?
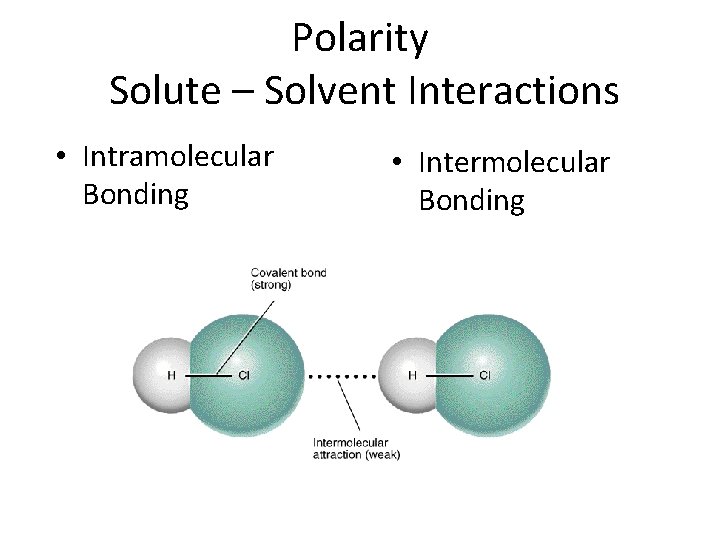
Polarity Solute – Solvent Interactions • Intramolecular Bonding • Intermolecular Bonding

Intramolecular Bonding • Intramolecular bonding refers to the chemical bonding that holds atoms together within a molecule of a compound Covalent bonding and ionic bonding are the two main types of intramolecular bonding

• Ionic bonding involves the transfer of valence electrons from one atom to another. • Electrostatic attraction between opposite charged ions holds the bond together in crystal lattice.
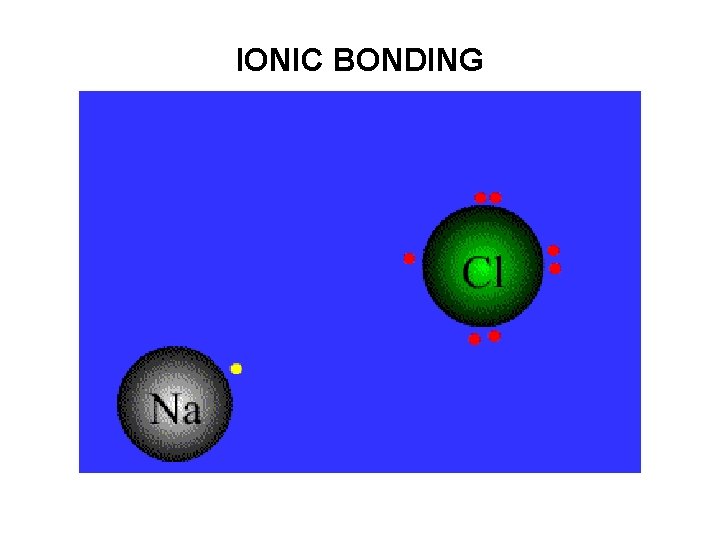
IONIC BONDING

Covalent bonding involves the sharing of valence electrons between two atoms. Eg. covalent bonding holds hydrogen and oxygen atoms together to form a water molecule, H 2 O. POLAR- unequal sharing of electrons NON POLAR – equal sharing of electrons
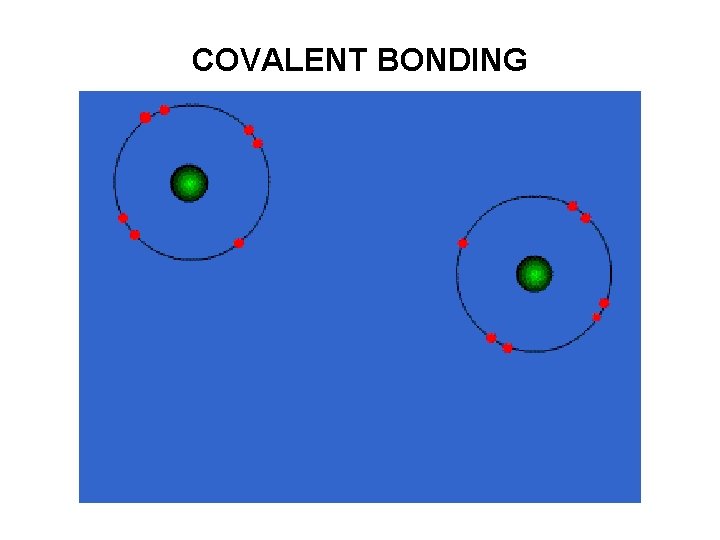
COVALENT BONDING

NON POLAR MOLECULE • Nonpolar molecules either have no positive and negative ends, because the bonds making up the molecule are nonpolar, or because the entire outer "edge" is negative while the core of the molecule is positive (or vice versa), thus having no oppositely charged ends.

