Solubility Equilibria involve the dissolution or precipitation of

Solubility Equilibria -- involve the dissolution or precipitation of “insoluble” salts Consider a saturated solution of a typical salt: MN(s) Mx+(aq) + Nx–(aq) MN 2(s) M 2 x+(aq) + 2 Nx–(aq) etc. For these cases, the solubility-product constant is equal to (respectively): Ksp = [Mx+] [Nx–] Ksp = [M 2 x+] [Nx–]2 Ksp expressions NEVER have denominators because… the “reactant” is always a solid, and we never write solids in equil. eqs.
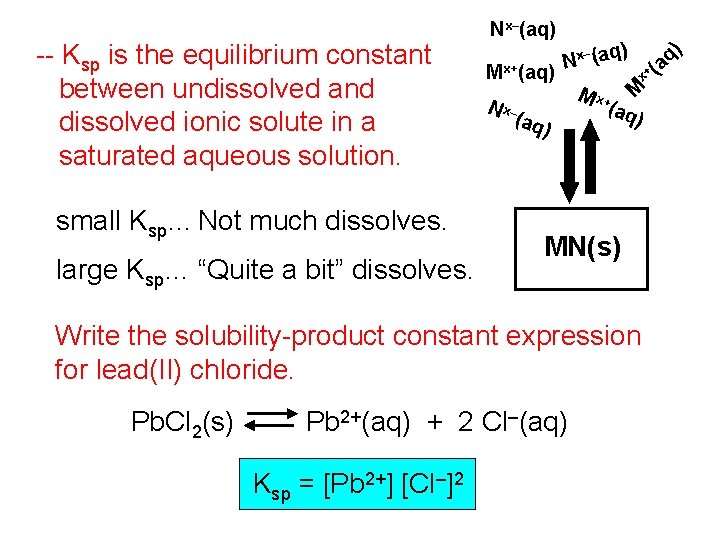
small Ksp… Not much dissolves. large Ksp… “Quite a bit” dissolves. N x– (aq ) q) (a + Mx+(aq) ) – Nx (aq Mx -- Ksp is the equilibrium constant between undissolved and dissolved ionic solute in a saturated aqueous solution. Nx–(aq) M x+ (aq ) MN(s) Write the solubility-product constant expression for lead(II) chloride. Pb. Cl 2(s) Pb 2+(aq) + 2 Cl–(aq) Ksp = [Pb 2+] [Cl–]2

Copper(II) azide has Ksp = 6. 3 x 10– 10. Find the solubility of Cu(N 3)2 in water, in g/L. Cu(N 3)2(s) Cu 2+(aq) + 2 N 3–(aq) Ksp = 6. 3 x 10– 10 = [Cu 2+] [N 3–]2 ** In “plain-old” Cu(N 3)2, : [N 3–] = 2 [Cu 2+] (Let [Cu 2+] = x) So… 6. 3 x 10– 10 = x (2 x)2 = 4 x 3 x = 5. 40 x 10– 4 M = [Cu 2+] From eq. at top, 5. 40 x 10– 4 M is also the [ ] of Cu(N 3)2 that dissolves. 5. 40 x 10– 4 ( ) mol Cu(N 3)2 147. 5 g L 1 mol = 8. 0 x 10– 2 g L

Find the solubility of zinc hydroxide (Ksp = 3. 0 x 10– 16) in a soln buffered at p. H = 11. 43. Zn(OH)2(s) Zn 2+(aq) + 2 OH–(aq) Ksp = 3. 0 x 10– 16 = [Zn 2+] [OH–]2 from given p. H… 3. 0 x 10– 16 = [Zn 2+] (2. 69 x 10– 3)2 [Zn 2+] = 4. 1 x 10– 11 M From eq. at top, 4. 1 x 10– 11 M is also the [ ] of Zn(OH)2 that dissolves.

