Solubility Explain how solutions are formed Define solubility

Solubility § Explain how solutions are formed. § Define solubility and interpret solubility graphs. § Describe factors that affect the concentration of solutions. § Compare and contrast solubility of solid, liquid, and gaseous matter.

Water and solutions § A solution is a mixture of two or more substances that is homogeneous at the molecular level. § Homogeneous means the particles are evenly distributed.

Water as a mixture § Muddy water is heterogeneous because it contains larger particles of soil or plant debris.

Colloids § Colloids are mixtures, and look like solutions, but their particles are too small to settle to the bottom of their container over time. § Examples of colloids are mayonnaise, egg whites, and gelatin.

Suspensions § In a mixture called a suspension the particles can range widely in size. § Muddy water, will settle when it is left still for a period of time.

Water and solutions § A solution contains at least two components: a solvent, and a solute. § The solvent is the part of a mixture that is present in the greatest amount. Which of these is the solvent?

Water and solutions § When the solute particles are evenly distributed throughout the solvent, we say that the solute has dissolved.
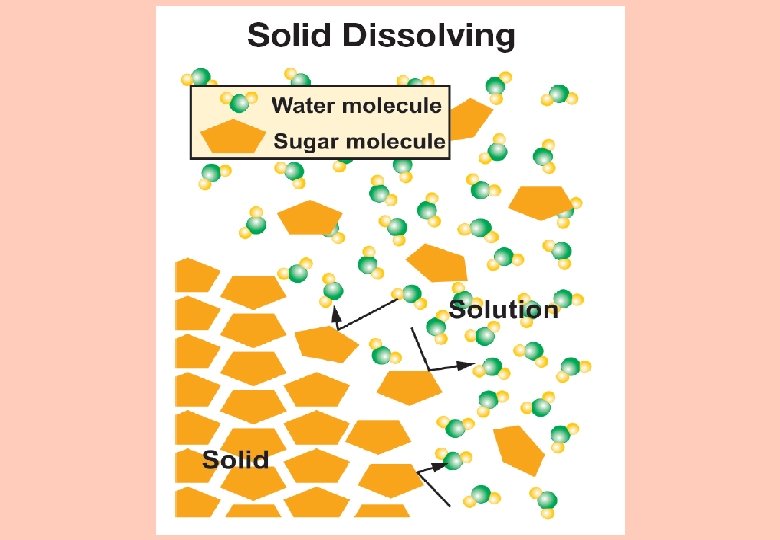

Solubility § The term solubility means the amount of solute (if any) that can be dissolved in a volume of solvent. Is there a limit for how much seltzer (solute) can dissolve in water (solvent)?

Solubility § Chalk and talc do not have solubility values. § These substances are insoluble in water because they do not dissolve in water.

Solubility § A solution is saturated if it contains as much solute as the solvent can dissolve. § Any solute added in excess of the substance’s solubility will not dissolve.

Solving Problems How much salt can dissolve in 200 m. L of water at 25 °C? 1. Looking for: § …grams of solute 2. Given § … v = 200 m. L; T = 25 C

Solving Problems 3. Relationships: § § Solubility table for reference 35. 9 g of salts in 100 m. L water at 25 °C 4. Solution § § § …if there are 35. 9 g salt/100 m. L water, then “x” g/ 200 m. L water = 71. 8 g salts are need in 200 m. L of water

Solubility § A solution is saturated if it contains as much solute as the solvent can hold. § An examples of a saturated solution is air. § Air can be saturated with water. (We call it humidity!)

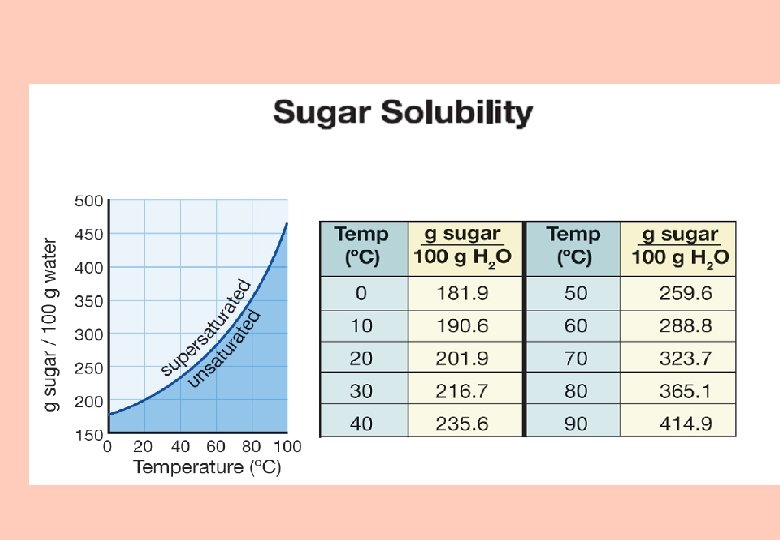
Solubility graphs § Solubility values for three solutes are plotted in this temperature-solubility graph.

Solubility § For something to dissolve in water, the water molecules need to break the bonds between the solute molecules. § Water dissolves each substance differently because the chemical bond strengths between atoms found in different solutes are not the same.

Equilibrium § When a solute like sugar is mixed with a solvent like water, two processes are actually going on continuously. § Molecules of solute dissolve and go into solution. § Molecules of solute come out of solution and become “undissolved. ” § When the rate of dissolving equals the rate of coming out of solution, we say equilibrium has been reached.

Solutions of gases and liquids § Some solutions have a gas as the solute. § In carbonated soda, the fizz comes from dissolved carbon dioxide gas (CO 2).

Solutions of gases and liquids § When temperature increases, the solubility of gases in liquid decreases.

Solutions of gases and liquids § The variety and no. or organisms is controlled somewhat by the relationship between dissolved oxygen and temperature.

Solutions of gases and liquids § Oil and vinegar salad dressing separates because oil is not soluble in vinegar (mostly water). § Liquids that are not soluble in water may be soluble in other solvents.

Solubility rules § A set of solubility rules helps predict when an ionic compound is soluble or insoluble.
- Slides: 23