Solubility Equilibrium Solubility Product Constant Ksp solubility product

Solubility Equilibrium

Solubility Product Constant Ksp: solubility product constant The product of the concentrations of the ions each raised to a power equal to the coefficient of the ion in the dissociation equation Smaller the numerical value, lower the solubility Pb. Cr. O 4 Pb 2+ + Cr. O 42 Ksp = [Pb 2+][Cr. O 42 -]= 1. 8 x 10 -14

Predicting Precipitants Ksp can be used to predict if precipitants will form If the product of the concentration of the two ions in soln is greater than the Ksp, a precipitant will form 0. 50 L of 0. 002 M Ba(NO 3)2 is mixed with 0. 50 L of 0. 008 MNa 2 SO 4. Insoluble compound: Ba. SO 4 Ksp: 1. 1 x 10 -10 (found in a chart) [ions] are ½ because the volume was doubled [Ba 2+]x[SO 42 -] (0. 001 M)(0. 004 M)=4 x 10 -6>1. 1 x 10 -10 (Ksp) Precipitant forms
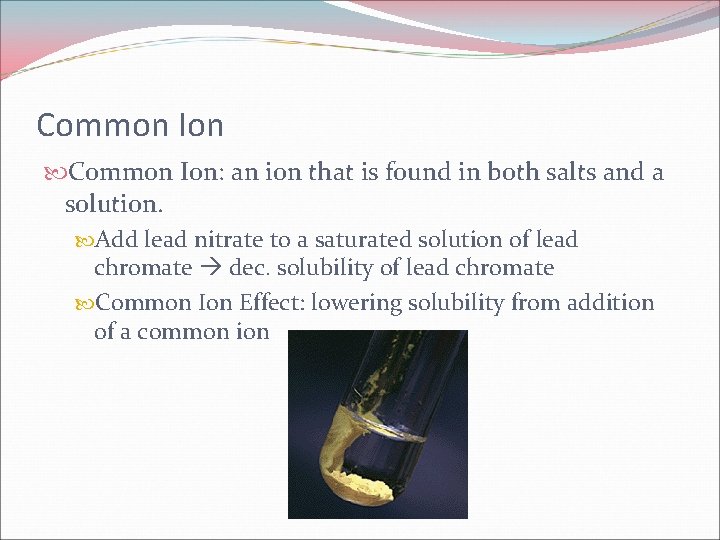
Common Ion: an ion that is found in both salts and a solution. Add lead nitrate to a saturated solution of lead chromate dec. solubility of lead chromate Common Ion Effect: lowering solubility from addition of a common ion

Entropy and Free Energy

Free Energy and Spontaneous Rxn. Free Energy: energy that is available to do work Engines ~30% efficient Most Living things only reach 70% efficiency Spontaneous Reaction: occurs naturally and favors the formation of products Produce substantial amounts of products at equilibrium and release free energy Nonspontaneous Reaction: doesn’t favor the formation of products nor give substantial amounts of products

Spontaneous and Nonspontaneous terms do not refer to reaction rates Some spontaneous reactions go so slowly they appear nonspontaneous Changes in temperature or pressure may be needed to determine if a reaction is spontaneous Coupling a nonspontaneous and a spontaneous reaction can allow a nonspontaneous reaction to occur

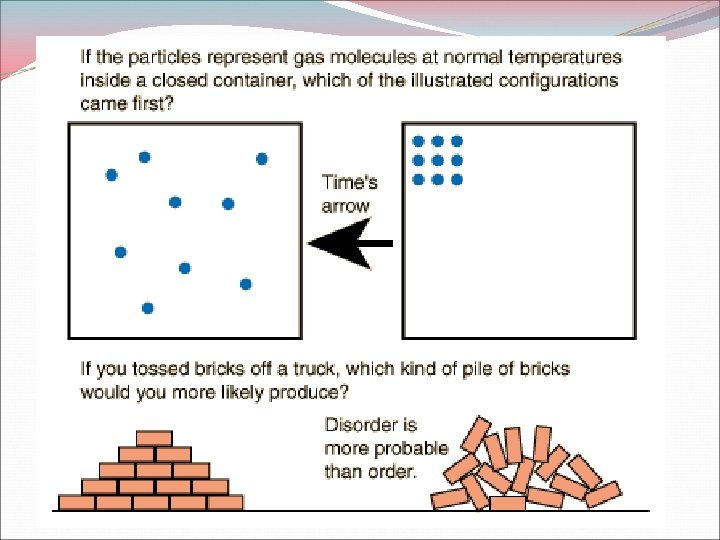
Entropy Measure of disorder Law of Disorder: the natural tendency is for a system to more in the direction of maximum disorder and randomness Increase in Entropy favors spontaneous reactions Decreases in Entropy favors nonspontaneous reactions

The size and direction of enthalpy changes and entropy changes together determine whether a reaction is spontaneous Favors products and free energy is releases Spontaneous if Exothermic: decrease enthalpy (H) Increase entropy ( S) Nonspontaneous if: Endothermic: increase enthalpy (H) Decrease entropy ( S)

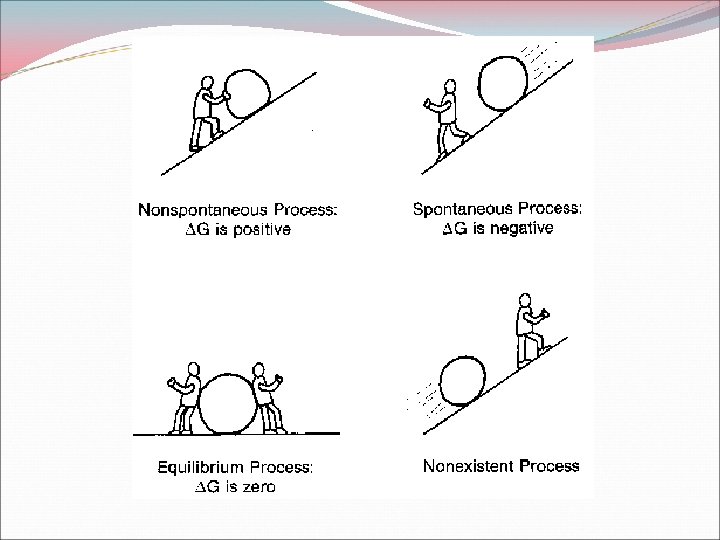
Gibbs Free Energy Maximum amount of energy that can be coupled to another process to do useful work. G = H - T S Temperature (K) G is negative: spontaneous process G is positive: nonspontaneous process
- Slides: 12