SOLUBILITY EQUILIBRIUM Solubility Product Constant Ksp CommonIon Effect

SOLUBILITY EQUILIBRIUM Solubility Product Constant, Ksp Common-Ion Effect Predicting Precipitation Free Energy of Dissolution
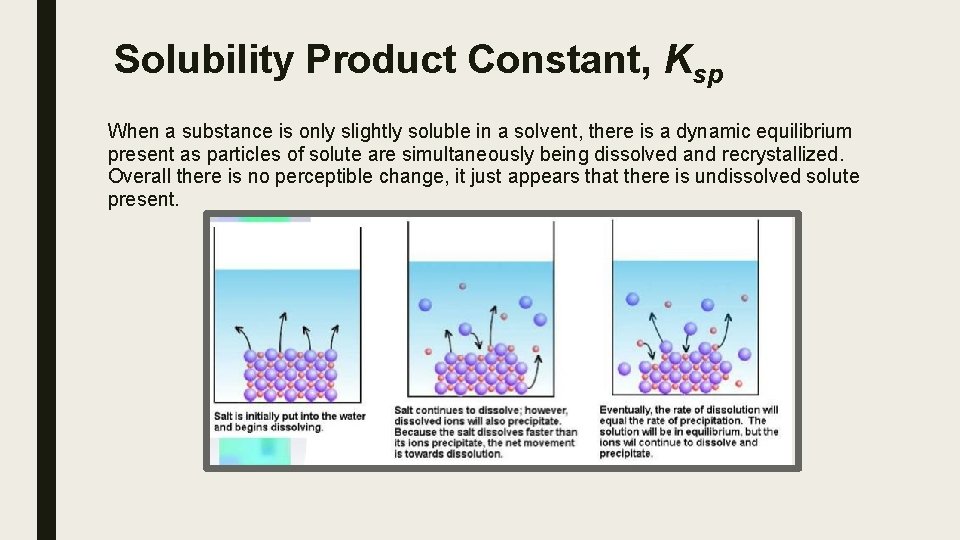
Solubility Product Constant, Ksp When a substance is only slightly soluble in a solvent, there is a dynamic equilibrium present as particles of solute are simultaneously being dissolved and recrystallized. Overall there is no perceptible change, it just appears that there is undissolved solute present.

For example barium sulfate is only slightly soluble in water: SO 42 -(aq) Ba. SO 4(s) ⇌ Ba 2+(aq) + This is an example of a heterogeneous equilibrium so the equilibrium constant is: Ksp = [Ba 2+(aq)][SO 42 -(aq)] Ksp is called the solubility product constant. It is temperature dependent. Generally, as temperature increases so too does Ksp. Molar solubility is the amount of solute in moles that can dissolve in one solvent. The unit for molar solubility is mol/L. litre of

Example 1: The solubility of Ag 2 CO 3 is 1. 3 x 10 -4 mol/L at 25 o. C. Calculate Ksp for Ag 2 CO 3.

Example 2: The value of Ksp for barium fluoride is 1. 5 x 10 -6 at 25 o. C. What is its molar solubility at 25 o. C?
■ AP practice questions

Common-Ion Effect Suppose you have an ionic compound dissolved in water. What will happen to its solubility if a solution containing one of the ions is added to it? For example: Suppose you have a solution of Ag. Cl (Ksp = 1. 8 x 10 -10) and you added some sodium chloride to it. What would happen to Ag. Cl’s solubility? Explain using Le Chatelier’s principle. Ø Sodium chloride is very soluble in water. Therefore, adding Na. Cl increases the [Cl-] causing a shift to the left. A shift to the left means that more Ag. Cl precipitates out, ie. less Ag. Cl is dissolved. Its solubility has decreased.

Example: Ksp for Ag. Cl is 1. 8 x 10 -10. Determine the molar solubility of Ag. Cl in a 0. 2 M solution of Na. Cl.
■ AP practice questions

p. H and Solubility p. H changes can affect the solubility of a substance. Use Le. Chatelier’s principle to analyze these situations. For example:

Another example:

Predicting Precipitation Ksp values can help you determine if a solution is saturated or unsaturated by comparing its value to the ion product, Qsp is the same idea as reaction quotient, except you are observing a dissolving process rather than a reaction. ■ If Qsp<Ksp, then: more solute can dissolve, no precipitation occurs ■ If Qsp=Ksp, then: unlikely! ■ If Qsp>Ksp, then: precipitation occurs

Example: A solution is prepared by adding 750. 0 m. L of 4. 00 x 10 -3 M Ce(NO 3)3 to 300. 0 m. L of 0. 0200 M KIO 3. Will Ce(IO 3)3 (Ksp = 1. 9 x 10 -10) precipitate from this solution?

Free Energy of Dissolution ■ Processes are said to be spontaneous if they occur without any outside intervention (note: spontaneous ≠ rapid). ■ “Non-spontaneous” events can occur only as long as they receive some sort of outside assistance. ■ For example: Ice melts spontaneously at room temperature while water boils only if constantly heated (ie non-spontaneous)

Spontaneity is influenced by 2 factors: 1. The enthalpy change (DH) Ø exothermic reactions tend to be spontaneous since lowering the energy of the system tends to be favored 2. The change in entropy (DS) Ø Entropy (S) is a measure of disorder; the higher the degree of disorder, the greater the entropy Ø entropy is temperature dependent; entropy increase is directly proportional to the amount of heat added Ø 2 kinds of disorder: ■ ■ positional disorder: based on the arrangement of particles in space thermal disorder: depending on how the total energy is distributed among the particles Example: In each case, use qualitative reasoning to decide which of the following will have the largest entropy: 1) A mole of ice at 0 o. C, or a mole of water at the same temp. 2) Solid ammonium chloride, or a solution of the salt in water.

Processes that increase entropy tend to be spontaneous (the second law of thermodynamics indicates that any spontaneous process must increase the entropy of the universe)

Free Energy Two factors – enthalpy and entropy – determine whether or not a given physical or chemical event will be spontaneous. Sometimes these two factors work together. For example, an exothermic reaction (DH < 0) that increases randomness (DS > 0)will always be spontaneous, while an endothermic reaction (DH > 0) that decreases randomness (DS < 0)will always be non-spontaneous. When ENTHALPY AND ENTROPY EFFECTS OPPOSE EACH OTHER, the result of the interaction between them is not immediately obvious. However, we can analyze the NET EFFECT of the two factors through the use of anothermodynamic quantity, called GIBB’S FREE ENERGY, G. Gibb’s Free Energy is defined as: G = H – TS. For a CHANGE at CONSTANT TEMPERTURE and PRESSURE, the equation becomes: Note: we will be looking more in-depth at entropy and Gibb’s Free Energy in Unit 9.
■ AP practice questions
- Slides: 18