Solubility Equilibria Chapter 15 Objectives Define key terms

Solubility Equilibria Chapter 15

Objectives • Define key terms and concepts • Calculate the solubility product for a compound. • Calculate concentration and solubility using the common ion and complex ion effects. • Develop and utilize a flow chart for use in qualitative analysis.

Solubility Equilibria • Solubility Product (Ksp) • The product of the molar concentrations of the ions that make-up a compound, each raised to the power of it’s stoichiometric coefficient. • The smaller the Ksp value, the less soluble the compound is in water. Ba. CO 3 ↔ Ba 2+ + CO 32 - Ksp = [Ba 2+] [CO 32 -] Ba. F 2 ↔ Ba 2+ + 2 F- Ksp = [Ba 2+] [F-]2

Solubility Equilibria • The saturation level of a solution can be determined using Ksp and Q. • If Q < Ksp, the solution is unsaturated • If Q > Ksp, the solution is supersaturated • If Q = Ksp, the solution is saturated

Write the solubility product expression for the following salts: • Ag. Cl • Hg 2 Cl 2 • Pb 3(As. O 4)2

A liter of a solution saturated at 25°C with calcium oxalate, Ca. C 2 O 4, is evaporated to dryness, giving a 0. 0061 g residue of Ca. C 2 O 4. Calculate the solubility product constant for the salt at 25°C.

By experiment, it is found that 1. 2 x 10 -3 mole of lead (II) iodide dissolves in 1 L of aqueous solution at 25°C. What is the solubility product constant at this temperature?

Solubility Equilibria • Molar Solubility • The number of moles of solute in 1 liter of saturated solution • Moles per liter • Solubility • The number of grams of solute in 1 liter of saturates solution • Grams per liter • These both refer to solutions at a specific temperature
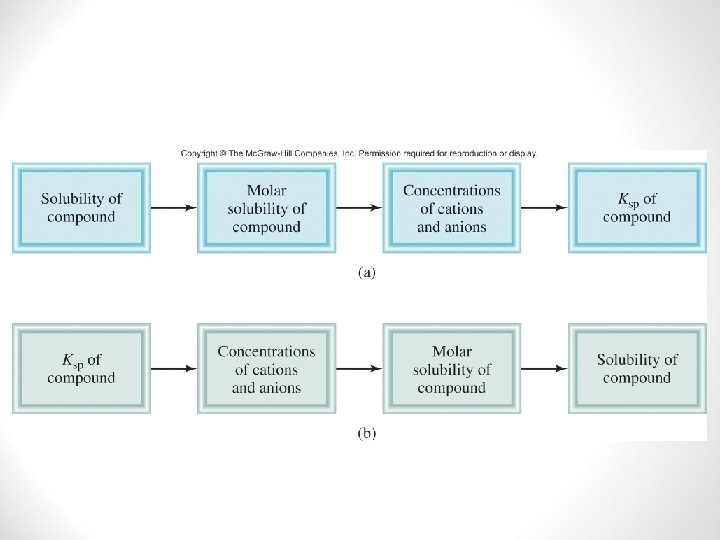

Solubility Product and Molar Solubility

Calculate the solubility in g/L of lead (II) chromate in water. The Ksp for Pb. Cr. O 4 is 1. 8 x 10 -14.
Calculate the solubility in g/L for lead (II) arsenate when dissolved in water. The Ksp for Pb 3 As. O 4)2 is 4. 0 x 10 -36.

Calculate the solubility product for potassium nitrate if the solubility of the compound in water is 133 g/L at 0°C.

If 0. 17 g of calcium fluoride is soluble in 1 L of solvent, what is the solubility product of calcium fluoride?

