Solubility Curves Solubility curves Solubility curves can be

Solubility Curves

Solubility curves • Solubility curves can be used to show the how much of a solid will dissolve at a given temperature • By convention solubility curves are graphed as solubility (g/100 g of H 2 O) • So how many grams of a solid can be dissolved in 100 g of water • 100 g of water =100 m. L of water
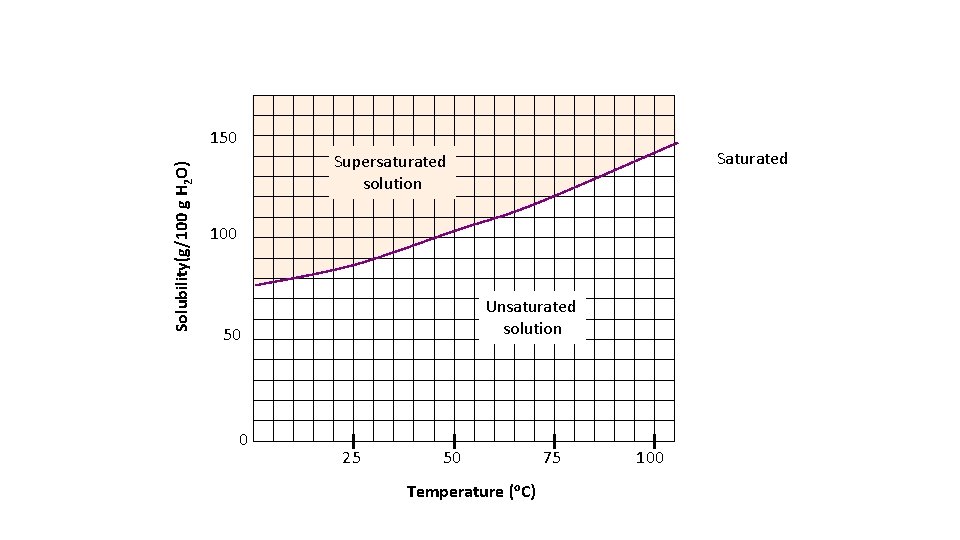
Solubility(g/100 g H 2 O) 150 Saturated Supersaturated solution 100 Unsaturated solution 50 0 25 50 Temperature (o. C) 75 100
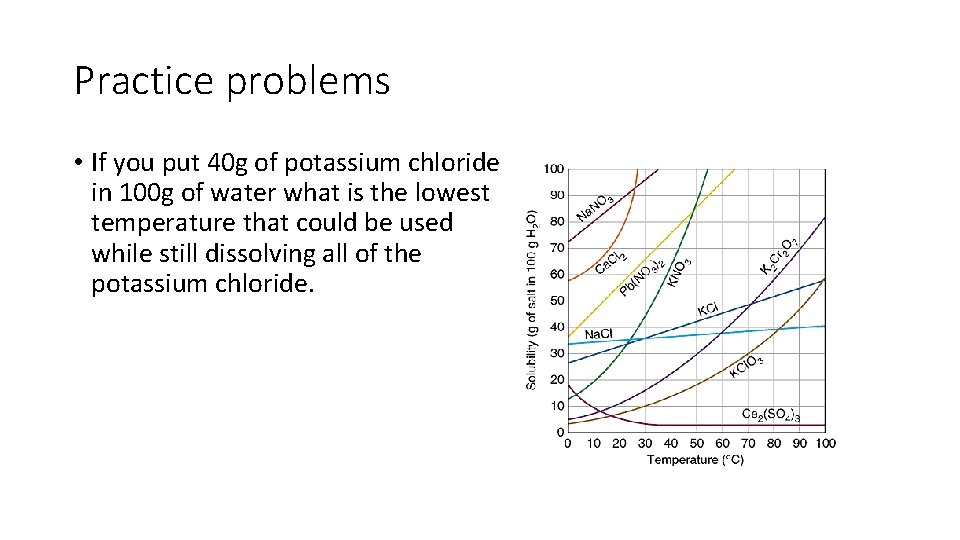
Practice problems • If you put 40 g of potassium chloride in 100 g of water what is the lowest temperature that could be used while still dissolving all of the potassium chloride.
