Solubility Equilibrium The Solubility Product Constant Most salts

Solubility Equilibrium

The Solubility Product Constant Most salts (ionic compounds) readily dissociate in water E. g. Ca 3(PO 4)2(s) ↔ 3 Ca 2+(aq) + 2 PO 43 -(aq) For the dissociation equilibrium equation: AB(s) ↔ b. B+(aq) + c. C-(aq) Ksp = [B+(aq)]b[C-(aq)]c • Recall: solids are not included in Ksp • Common Ksp values at SATP are listed on p. 802 • Remember: Ksp is temperature dependent

Magnitude of Ksp >> 1 Product favoured Mostly ions Soluble ionic compounds Ksp << 1 Reactant favoured Very low [ions] Ionic compounds with low solubility

E. g. 1: Calculate Ksp for magnesium fluoride at 25◦C, given a solubility of 1. 72 X 10 -3 g/100 m. L

E. g. 2: Calculate the solubility of zinc hydroxide, in mol/L, given a Ksp of 7. 7 X 10 -17 at 25◦C.

Practice! • P. 486 #1 -4 • Ksp worksheet (1 st page only)
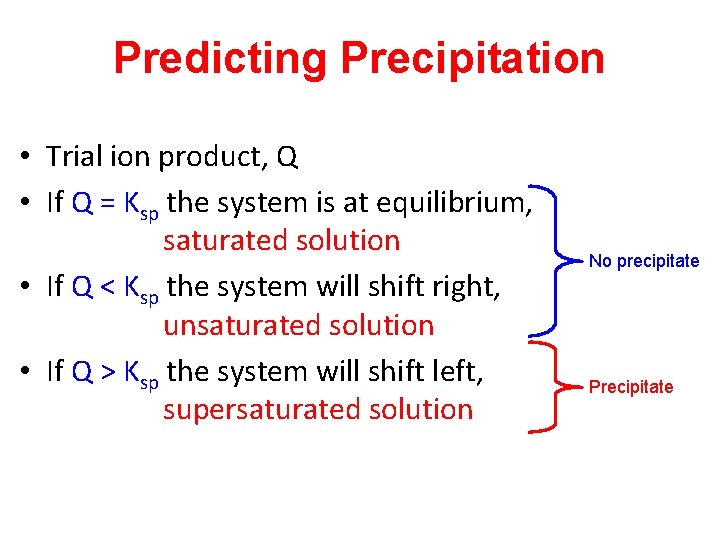
Predicting Precipitation • Trial ion product, Q • If Q = Ksp the system is at equilibrium, saturated solution • If Q < Ksp the system will shift right, unsaturated solution • If Q > Ksp the system will shift left, supersaturated solution No precipitate Precipitate

The Common Ion Effect • Equilibrium can be shifted by dissolving a salt with a common ion or a compound that reacts with one of the ions in solution (Le Chatelier’s Principle) • E. g. Na. Cl(s) ↔ Na+(aq) + Cl-(aq) • Explain the effect on the equilibrium system above when: – – HCl(aq) is added Na. NO 3(s) is added Ca(NO 3)2(aq) is added Pb(NO 3)2(s) is added

The Common Ion Effect E. g. What is the solubility of Pb. Cl 2(s) in a 0. 20 mol/L Na. Cl(aq) solution at SATP

Practice! • • P. 489 #5 -6 P. 492 #7 -12 P. 493 #1 -11 Second page of worksheet
- Slides: 10