POLAR MOLECULE • A polar molecule will have one end of the molecule bearing a partial positive charge while another end carries a partial negative charge. Polar molecules must contain polar bonds.
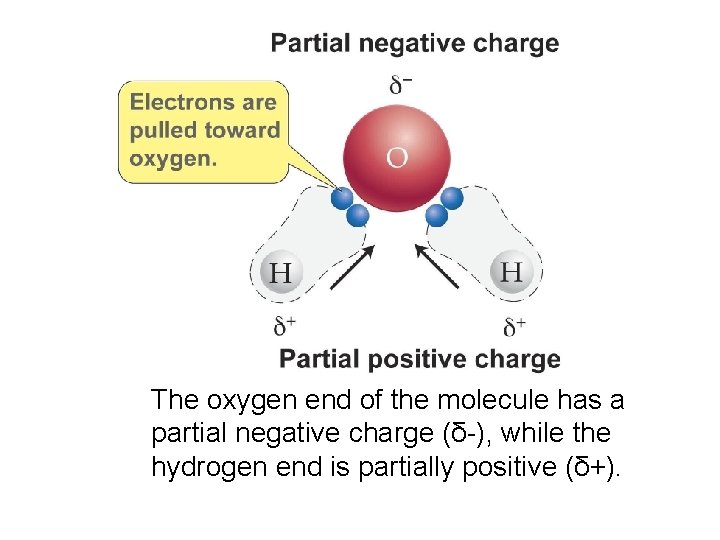
The oxygen end of the molecule has a partial negative charge (δ-), while the hydrogen end is partially positive (δ+).
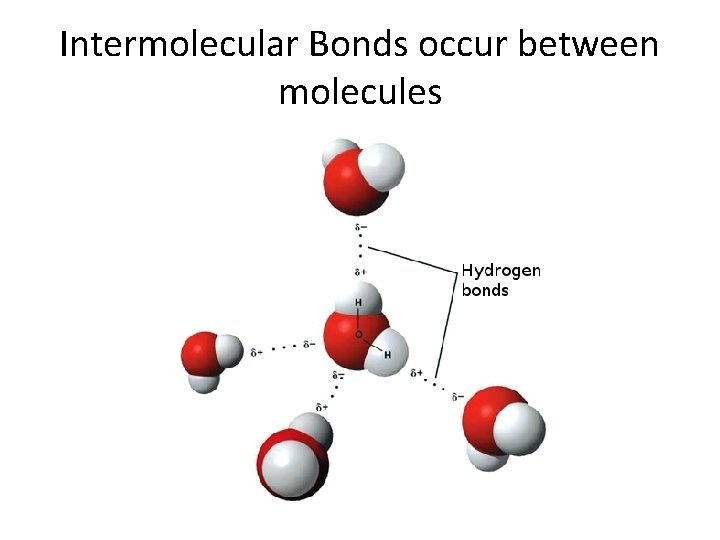
Intermolecular Bonds occur between molecules

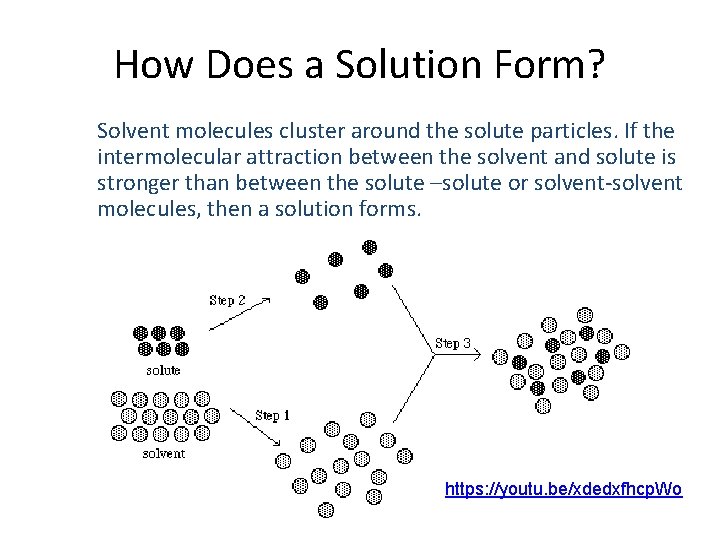
How Does a Solution Form? Solvent molecules cluster around the solute particles. If the intermolecular attraction between the solvent and solute is stronger than between the solute –solute or solvent-solvent molecules, then a solution forms. https: //youtu. be/xdedxfhcp. Wo