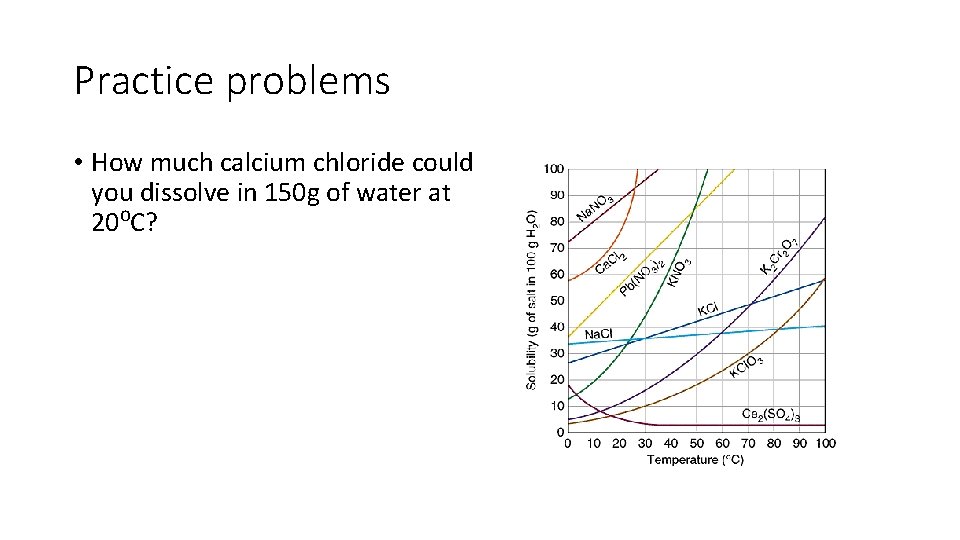
Practice problems • How much calcium chloride could you dissolve in 150 g of water at 20⁰C?
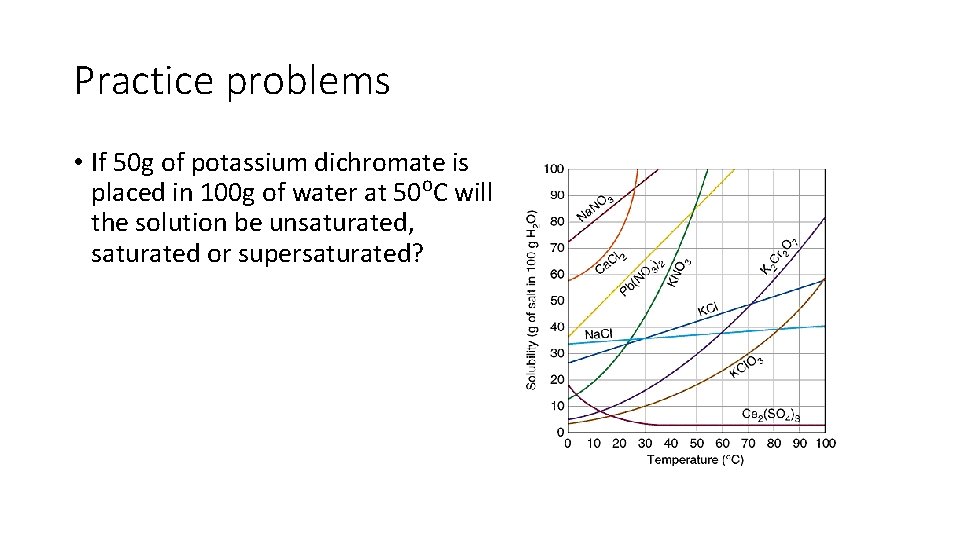
Practice problems • If 50 g of potassium dichromate is placed in 100 g of water at 50⁰C will the solution be unsaturated, saturated or supersaturated?
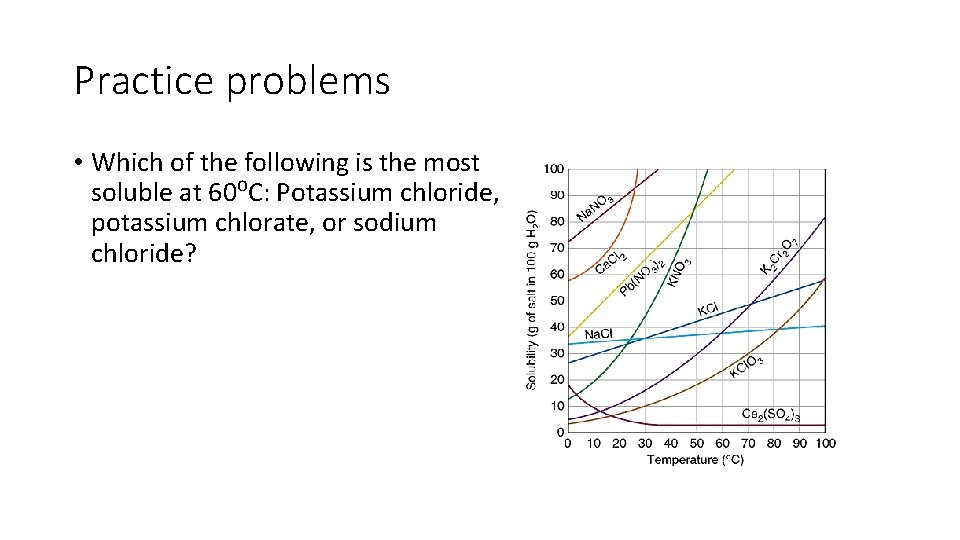
Practice problems • Which of the following is the most soluble at 60⁰C: Potassium chloride, potassium chlorate, or sodium chloride?
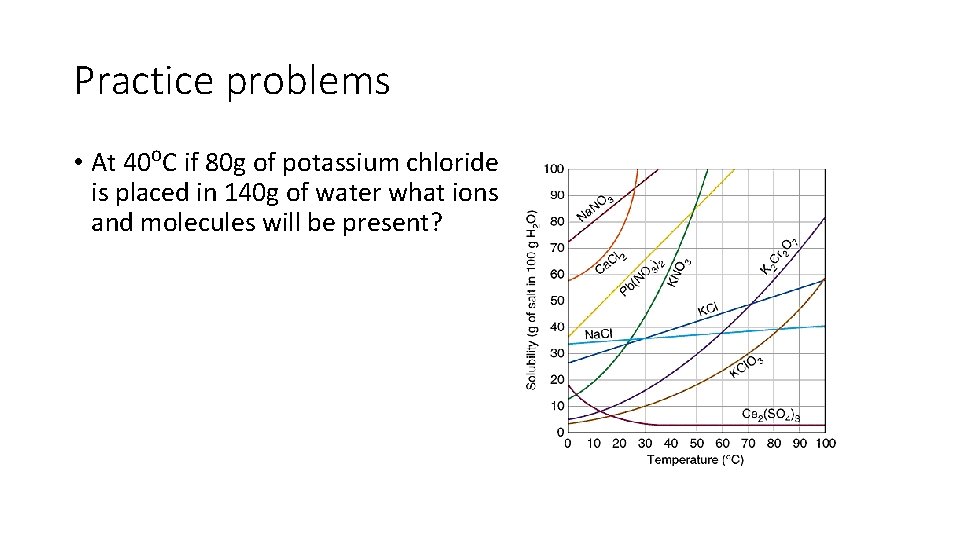
Practice problems • At 40⁰C if 80 g of potassium chloride is placed in 140 g of water what ions and molecules will be present?

