Solubility Particles move back and forth from being

Solubility

Particles move back and forth from being dissolved to leaving a solution—equilibrium is reached when this movement is balanced. A point is reached where adding more solute to a solution will prevent the solute from dissolving in the solution.

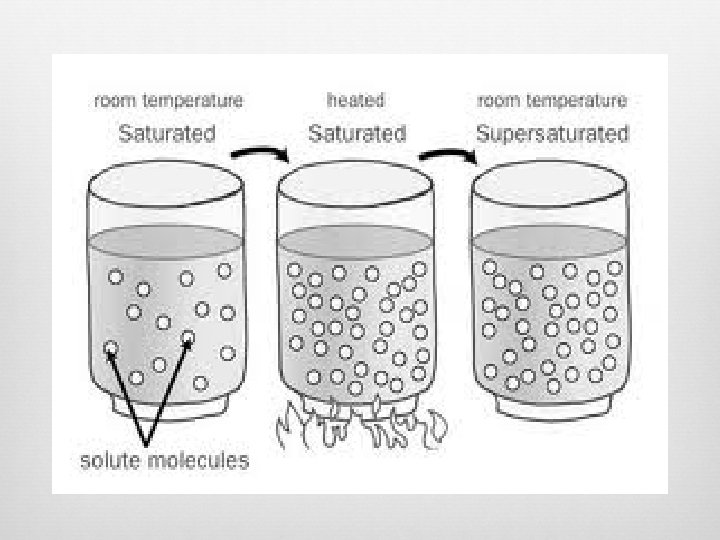
Types of Solutions 1) Saturated Maximum amount of solute that can be dissolved in a solvent Certain temperature and pressure 2) Unsaturated Solution with LESS solute than the maximum solute amount at a certain temperature and pressure More solute can be added and dissolved in the solution 3) Supersaturated Contains MORE solute than the maximum solute amount

Like Dissolves Like • Water is polar. It has an unequal charge distribution. • Polar and ionic substances will dissolve in water. • Nonpolar substances will not dissolve in water, but will dissolve in nonpolar solvents. • Liquids that will not dissolve in each other are immiscible.

Solubility How much solute dissolves in a solution to produce a saturated solution Temperature and Pressure dependent

Solubility Diagrams

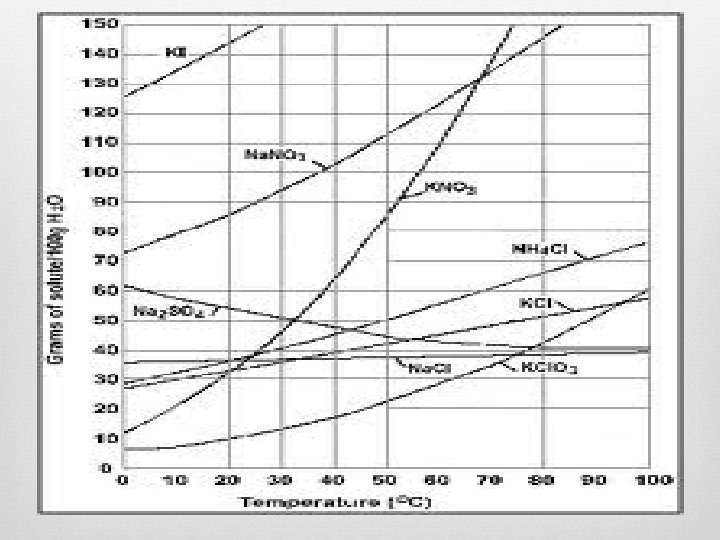

Solubility Rules How do we determine which compounds will dissolve in water or not? Based on data, patterns have been observed and helped develop the rules we follow

Which compounds are soluble in water? Ba. Cl 2 Pb. Cr. O 4 Na 2 S
Your Turn……. Na. Cl KBr KNO 3 Fe(OH)3
- Slides: 12