Precipitation Reactions • If the ion product exceeds the Ksp, a precipitate will form. Soluble Compounds Almost all salts of Na+, K+, and NH 4+ Salts of NO 3 -, Cl. O 4 -, C 2 H 3 O 2 Almost all salts of Cl-, Br-, ICompounds containing FSalts of SO 42 Insoluble Compounds Most salts of CO 32 -, PO 43 -, C 2 O 42 -, Cr. O 42 Most metal sulfides, S 2 Most metal hydroxides and oxides Exceptions Halides of Ag+, Hg 2+, Pb 2+ Fluorides of Mg 2+, Ca 2+, Sr 2+, Ba 2+, and Pb 2+ Sulfates of Ca 2+, Sr 2+, Ba 2+, Pb 2+ Exceptions Salts of NH 4+ and alkali metal cations

Common Ion Effect and Solubility • A compound can be precipitated out of solution if another compound containing a common is added to the solution. • The saturation level of a solution can be determined using Ksp and Q (ion product). • If Q < Ksp, the solution is unsaturated • If Q > Ksp, the solution is supersaturated • If Q = Ksp, the solution is saturated

Common Ion Effect and Solubility

o Which of the following compounds should most affect the solubility of lead (II) sulfate in water to which it has been added? a) Na. Cl b) Na 2 SO 4 c) Pb. S o Which of the following compounds should most affect the solubility of silver chloride in water to which it has been added? a) Na. Cl b) Na 2 SO 4 c) Pb. S

The concentration of calcium ion in blood plasma is 0. 0025 M. If the concentration of oxalate ion is 1. 0 x 10 -7 M, do you expect calcium oxalate to precipitate? Ksp for calcium oxalate is 2. 3 x 10 -9.

Ag. Cl will be dissolved into a solution with is ALREADY 0. 0100 M in chloride ion. What is the solubility of Ag. Cl? The Ksp =1. 77 x 10¯ 10

What is the solubility of Ca(OH)2 in 0. 0860 M Ba(OH)2? The Ksp for Ca(OH)2 is known to be 4. 68 x 10¯ 6.

Will silver chloride precipitate from a solution that is 0. 01 M Ag. NO 3 and 0. 01 M Na. Cl? The Ksp for silver chloride is 8. 3 x 10 -17

Will silver iodide precipitate from a solution that is 0. 0045 M Ag. NO 3 and 0. 15 M Na. I? The Ksp for silver iodide is 1. 8 x 10 -10.

Sulfate ion in solution is often determined quantitatively by precipitating it as barium sulfate. The sulfite ion may have been formed from a sulfur compound. Analysis for the amount of sulfate ion then indicates the percentage of sulfur in the compound. Is a precipitate expected to form at equilibrium when 50 m. L of 0. 0010 M barium chloride is added to 50 m. L of 0. 00010 M sodium sulfate? The solubility product constant for barium sulfate is 1. 1 x 10 -10.

A. B. C. If 1. 1 x 10 -4 g of Cr(OH)3 is added to 120 L of water at 25 °C, will all of the solute dissolve? The of chromium (III) hydroxide is Ksp = 6. 7 x 10 -31. If 4. 0 x 10 -4 g of Na. OH is added to the solution described above, will a precipitate form? Calculate the molar solubility of Cr(OH)3 in a solution buffered at p. H = 11. 00.

