Solubility Equilibria Lead II iodide precipitates when potassium
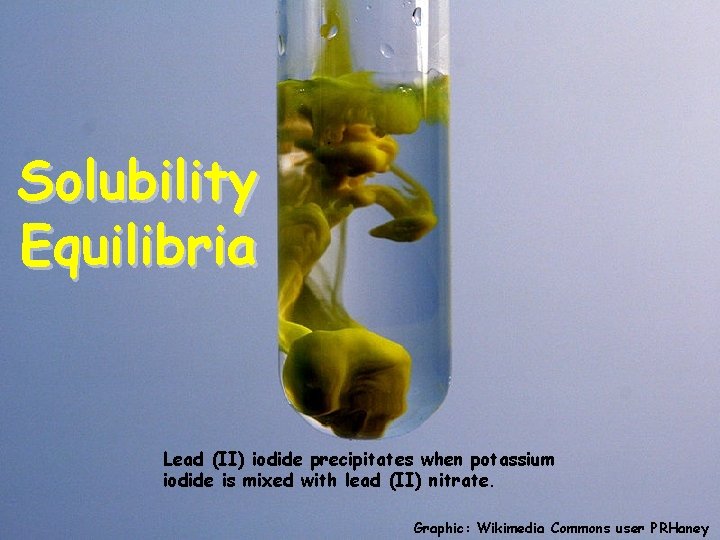
Solubility Equilibria Lead (II) iodide precipitates when potassium iodide is mixed with lead (II) nitrate. Graphic: Wikimedia Commons user PRHaney
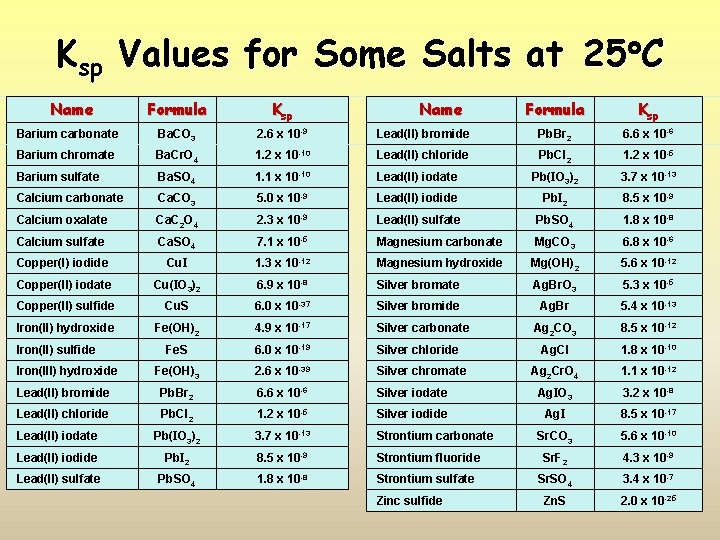
Ksp Values for Some Salts at 25 C Name Formula Ksp Barium carbonate Ba. CO 3 2. 6 x 10 -9 Barium chromate Ba. Cr. O 4 Barium sulfate Formula Ksp Lead(II) bromide Pb. Br 2 6. 6 x 10 -6 1. 2 x 10 -10 Lead(II) chloride Pb. Cl 2 1. 2 x 10 -5 Ba. SO 4 1. 1 x 10 -10 Lead(II) iodate Pb(IO 3)2 3. 7 x 10 -13 Calcium carbonate Ca. CO 3 5. 0 x 10 -9 Lead(II) iodide Pb. I 2 8. 5 x 10 -9 Calcium oxalate Ca. C 2 O 4 2. 3 x 10 -9 Lead(II) sulfate Pb. SO 4 1. 8 x 10 -8 Calcium sulfate Ca. SO 4 7. 1 x 10 -5 Magnesium carbonate Mg. CO 3 6. 8 x 10 -6 Copper(I) iodide Cu. I 1. 3 x 10 -12 Magnesium hydroxide Mg(OH)2 5. 6 x 10 -12 Copper(II) iodate Cu(IO 3)2 6. 9 x 10 -8 Silver bromate Ag. Br. O 3 5. 3 x 10 -5 Copper(II) sulfide Cu. S 6. 0 x 10 -37 Silver bromide Ag. Br 5. 4 x 10 -13 Iron(II) hydroxide Fe(OH)2 4. 9 x 10 -17 Silver carbonate Ag 2 CO 3 8. 5 x 10 -12 Fe. S 6. 0 x 10 -19 Silver chloride Ag. Cl 1. 8 x 10 -10 Fe(OH)3 2. 6 x 10 -39 Silver chromate Ag 2 Cr. O 4 1. 1 x 10 -12 Lead(II) bromide Pb. Br 2 6. 6 x 10 -6 Silver iodate Ag. IO 3 3. 2 x 10 -8 Lead(II) chloride Pb. Cl 2 1. 2 x 10 -5 Silver iodide Ag. I 8. 5 x 10 -17 Lead(II) iodate Pb(IO 3)2 3. 7 x 10 -13 Strontium carbonate Sr. CO 3 5. 6 x 10 -10 Lead(II) iodide Pb. I 2 8. 5 x 10 -9 Strontium fluoride Sr. F 2 4. 3 x 10 -9 Lead(II) sulfate Pb. SO 4 1. 8 x 10 -8 Strontium sulfate Sr. SO 4 3. 4 x 10 -7 Zn. S 2. 0 x 10 -25 Iron(II) sulfide Iron(III) hydroxide Name Zinc sulfide