Precipitation of Ions For Ba. SO 4(s) Ba 2+(aq) + SO 42–(aq). . . Ksp = [Ba 2+] [SO 42–] We could reach equilibrium from the left. . . i. e. , start with Ba. SO 4(s) or from the right. . . i. e. , start w/Ba 2+ (say, from Ba(NO 3)2(aq)) and SO 42– (say, from Na 2 SO 4(aq)) At any given time, the ion product Q = [Ba 2+] [SO 42–] If Q > Ksp. . . more ppt will form. If Q < Ksp. . . more ions will dissolve. If Q = Ksp. . . ions and ppt are in eq.
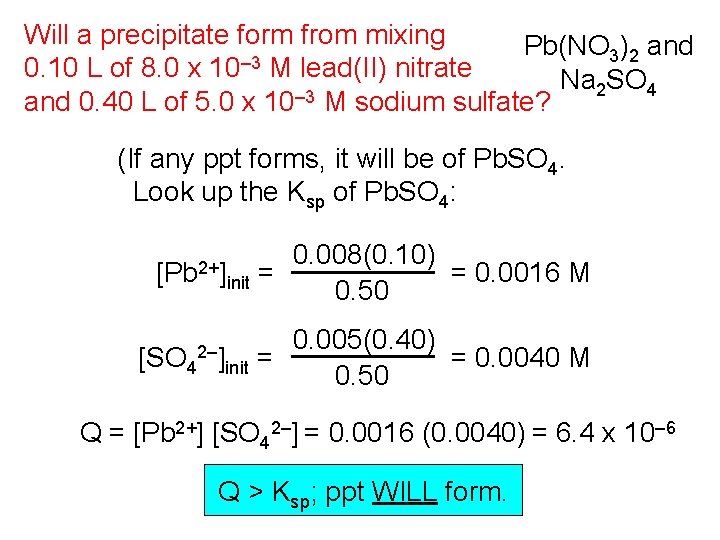
Will a precipitate form from mixing Pb(NO ) and 3 2 0. 10 L of 8. 0 x 10– 3 M lead(II) nitrate Na 2 SO 4 – 3 and 0. 40 L of 5. 0 x 10 M sodium sulfate? (If any ppt forms, it will be of Pb. SO 4. Look up the Ksp of Pb. SO 4: 6. 3 x 10– 7. ) [Pb 2+] [SO 4 2–] 0. 008(0. 10) = 0. 0016 M init = 0. 50 0. 005(0. 40) = 0. 0040 M init = 0. 50 Q = [Pb 2+] [SO 42–] = 0. 0016 (0. 0040) = 6. 4 x 10– 6 Q > Ksp; ppt WILL form.

selective precipitation: using the different solubilities of ions to separate them Say we want to separate the Ag+ and Cu 2+ in a soln that contains both. -- Add HCl. Cu. Cl 2 is… soluble, which means it won’t ppt out. Ag. Cl is… insoluble (its Ksp is 1. 8 x 10– 10) so MOST of it will ppt out when we add HCl. long-stem funnel -- Separate by. . . filtering out filter paper Ag. Cl(s) the Ag. Cl(s). Erlenmeyer flask silver Cu 2+ ions still in soln (along w/a “wee-little” Ag+) chloride

Factors That Affect Solubility 1. For solids, as temperature increases, solubility. . . 2. common-ion effect Use Le Chatelier’s principle. For example, with. . . Ca. F 2(s) Ca 2+(aq) + 2 F–(aq) If you add Ca 2+ or F–, shift . (Sol. ) 3. p. H and solubility Compounds with anions exhibiting basic properties (e. g. , Mg(OH)2 / OH–, Ca. CO 3 / CO 32–, Ca. F 2 / F–) ____ in solubility as solution becomes more acidic.
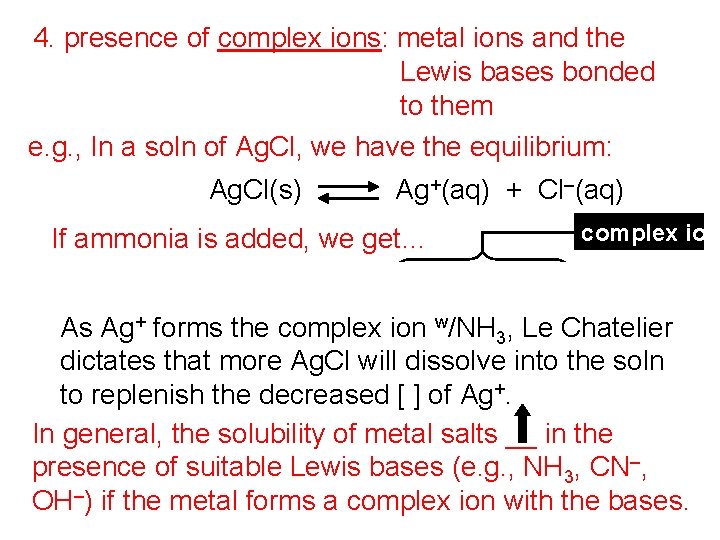
4. presence of complex ions: metal ions and the Lewis bases bonded to them e. g. , In a soln of Ag. Cl, we have the equilibrium: Ag. Cl(s) Ag+(aq) + Cl–(aq) If ammonia is added, we get… Ag+(aq) + 2 NH 3(aq) complex ion Ag(NH 3)2+(aq) As Ag+ forms the complex ion w/NH 3, Le Chatelier dictates that more Ag. Cl will dissolve into the soln to replenish the decreased [ ] of Ag+. In general, the solubility of metal salts __ in the presence of suitable Lewis bases (e. g. , NH 3, CN–, OH–) if the metal forms a complex ion with the bases.

