Solubility Equilibria Solubility Product Constant Ksp for saturated

Solubility Equilibria Solubility Product Constant Ksp for saturated solutions at equilibrium 1
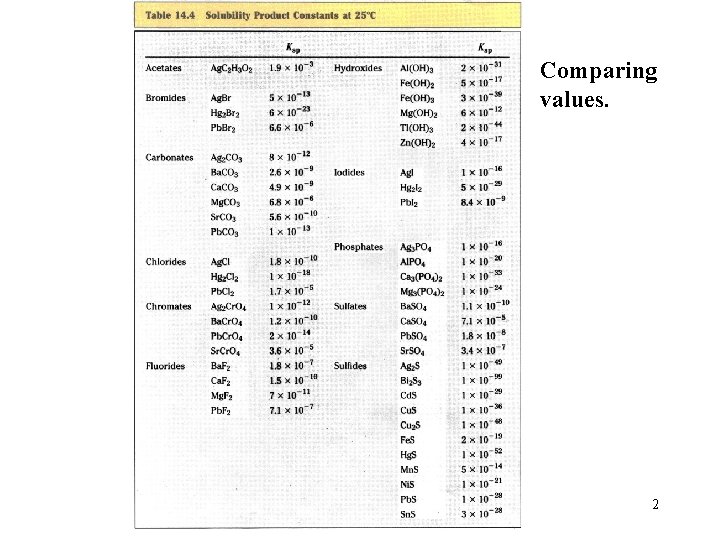
Comparing values. 2
![Solubility Product (Ksp) = [products]x/[reactants]y but. . . reactants are in solid form, so Solubility Product (Ksp) = [products]x/[reactants]y but. . . reactants are in solid form, so](http://slidetodoc.com/presentation_image_h2/3c20363c1a1a5bc4bf6996ac948d7bc9/image-3.jpg)
Solubility Product (Ksp) = [products]x/[reactants]y but. . . reactants are in solid form, so Ksp=[products]x i. e. A 2 B 3(s) 2 A 3+ + 3 B 2– Given: Ksp=[A 3+]2 [B 2–]3 Ag. Br(s) Ag+ + Br– In a saturated solution @25 o. C, the [Ag+] = [Br– ]= 5. 7 x 10– 7 M. Determine the Ksp value. 3

Problem: A saturated solution of silver chromate was to found contain 0. 022 g/L of Ag 2 Cr. O 4. Find Ksp Eq. Expression: Ag 2 Cr. O 4 (s) 2 Ag+ + Cr. O 42– Ksp = [Ag+]2[Cr. O 42–] So we must find the concentrations of each ion and then solve for Ksp. 4

Problem: A saturated solution of silver chromate was to contain 0. 022 g/L of Ag 2 Cr. O 4. Find Ksp Eq. Expression: Ag 2 Cr. O 4 (s) 2 Ag+ + Cr. O 42– Ksp = [Ag+]2[Cr. O 42–] Ag+: 0. 022 g Ag 2 Cr. O 4 L Cr. O 4– 2: 0. 022 g Ag 2 Cr. O 4 L 1. 33 x 10– 4 6. 63 x 10– 5 5

Problems working from Ksp values. Given: Ksp for Mg. F 2 is 6. 4 x 10– 9 @ 25 o. C Find: solubility in mol/L and in g/L Ksp = [Mg 2+][F–]2 Mg. F 2(s) Mg 2+ + 2 F– I. C. E. N/A N/A 0 +x +x 0 +2 x Ksp= [x][2 x]2 = 4 x 3 6. 4 x 10– 9 = 4 x 3 now for g/L: 7. 3 x 10– 2 6
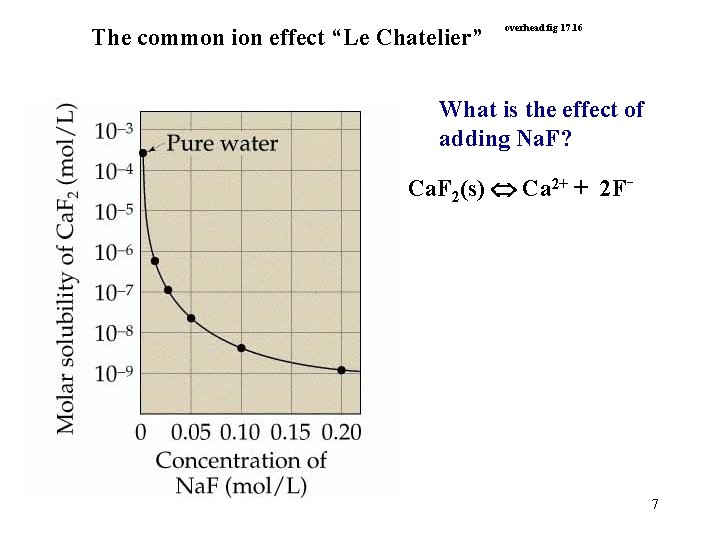
The common ion effect “Le Chatelier” overhead fig 17. 16 What is the effect of adding Na. F? Ca. F 2(s) Ca 2+ + 2 F- 7
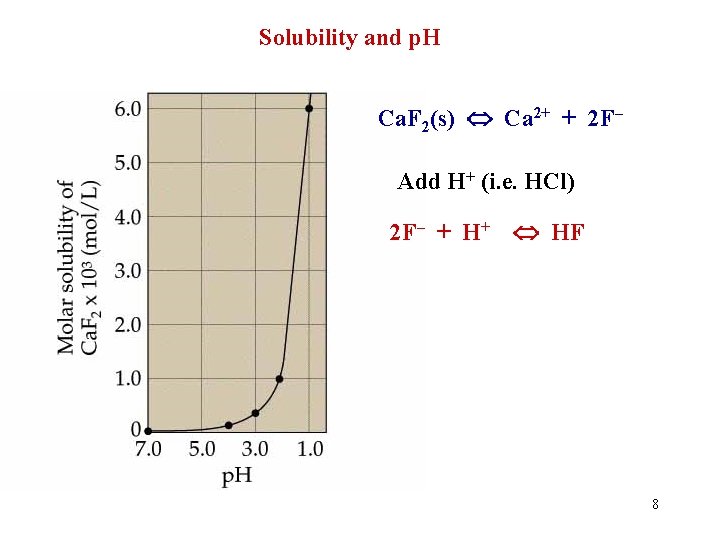
Solubility and p. H Ca. F 2(s) Ca 2+ + 2 F– Add H+ (i. e. HCl) 2 F– + H+ HF 8