Solving Solubility Problems For the salt Ag. I at 25 C, Ksp = 1. 5 x 10 -16 Ag. I(s) Ag+(aq) + I-(aq) I O O C +x +x E x x 1. 5 x 10 -16 = x 2 x = solubility of Ag. I in mol/L = 1. 2 x 10 -8 M

Solving Solubility Problems For the salt Pb. Cl 2 at 25 C, Ksp = 1. 6 x 10 -5 Pb. Cl 2(s) Pb 2+(aq) + 2 Cl-(aq) I O O C +x +2 x E x 2 x 1. 6 x 10 -5 = (x)(2 x)2 = 4 x 3 x = solubility of Pb. Cl 2 in mol/L = 1. 6 x 10 -2 M

Solving Solubility with a Common Ion For the salt Ag. I at 25 C, Ksp = 1. 5 x 10 -16 What is its solubility in 0. 05 M Na. I? Ag. I(s) Ag+(aq) + I-(aq) I O 0. 05 C +x 0. 05+x E x 0. 05+x 1. 5 x 10 -16 = (x)(0. 05+x) (x)(0. 05) x = solubility of Ag. I in mol/L = 3. 0 x 10 -15 M
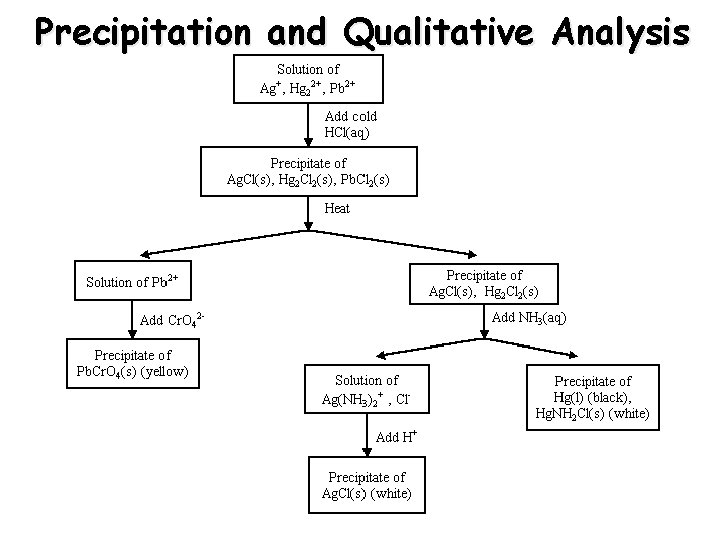
Precipitation and Qualitative Analysis

