SOLUBILITY EQUILIBRIA SCH 4 UO The Solubility Product

SOLUBILITY EQUILIBRIA SCH 4 U/O

The Solubility Product Constant, Ksp • Many important ionic compounds are only slightly soluble in water and equations are written to represent the equilibrium between the compound and the ions present in a saturated aqueous solution. • The solubility product constant, Ksp, is the product of the concentrations of the ions involved in a solubility equilibrium, each raised to a power equal to the stoichiometric coefficient of that ion in the chemical equation for the equilibrium.

The Solubility Equilibrium Equation and Ksp • Mn. Xm(s) n. Mm+(aq) + m. Xn−(aq) Ksp = [Mm+]n[Xn−]m • Ca. F 2(aq) Ca 2+ (aq) + 2 F- (aq) Ksp = [Ca 2+][F-]2 Ksp = 5. 3 x 10 -9 • As 2 S 3(s) 2 As 3+(aq) + 3 S 2 - (aq) Ksp = [As 3+]2[S 2 -]3

Sample Ksp values at 25°C

Practice Time! • For each of the following salts below, write a balanced equation showing the dissociation in water. Then, write the Ksp expression for the salt. • A) Iron (III) hydroxide, Fe(OH)3 • B) Nickel sulfide, Ni. S • C) Silver chromate, Ag 2 Cr. O 4 • D) Zinc carbonate, Zn. CO 3 • E) Calcium fluoride, Ca. F 2

Ksp and Molar Solubility • The solubility product constant (Ksp) is related to the solubility of an ionic solute. • Molar Solubility - the molarity of a solute in a saturated aqueous solution • Ksp and Molar solubility are not the same thing. • Calculating solubility equilibria fall into two categories: • determining a value of Ksp from experimental data • calculating equilibrium concentrations when Ksp is known.

1. Calculating Ksp from Molar Solubility It is found that 1. 2 x 10 -3 mol of lead (II) iodide, Pb. I 2, dissolves in 1. 0 L of aqueous solution at 25 o. C. What is the K at this temperature? sp

1. Calculating Ksp from Molar Solubility It is found that 1. 2 x 10 -3 mol of lead (II) iodide, Pb. I 2, dissolves in 1. 0 L of aqueous solution at 25 o. C. What is the K at this temperature? sp Pb. I 2 Pb 2+ + 2 IKsp = [Pb 2+][I-]2 Ksp = [1. 2 x 10 -3] [2(1. 2 x 10 -3)]2 Ksp = 6. 9 x 10 -9 2 iodide ions form, so you must multiply molarity by 2!

2. Calculating Molar Solubility from Ksp • Calculate the molar solubility of silver chromate, Ag 2 Cr. O 4, in water from Ksp = 1. 1 x 10 -12 for Ag 2 Cr. O 4.

2. Calculating Molar Solubility from Ksp • Calculate the molar solubility of silver chromate, Ag 2 Cr. O 4, in water from Ksp = 1. 1 x 10 -12 for Ag 2 Cr. O 4 (s) 2 Ag+ Cr. O 42 - 0 0 +2 x +x 2 x x Ksp = [Ag+]2[Cr. O 4 -2] 1. 1 x 10 -12 = (x)(2 x)2 1. 1 x 10 -12 = 4 x 3 x = 6. 5 x 10 -5 M = [Ag 2 Cr. O 4] = [Cr. O 42 -] [Ag+] = 2(6. 5 x 10 -5)

