Solubility Product Constant Factors Affecting Solubility Selective Precipitation

Solubility Product Constant Factors Affecting Solubility Selective Precipitation

• Objectives ▫ Today I will be able to: Calculate the solubility product constant for a compound Explain the factors that affect solubility Determine the compounds that selectively precipitate from a solution when placed under stresses • Informal assessment – monitoring student interactions and questions as they complete the practice problems • Formal assessment – analyzing student responses to the practice problems

Lesson Sequence • • Evaluate: Warm Up Explain: Notes Elaborate: Practice Problems Evaluate: Exit Ticket

Warm Up

Objectives • Today I will be able to: ▫ Calculate the solubility product constant for a compound ▫ Explain the factors that affect solubility ▫ Determine the compounds that selectively precipitate from a solution when placed under stresses

Homework • • Continue working practice problems Ch. 17 49, 51, 53, 55, 57, 59 69, 70, 75, 76

Agenda • • Warm Up Notes Practice Problems Exit Ticket

Solubility Product Constant Factors Affecting Solubility Selective Precipitation Notes

Solubility Equilibria

Solubility Product Constant • Consider the equilibrium that exists in a saturated solution of Ba. SO 4 in water: Ba. SO 4(s) Ba 2+(aq) + SO 42−(aq)

Solubility Product Constant Expression The equilibrium constant expression for this equilibrium is Ksp = [Ba 2+] [SO 42−] where the equilibrium constant, Ksp, is called the solubility product.

Ksp is not the same as solubility

What is solubility? What is Ksp? • Solubility is generally expressed as the mass of solute dissolved in 1 L (g/L) or 100 m. L (g/m. L) of solution, or in mol/L (M). • Equilibrium constant for equilibrium between an ionic sold and its saturated solution • Unitless • Larger magnitude means more dissolving occurred • Only one value per temperature • Does not change with p. H or other ions present in solution like solubility

Calculation • Solid silver chromate is added to pure water at 25 o. C and some solid remains undissolved. The mixture is stirred for several days to ensure that equilibrium is achieved between the undissolved Ag 2 Cr. O 4 and the solution. Analysis of the equilibrated solution shows that its silver ion concentration is 1. 3 x 10 -4 M. Assuming that the Ag 2 Cr. O 4 solution is saturated , calculate Ksp for the compound.

Calculation • The Ksp of Ca. F 2 is 3. 9 x 1 o-11 at 25 o. C. Assuming equilibrium is established between solid and dissolved Ca. F 2, calculate the solubility of Ca. F 2 in grams/liter

Factors impacting solubility

1. Common Ion Effect • If one of the ions in a solution equilibrium is already dissolved in the solution, the equilibrium will shift to the left and the solubility of the salt will decrease Ba. SO 4(s) Ba 2+(aq) + SO 42−(aq)
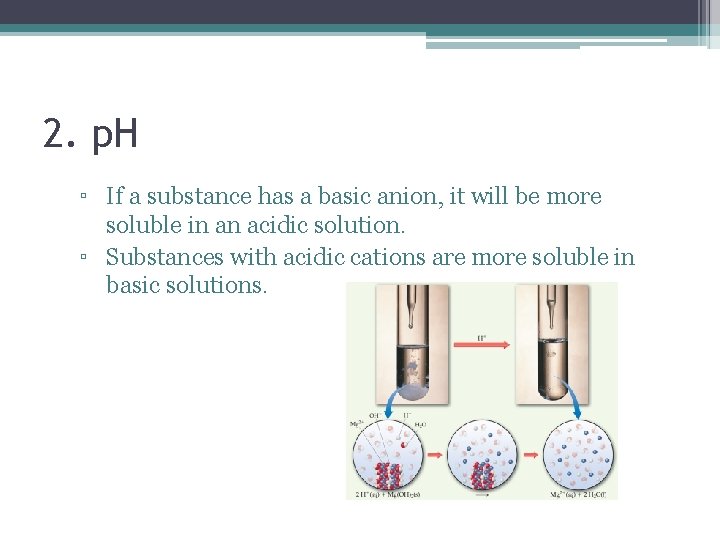
2. p. H ▫ If a substance has a basic anion, it will be more soluble in an acidic solution. ▫ Substances with acidic cations are more soluble in basic solutions.