Complex Ions A Complex ion is a charged species composed of: 1. A metallic cation 2. Ligands – Lewis bases that have a lone electron pair that can form a covalent bond with an empty orbital belonging to the metallic cation
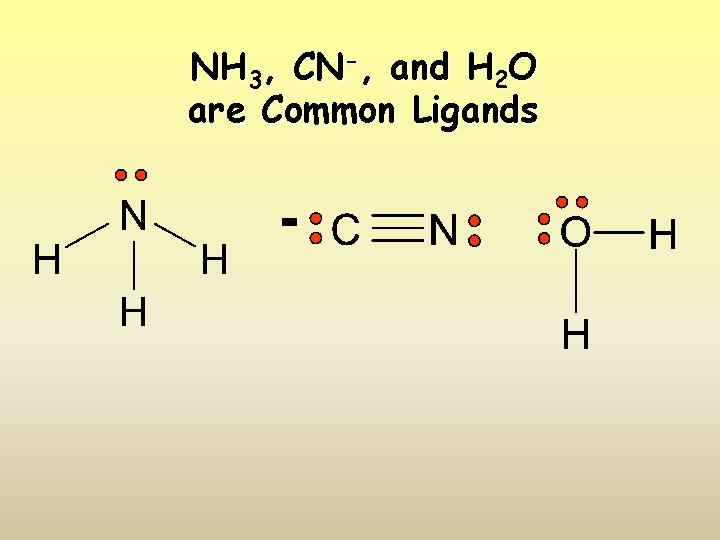
NH 3, CN-, and H 2 O are Common Ligands

Coordination Number q Coordination number refers to the number of ligands attached to the cation q 2, 4, and 6 are the most common coordination numbers Coordination Example(s) number 2 Ag(NH 3)2+ 4 Co. Cl 42 - 6 Co(H 2 O)62+ Cu(NH 3)42+ Ni(NH 3)62+
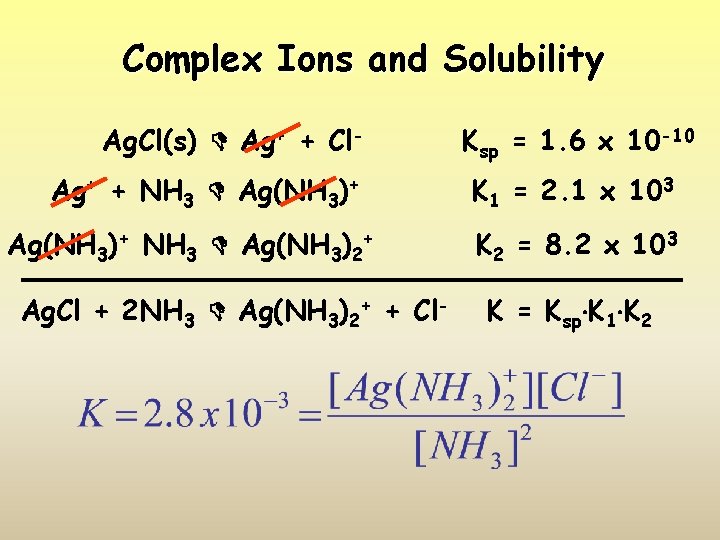
Complex Ions and Solubility Ag. Cl(s) Ag+ + Cl. Ag+ + NH 3 Ag(NH 3)+ NH 3 Ag(NH 3)2+ Ag. Cl + 2 NH 3 Ag(NH 3)2+ + Cl- Ksp = 1. 6 x 10 -10 K 1 = 2. 1 x 103 K 2 = 8. 2 x 103 K = Ksp K 1 K 2
- Slides: 10