Solubility and p. H Mg(OH)2(s) Mg 2+ + 2 OH– Adding Na. OH? Adding HCl? 9

The common ion effect “Le Chatelier” Why is Ag. Cl less soluble in sea water than in fresh water? Ag. Cl(s) Ag+ + Cl– Seawater contains Na. Cl 10

Problem: The solubility of Ag. Cl in pure water is 1. 3 x 10– 5 M. What is its solubility in seawater where the [Cl–] = 0. 55 M? (Ksp of Ag. Cl = 1. 8 x 10– 10) I. C. E. Ag. Cl(s) Ag+ + Cl– N/A 0 0. 55 N/A +x +x N/A +x 0. 55 + x Ksp= [x][0. 55 + x] Ksp= [Ag+][Cl–] try dropping this x Ksp = 0. 55 x 1. 8 x 10– 10 = 0. 55 x x = 3. 3 x 10– 10 = [Ag+]=[Ag. Cl] “Ag. Cl is much less soluble in seawater” 11

more Common ion effect: a. What is the solubility of Ca. F 2 in 0. 010 M Ca(NO 3)2? Ksp(Ca. F 2) = 3. 9 x 10– 11 Ca. F 2(s) Ca 2+ + 2 F– [Ca 2+] [F–] I. 0. 010 0 C. +x +2 x 2 x E. 0. 010 + x Ksp=[Ca 2+][F-]2 Ksp= [0. 010 + x][2 x]2 [0. 010][2 x]2 = 0. 010(4 x 2) 3. 9 x 10– 11 = 0. 010(4 x 2) x = 3. 1 x 10– 5 M Ca 2+ from Ca. F 2 so = M of Ca. F 2 Now YOU determine the solubility of Ca. F 2 in 0. 010 M Na. F. 12
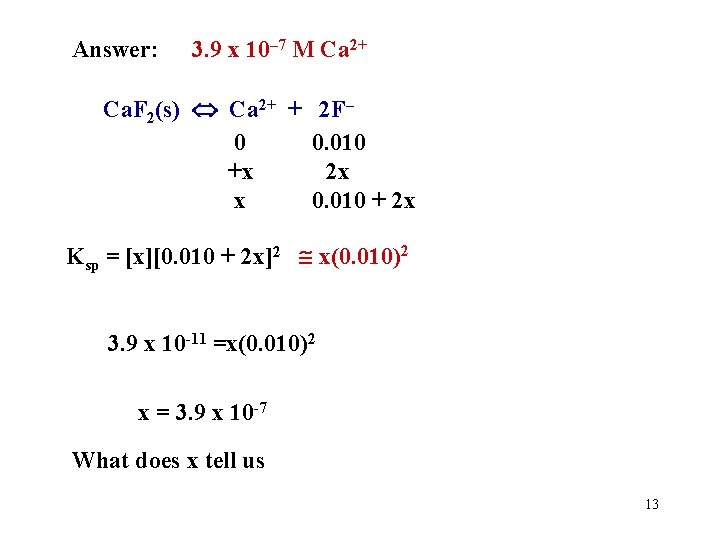
Answer: 3. 9 x 10– 7 M Ca 2+ Ca. F 2(s) Ca 2+ + 2 F– 0 0. 010 +x 2 x x 0. 010 + 2 x Ksp = [x][0. 010 + 2 x]2 x(0. 010)2 3. 9 x 10 -11 =x(0. 010)2 x = 3. 9 x 10 -7 What does x tell us 13