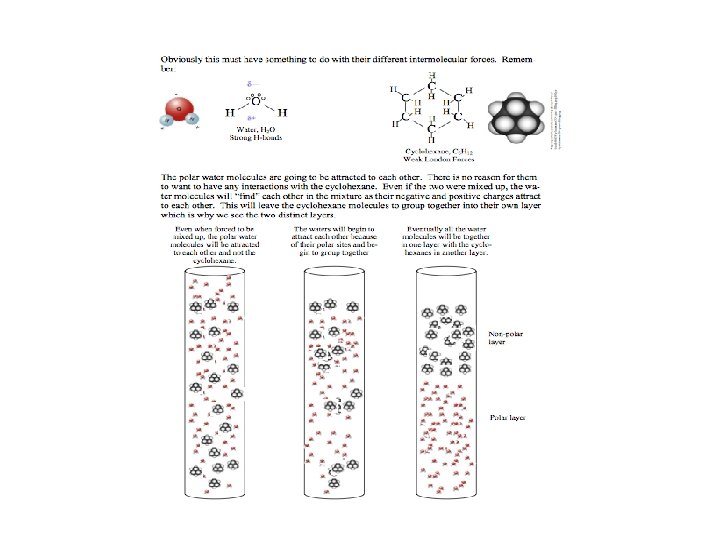
Take home message“like dissolves like” Two substances with similar intermolecular forces are likely to be soluble in each other. • non-polar molecules are soluble in non-polar solvents CCl 4 in C 6 H 6 • polar molecules are soluble in polar solvents C 2 H 5 OH in H 2 O • ionic compounds are more soluble in polar solvents Na. Cl in H 2 O or NH 3 (l) 12. 2
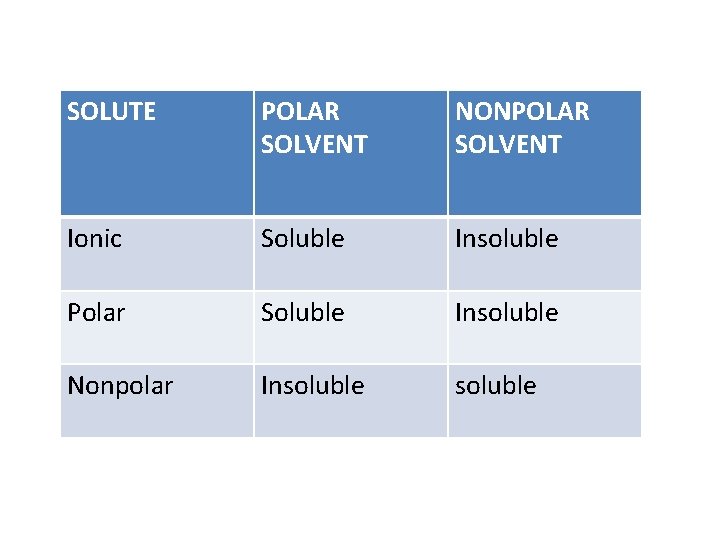
SOLUTE POLAR SOLVENT NONPOLAR SOLVENT Ionic Soluble Insoluble Polar Soluble Insoluble Nonpolar Insoluble

Making Connections Vitamins • Water Soluble – Vitamin C – Must be replenished regularly • Fat Soluble – Can overdose (liver damage, hair loss, blurred vision, numbness) – Vitamin A – Can be ingested periodically, stored in body fat
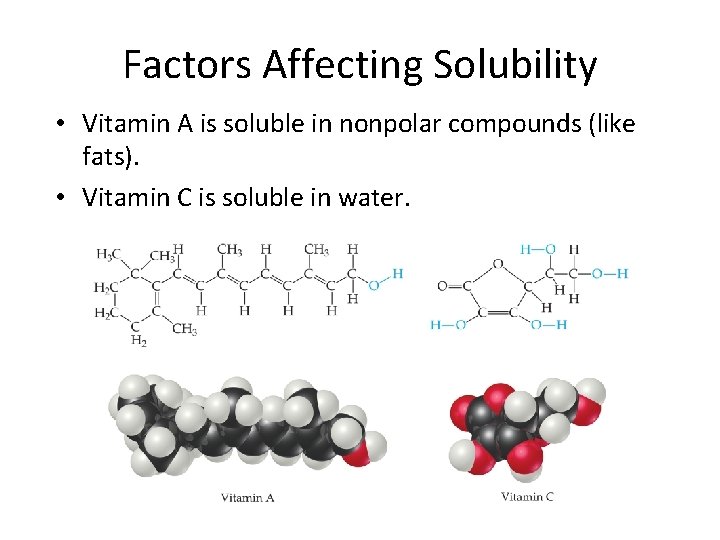
Factors Affecting Solubility • Vitamin A is soluble in nonpolar compounds (like fats). • Vitamin C is soluble in water.

How Salt Dissolves? ION-POLE ATTRACTION Watch video and write down explanation on sheet provided.

How Salt Dissolves? ION-POLE ATTRACTION • Each ion in the solute is surrounded by the partial positive and negative solvent molecules in the water. • Solute is pulled out of its lattice and solvent molecules spread the solute out throughout the solution.
- Slides: 37