Common ion effect • What happens if we add a solid to a solution which contains a common ion or molecule? • Ex. A solution containing 0. 100 M F 2 has solid calcium fluoride added. What is the molar solubility of calcium fluoride when placed into this solution? Ca 2+ I C E F 2

Ex. 2 • A solution of 2. 0 M beryllium hydroxide is prepared and completely dissociates into its ions. Solid silver hydroxide is added to the solution, what is the molar solubility of the silver hydroxide in this solution? Ag+ I C E OH-

Common Ion Effect • When a solution is made with two compounds that share a common ion the molar solubility (s) is lowered. • For example if sodium hydroxide (which completely dissociates) and silver hydroxide are placed in solution they will dissociate into their respective ions Na+(aq)+ OH-(aq) Common Ion Na. OH(s) Ag+(aq) + OH-(aq) • Because Ag. OH is not very soluble there may be some solid Ag. OH dependant upon how much is added.

Example problem 3: • Silver hydroxide is added to a solution of 0. 200 mol/L sodium hydroxide. Find the molar solubility of silver hydroxide in this solution. • Step 1: State the separate chemical equations for the dissociation of sodium hydroxide and silver hydroxide Na. OH(s) Na+(aq)+ OH-(aq) Ag. OH(s) Ag+(aq) + OH-(aq)