The Common Ion Effect in Solubility Equilibria • The common ion effect also affects solubility equilibria. • Le Châteliers principle is followed for the shift in concentration of products and reactants upon addition of either more products or more reactants to a solution. • The solubility of a slightly soluble ionic compound is lowered when a second solute that furnishes a common is added to the solution. • for example, addition of Na. Cl to the solubility equilibrium of solid Pb. Cl 2 decreases the solubility of Pb. Cl 2: Pb. Cl 2(s) Pb 2+(aq) + 2 Cl−(aq) addition of Cl− shifts the equilibrium to the left
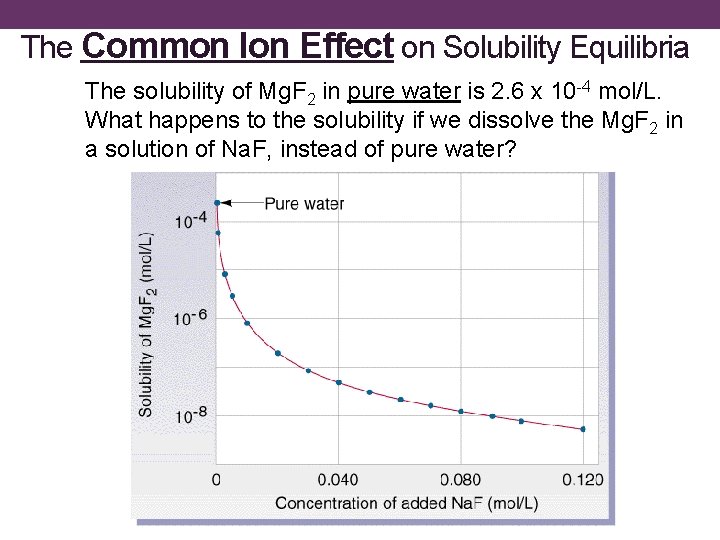
The Common Ion Effect on Solubility Equilibria The solubility of Mg. F 2 in pure water is 2. 6 x 10 -4 mol/L. What happens to the solubility if we dissolve the Mg. F 2 in a solution of Na. F, instead of pure water?

3. Common Ion Effect Calculation • Calculate the molar solubility of Ca. F 2 in 0. 100 M Na. F at 25°C

3. Common Ion Effect Calculation • Calculate the molar solubility of Ca. F 2 in 0. 100 M Na. F at 25°C Ca. F 2(s) Ca 2+(aq) + 2 F−(aq) [Ca. F 2] [Ca 2+] [F−] 0 0. 100 +x +2 x x 0. 100 + 2 x Ksp = [Ca 2+][F−]2 Ksp = (x)(0. 100 + 2 x)2

The Effect of p. H on Solubility • for insoluble ionic hydroxides, the higher the p. H, the lower the solubility of the ionic hydroxide ü and the lower the p. H, the higher the solubility ü higher p. H = increased [OH−] • M(OH)n(s) Mn+(aq) + n. OH−(aq) for insoluble ionic compounds that contain anions of weak acids, the lower the p. H, the higher the solubility M 2(CO 3)n(s) 2 Mn+(aq) + n. CO 32−(aq) H 3 O+(aq) + CO 32− (aq) HCO 3− (aq) + H 2 O(l)

Determining Whether Precipitation Occurs • precipitation will occur when the concentrations of the • • ions exceed the solubility of the ionic compound if we compare the reaction quotient, Q, for the current solution concentrations to the value of Ksp, we can determine if precipitation will occur ü Q = Ksp, the solution is saturated, no precipitation ü Q < Ksp, the solution is unsaturated, no precipitation ü Q > Ksp, the solution would be above saturation, the salt above saturation will precipitate some solutions with Q > Ksp will not precipitate unless disturbed – these are called supersaturated solutions

precipitation occurs if Q > Ksp a supersaturated solution will precipitate if a seed crystal is added

Determining Whether Precipitation Occurs- An Example The concentration of calcium ion in blood plasma is 0. 0025 M. If the concentration of oxalate ion is 1. 0 x 10 -7 M, do you expect calcium oxalate to precipitate? Ksp = 2. 3 x 10 -9. Three steps: (1) Determine the initial concentrations of ions. (2) Evaluate the reaction quotient Qip. (3) Compare Qsp with Ksp.

Determining Whether Precipitation Occurs The concentration of calcium ion in blood plasma is 0. 0025 M. If the concentration of oxalate ion is 1. 0 x 10 -7 M, do you expect calcium oxalate, Ca. C 2 O 4, to precipitate? Ksp = 2. 3 x 10 -9. Qsp = [Ca 2+][C 2 O 42 -] = (0. 0025)(1. 0 x 10 -7) Qsp = 2. 5 x 10 -10 Qsp < Ksp therefore, no precipitate will form!!!

Determining Whether Precipitation Occurs • The concentration of calcium ion in blood plasma is 0. 0025 M. If the concentration of oxalate ion is 1. 0 x 10 -7 M, do you expect calcium oxalate, Ca. C 2 O 4, to precipitate? Ksp = 2. 3 x 10 -9.