3. Complex Ions • Complex Ions ▫ Metal ions can act as Lewis acids and form complex ions with Lewis bases in the solvent.
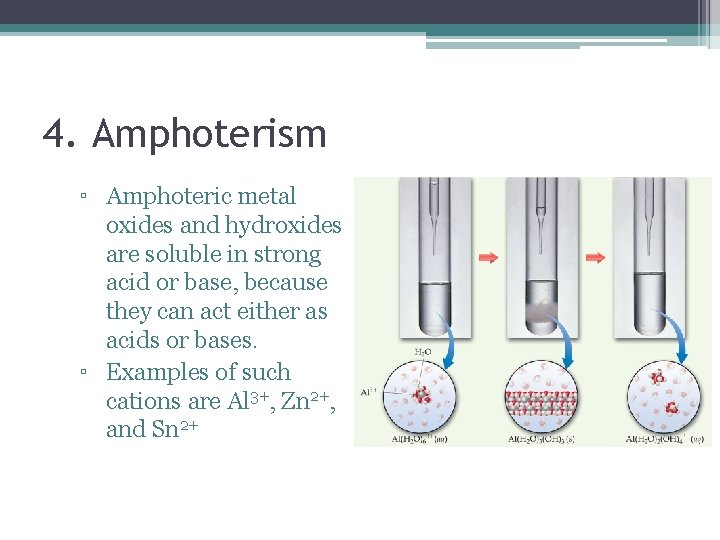
4. Amphoterism ▫ Amphoteric metal oxides and hydroxides are soluble in strong acid or base, because they can act either as acids or bases. ▫ Examples of such cations are Al 3+, Zn 2+, and Sn 2+

Complex Ions Cont. • The formation of these complex ions increases the solubility of these salts.

Precipitation and Separation of Ions

Will a Precipitate Form? • In a solution, ▫ If Q = Ksp, the system is at equilibrium and the solution is saturated. ▫ If Q < Ksp, more solid will dissolve until Q = Ksp. ▫ If Q > Ksp, the salt will precipitate until Q = Ksp.

Sample Calculation • The Ksp for Ba. SO 4 is 1. 1 x 10¯ 10 and that for Ba. Se. O 4 is 2. 8 x 10¯ 11. A 1. 0 M solution of Ba. Cl 2 is added slowly to a solution that is 1. 0 x 10¯ 4 M in sodium sulfate, Na 2 SO 4 and 1. 0 x 10¯ 4 M in sodium selenate, Na 2 Se. O 4. What is the approximate percentage of one anion has precipitated at the point which the second anion just begins to precipitate? (Assume the addition of the Ba. Cl 2 solution does not change the overall solution volume to any significant degree. )
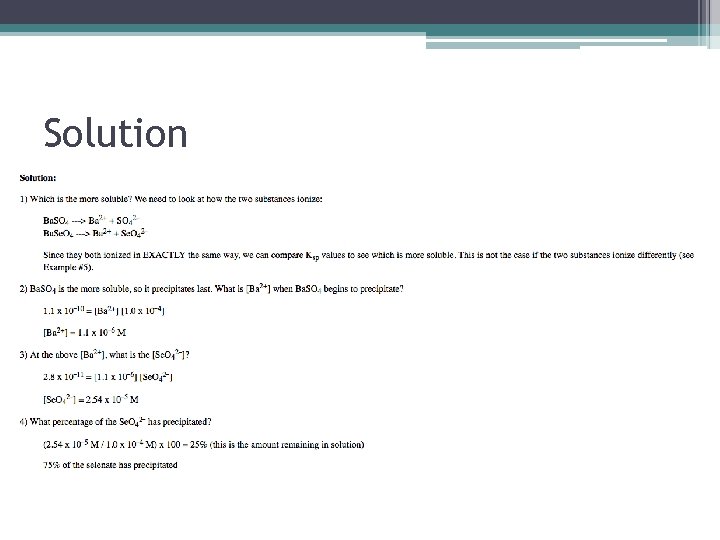
Solution

Selective Precipitation of Ions One can use differences in solubilities of salts to separate ions in a mixture.

Selective Precipitation Practice Problems • Visit the website and walk through the sample calculations • Try to solve the problem on your own and then walk through the worked out solution • http: //www. chemteam. info/Equilibrium/Selecti ve. Precip. html
Practice Problems Ch. 17 49, 51, 53, 55, 57, 59 69, 70, 75, 76

Closure • Which problem did you find most challenging today?
- Slides: 29