Example problem: • Step 2: Set up an Ice table for the compound you are finding molar solubility for Ag+ OH- I C E • Step 3: Determine the concentration of the common ion. Remember to look at the coefficients of the original solution. This is your Initial concentration on the ice table.
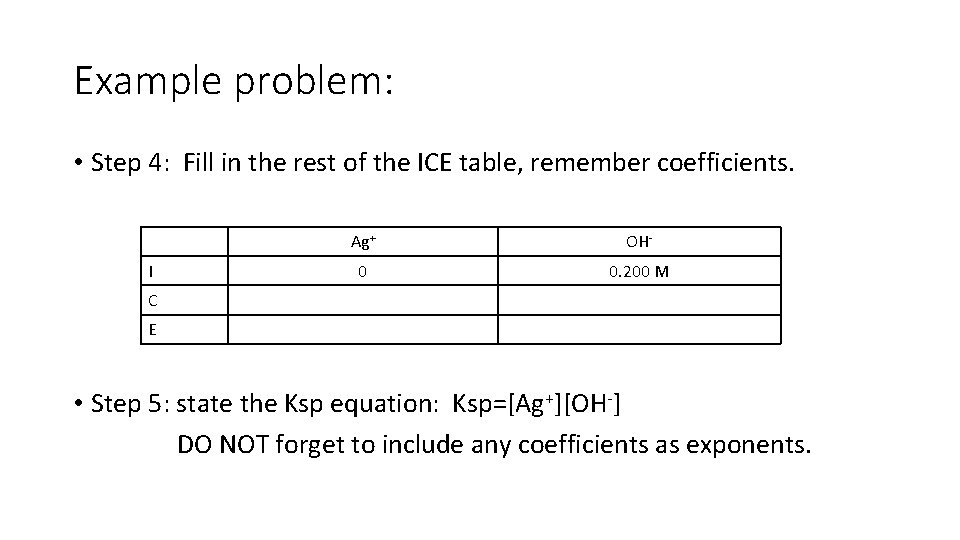
Example problem: • Step 4: Fill in the rest of the ICE table, remember coefficients. I Ag+ OH- 0 0. 200 M C E • Step 5: state the Ksp equation: Ksp=[Ag+][OH-] DO NOT forget to include any coefficients as exponents.

Example problem: • Step 6: substitute concentrations from equilibrium (E) into the Ksp equation, state what Ksp for this compound is. Ag+ OH- I 0 0. 200 M C +s +s E S 0. 200+s • Ksp = (s) (0. 200+s) Ksp= • Step 7: Assume “s” is small because Ksp is small, 0. 200 + s still equals 0. 200, just like the “x” is small approximation.
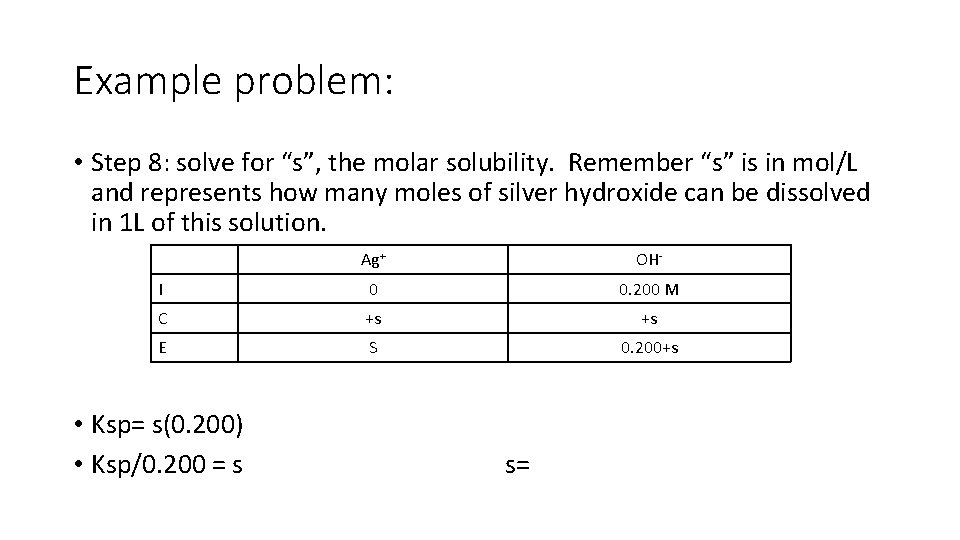
Example problem: • Step 8: solve for “s”, the molar solubility. Remember “s” is in mol/L and represents how many moles of silver hydroxide can be dissolved in 1 L of this solution. Ag+ OH- I 0 0. 200 M C +s +s E S 0. 200+s • Ksp= s(0. 200) • Ksp/0. 200 = s s=
- Slides: 16