Determining Whether Precipitation Occurs In applying the precipitation criteria, the effect of dilution when solutions are mixed must be considered. Example: A 250. 0 m. L sample of 0. 0012 M Pb(NO 3)2 (aq) is mixed with 150. 0 m. L of 0. 0640 M Na. I (aq). Should precipitation of Pb. I 2 (s), Ksp = 7. 1 x 10 -9, occur? Calculate new concentrations in total volume of 400 mls = 0. 4 L [Pb 2+] = (0. 250 L)(0. 0012 M)/(0. 400 L) = 7. 5 x 10 -4 M [I-] = (0. 150 L)(0. 0640 M)/(0. 400 L) = 0. 024 M Qsp = [Pb 2+][I-]2 = (7. 5 x 10 -4)(0. 024)2 = 4. 32 x 10 -7 Qsp > Ksp therefore a precipitate will form!

Comparing Solubilities • Which salt will be the most soluble in water. Molar solubility 1. Cu. S Ksp = 8. 5 x 10 -45 x = 9. 2 x 10 -23 Ksp = x 2 2. Ag 2 S Ksp = 1. 6 x 10 -49 x = 3. 4 x 10 -17 Ksp = (2 x)2 x = 4 x 3 3. Bi 2 S 3 Ksp = 1. 1 x 10 -73 x = 1. 0 x 10 -15 Ksp = (2 x)2(3 x)3 = 108 x 5 Most Soluble = highest molar solubility!!!! Do not use Ksp!!

Selective Precipitation • a solution containing several different cations can often be separated by addition of a reagent that will form an insoluble salt with one of the ions, but not the others • a successful reagent can precipitate with more than one of the cations, as long as their Ksp values are significantly different

Selective Precipitation a) The first precipitate to form when Ag. NO 3(aq) is added to an aqueous solution containing Cland I- is yellow Ag. I(s). b) Essentially all the I- has precipitated before the precipitation of white Ag. Cl(s) begins. Ag. Cl (s) Ag+ (aq) + Cl- (aq) Ag. I (s) Ag+ (aq) + I- (aq) Ksp = 1. 8 x 10 -10 Ksp = 8. 5 x 10 -17

Selective Precipitation Calculation • The magnesium and calcium ions present in seawater can be separated by selective precipitation with KOH. What is the minimum [OH−] necessary to just begin to precipitate Mg 2+ (with [0. 059]) from seawater? Ksp for Mg(OH)2 is 2. 06 x 10 -13

Selective Precipitation Calculation • The magnesium and calcium ions present in seawater can be separated by selective precipitation with KOH. What is the minimum [OH−] necessary to just begin to precipitate Mg 2+ (with [0. 059]) from seawater?
![Selective Precipitation Calculation #2 • What is the [Mg 2+] when Ca 2+ (with Selective Precipitation Calculation #2 • What is the [Mg 2+] when Ca 2+ (with](http://slidetodoc.com/presentation_image_h2/e688363bf816c7542add1244c29b08c4/image-27.jpg)
Selective Precipitation Calculation #2 • What is the [Mg 2+] when Ca 2+ (with [0. 011]) just begins to precipitate from seawater? Ksp for Ca(OH)2 is 4. 58 x 10 -6
![Selective Precipitation Calculation #2 • What is the [Mg 2+] when Ca 2+ (with Selective Precipitation Calculation #2 • What is the [Mg 2+] when Ca 2+ (with](http://slidetodoc.com/presentation_image_h2/e688363bf816c7542add1244c29b08c4/image-28.jpg)
Selective Precipitation Calculation #2 • What is the [Mg 2+] when Ca 2+ (with [0. 011]) just begins to precipitate from seawater?
![when Ca 2+ just begins to precipitate out, the [Mg 2+] has dropped from when Ca 2+ just begins to precipitate out, the [Mg 2+] has dropped from](http://slidetodoc.com/presentation_image_h2/e688363bf816c7542add1244c29b08c4/image-29.jpg)
when Ca 2+ just begins to precipitate out, the [Mg 2+] has dropped from 0. 059 M to 4. 8 x 10 -10 M