Complex Ion Equilibria and Solubility • Complex Ion • An ion containing a central metal cation bonded to one or more molecules or ion. • Commonly formed by transition metals • Formation Constant (Kf) • The equilibrium constant for complex ion formation • The larger the Kf, the more stable the complex
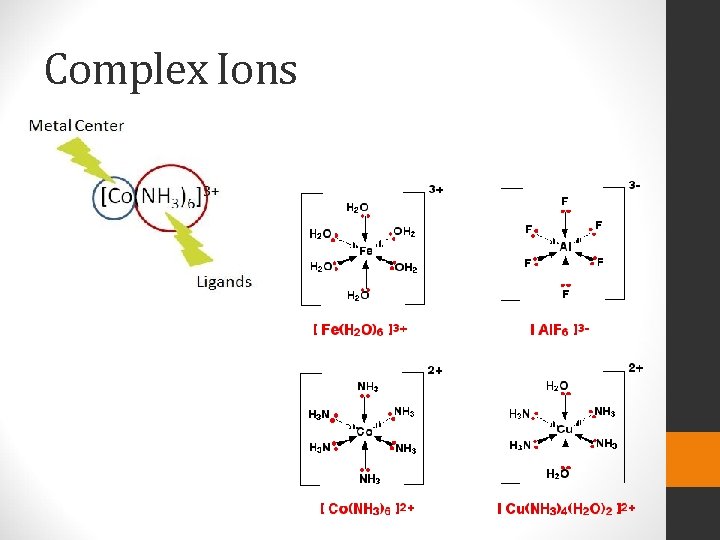
Complex Ions
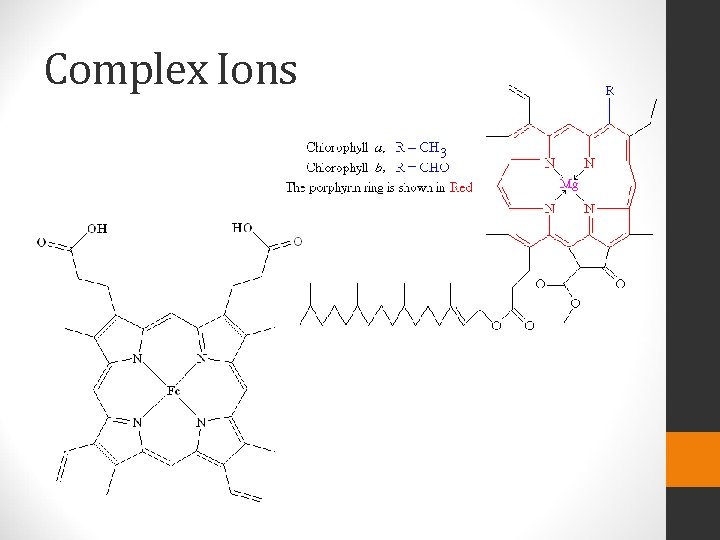
Complex Ions
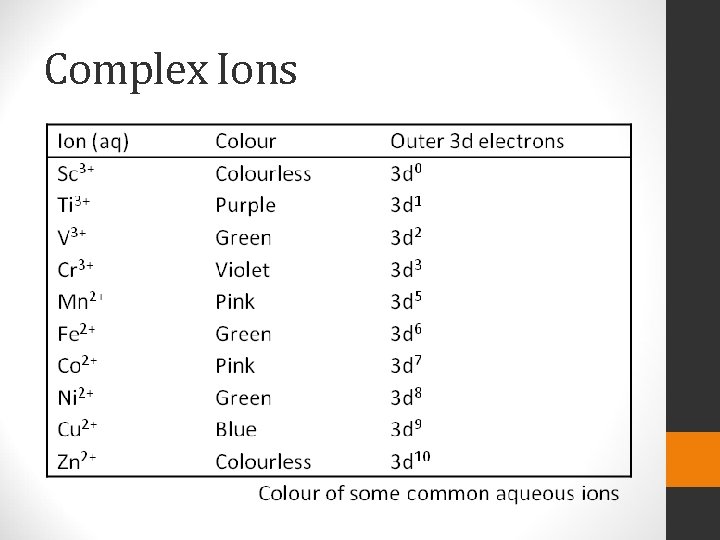
Complex Ions

Complex Ions Cu 2+ + 4 NH 32+ Cu(NH 3)42+ (solutions turns a deep blue) Ni 2+ + 6 NH 3 Ni(NH 3)62+ (Solutions is blue)
![Calculate the silver ion concentration, [Ag+], of a solution prepared by dissolving 1. 00 Calculate the silver ion concentration, [Ag+], of a solution prepared by dissolving 1. 00](http://slidetodoc.com/presentation_image/1b54ce05c6e4673ee45f249c413561ab/image-31.jpg)
Calculate the silver ion concentration, [Ag+], of a solution prepared by dissolving 1. 00 g of silver nitrate and 10. 0 g of potassium cyanide in 1. 00 of solution.
![Calculate the [Hg. Cl 42 -] in a solution prepared by adding 0. 0200 Calculate the [Hg. Cl 42 -] in a solution prepared by adding 0. 0200](http://slidetodoc.com/presentation_image/1b54ce05c6e4673ee45f249c413561ab/image-32.jpg)
Calculate the [Hg. Cl 42 -] in a solution prepared by adding 0. 0200 M of Na. Cl to 0. 250 L of a 0. 100 M Hg. Cl 2 solution.

What are your questions?
- Slides: 33