Reaction Quotient (Q): will a ppt. occur? Use Q (also called ion product) and compare to Ksp Q < Ksp reaction goes Q = Ksp Equilibrium Q > Ksp reaction goes 14

Problem: A solution is 1. 5 x 10– 6 M in Ni 2+. Na 2 CO 3 is added to make the solution 6. 0 x 10– 4 M in CO 32–. Ksp(Ni. CO 3) = 6. 6 x 10– 9. Will Ni. CO 3 ppt? We must compare Q to Ksp. Ni. CO 3 Ni 2+ + CO 32– Ksp = [Ni 2+][CO 32–] Q = [1. 5 x 10– 6][6. 0 x 10– 4] = 9. 0 x 10– 10 Q < Ksp no ppt. 15

Problem: 0. 50 L of 1. 0 x 10– 5 M Pb(OAc)2 is combined with 0. 50 L of 1. 0 x 10– 3 M K 2 Cr. O 4. a. Will a ppt occur? Ksp(Pb. Cr. O 4) = 1. 8 x 10– 14 Pb(OAc)2(aq) + K 2 Cr. O 4(aq) Pb. Cr. O 4(s) + 2 KOAc(aq) then: Pb. Cr. O 4(s) Pb 2+ + Cr. O 42– Ksp= [Pb 2+][Cr. O 42–] [Pb 2+]: 0. 50 L 1 L [Cr. O 42 -]: 0. 50 L 1 L 5. 0 x 10– 6 5. 0 x 10 -4 Q = [5. 0 x 10 -6][5. 0 x 10 -4] = 2. 5 x 10 -9 compare to Ksp: Q > Ksp so a ppt. will occur 16

b. find the Eq. conc. of Pb 2+ remaining in solution after the Pb. Cr. O 4 precipitates. Ksp(Pb. Cr. O 4) = 1. 8 x 10– 14 Since [Pb 2+] = 5. 0 x 10 -6 and [Cr. O 42 -] = 5. 0 x 10 -4 and there is a 1: 1 stoichiometry, Pb 2+ is the limiting reactant. Pb. Cr. O 4(s) Pb 2+ + Cr. O 42– 0 5. 0 x 10 -4 - 5. 0 x 10 -6 = 5. 0 x 10 -4 I. (after ppt. ) +x +x C. x 5. 0 x 10– 4 + x E. Ksp = [x][5. 0 x 10– 4 + x] Try dropping the “+ x” term. 1. 8 x 10– 14 = x(5. 0 x 10 -4) x = [Pb 2+] = 3. 6 x 10– 11 This is the concentration of Pb 2+ remaining in solution. 17

Complex ion formation: Ag. Cl(s) Ag+ + Cl– Ksp= 1. 8 x 10– 10 Ag+(aq) + NH 3(aq) Ag(NH 3)+(aq) + NH 3(aq) Ag(NH 3)2+(aq) Ag+(aq) + 2 NH 3(aq) {Ag(NH 3)2}+(aq) formation or stability constant: For Cu 2+: Cu 2+ + 4 NH 3 [Cu(NH 3)4]2+(aq) K 1 x K 2 x K 3 x K 4 = Kf = 6. 8 x 1012 18

Solubility and complex ions: Problem: How many moles of NH 3 must be added to dissolve 0. 050 mol of Ag. Cl in 1. 0 L of H 2 O? (Ksp. Ag. Cl = 1. 8 x 10– 10 ; Kf[Ag(NH 3)2]+ = 1. 6 x 107) Ag. Cl(s) Ag+ + Cl– Ag+(aq) + 2 NH 3(aq) Ag(NH 3)2+(aq) sum of RXNS: Ag. Cl(s) + 2 NH 3 Ag(NH 3)2+(aq) + Cl– = 2. 9 x 10– 3 Now use Koverall to solve the problem: Koverall= 2. 9 x 10– 3 = [NH 3]eq = 0. 93 but. . . How much NH 3 must we add? [NH 3]total= 0. 93 + (2 x 0. 050) = 1. 03 M 2 ammonia’s for each Ag+ 19

Fractional Precipitation: “ppting” one ion at a time. Compounds must have different Ksp values (i. e. different solubilities) Example: Ksp Cd. S = 3. 9 x 10– 29 and Ksp. Ni. S = 3. 0 x 10– 21 ? Which will ppt. first? least soluble Problem: A solution is 0. 020 M in both Cd 2+ and Ni 2+. Just before Ni. S begins to ppt. , what conc. of Cd 2+ will be left in solution? Approach: Find conc. of S 2– ion when Ni 2+ just begins to ppt. since Cd 2+ will already be ppting. Then use this S 2– conc. to find Cd 2+. Ni. S(s) Ni 2+ + S 2– Ksp= 3. 0 x 10– 21 = [0. 020][S 2–] = 1. 5 x 10– 19 M when Ni 2+ just begins to ppt. So: Cd. S(s) Cd 2+ + S 2– Ksp= 3. 9 x 10– 29 = [Cd 2+][1. 5 x 10– 19] [Cd 2+] = 2. 6 x 10– 10 M when Ni. S starts to ppt. 20
- Slides: 20