Qualitative Inorganic Analysis • an analytical scheme that utilizes selective precipitation to identify the ions present in a solution is called a qualitative analysis scheme üwet chemistry • a sample containing several ions is • subjected to the addition of several precipitating agents addition of each reagent causes one of the ions present to precipitate out
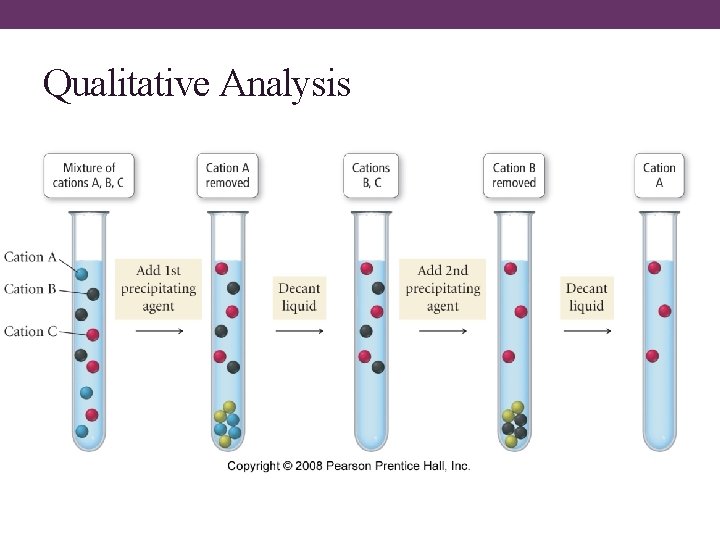
Qualitative Analysis
32

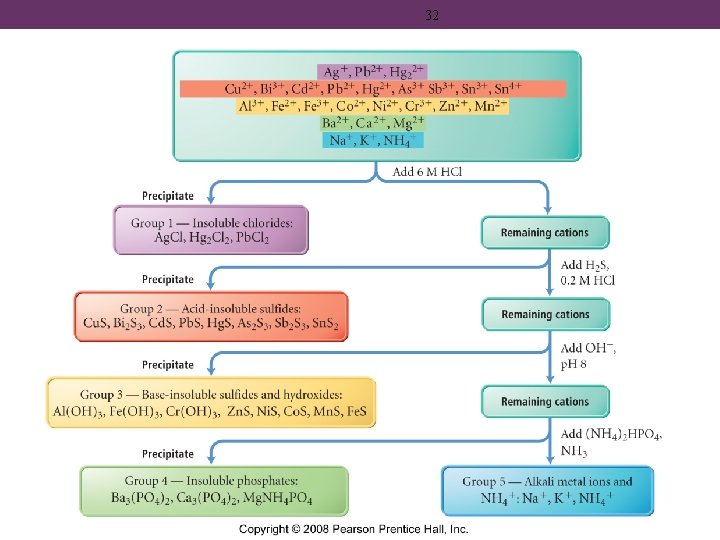
Group 1 • group one cations are Ag+, Pb 2+, and Hg 2+ • all these cations form compounds with Cl− that are insoluble in water • as long as the concentration is large enough • Pb. Cl 2 may be borderline • molar solubility of Pb. Cl 2 = 1. 43 x 10 -2 M • precipitated by the addition of HCl

Group 2 • group two cations are Cd 2+, Cu 2+, Bi 3+, Sn 4+, As 3+, Pb 2+, Sb 3+, and Hg 2+ • all these cations form compounds with HS− and S 2− that are insoluble in water at low p. H • precipitated by the addition of H 2 S in HCl

35 Group 3 • group three cations are Fe 2+, Co 2+, Zn 2+, Mn 2+, Ni 2+ precipitated as sulfides; as well as Cr 3+, Fe 3+, and Al 3+ precipitated as hydroxides • all these cations form compounds with S 2− that are insoluble in water at high p. H • precipitated by the addition of H 2 S in Na. OH

Group 4 • group four cations are Mg 2+, Ca 2+, Ba 2+ • all these cations form compounds with PO 43− that are insoluble in water at high p. H • precipitated by the addition of (NH 4)2 HPO 4

Group 5 • group five cations are Na+, K+, NH 4+ • all these cations form compounds that are soluble in water – they do not precipitate • identified by the color of their flame
- Slides: 37