Qualitative Analysis for Metallic Elements determines the presence or absence of a particular ion (as opposed to quantitative analysis) Steps: (1) separate ions into broad groups by solubility (2) specific ions are then separated from the group and identified by specific tests
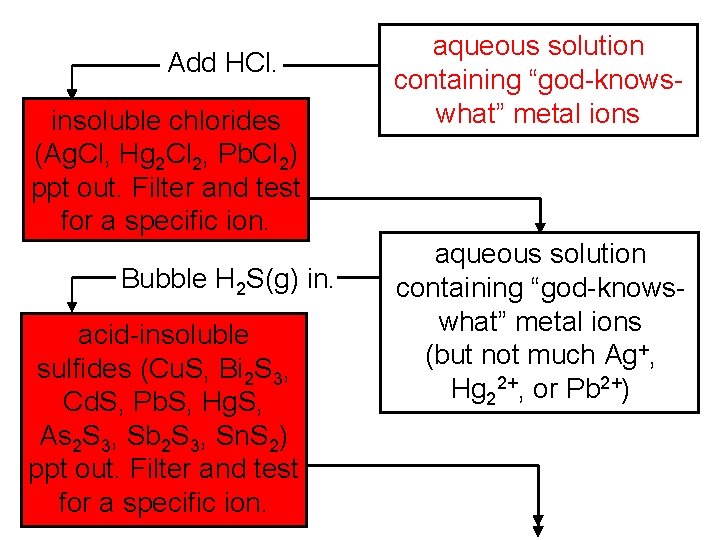
Add HCl. insoluble chlorides (Ag. Cl, Hg 2 Cl 2, Pb. Cl 2) ppt out. Filter and test for a specific ion. Bubble H 2 S(g) in. acid-insoluble sulfides (Cu. S, Bi 2 S 3, Cd. S, Pb. S, Hg. S, As 2 S 3, Sb 2 S 3, Sn. S 2) ppt out. Filter and test for a specific ion. aqueous solution containing “god-knowswhat” metal ions (but not much Ag+, Hg 22+, or Pb 2+)
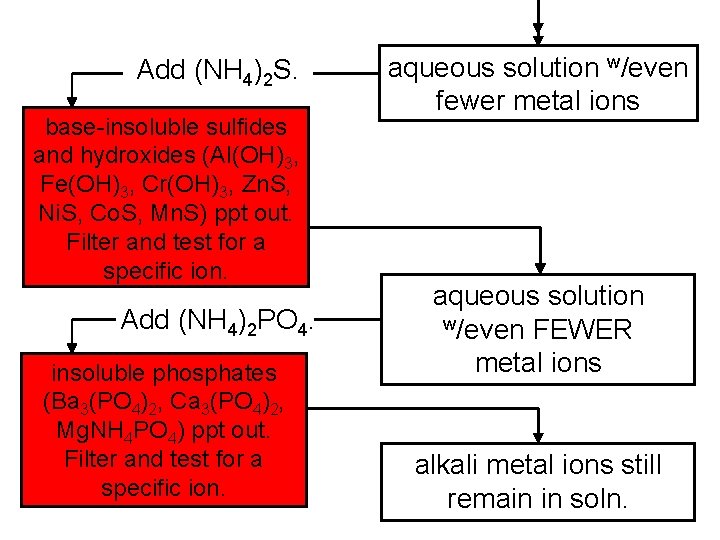
Add (NH 4)2 S. base-insoluble sulfides and hydroxides (Al(OH)3, Fe(OH)3, Cr(OH)3, Zn. S, Ni. S, Co. S, Mn. S) ppt out. Filter and test for a specific ion. Add (NH 4)2 PO 4. insoluble phosphates (Ba 3(PO 4)2, Ca 3(PO 4)2, Mg. NH 4 PO 4) ppt out. Filter and test for a specific ion. aqueous solution w/even fewer metal ions aqueous solution w/even FEWER metal ions alkali metal ions still remain in soln.

- Slides: 13