Solubility Curves How much solute can a solvent

Solubility Curves

How much solute can a solvent (H 2 O) hold? • Depends on amount of solvent & temperature (Pressure for gases) – Increasing the temperature increases the solubility of a solid solute – Increasing the temperature decreases the solubility of a gaseous solute – Increasing the pressure increases the solubility of a gaseous solute
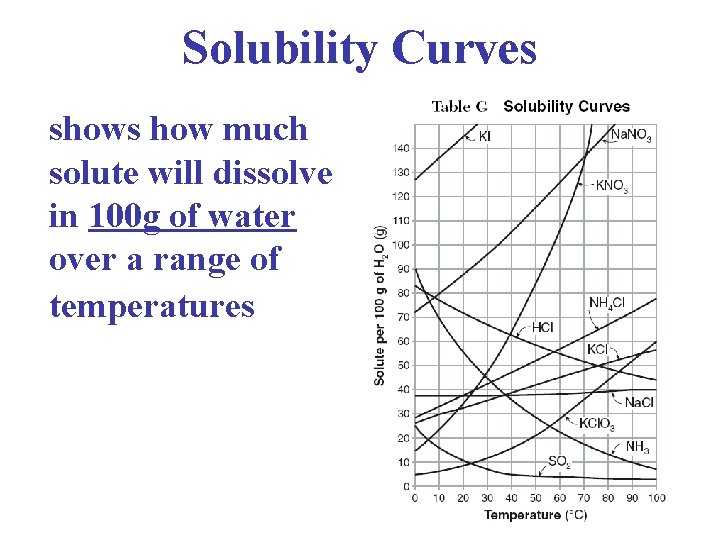
Solubility Curves shows how much solute will dissolve in 100 g of water over a range of temperatures
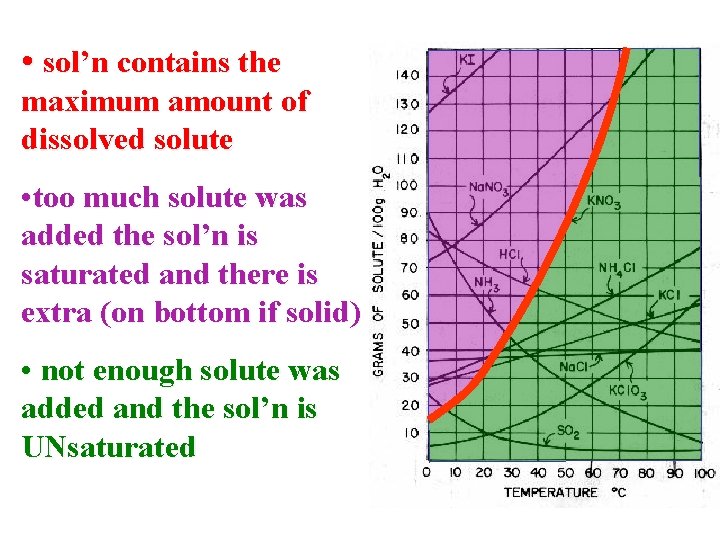
• sol’n contains the maximum amount of dissolved solute • too much solute was added the sol’n is saturated and there is extra (on bottom if solid) • not enough solute was added and the sol’n is UNsaturated
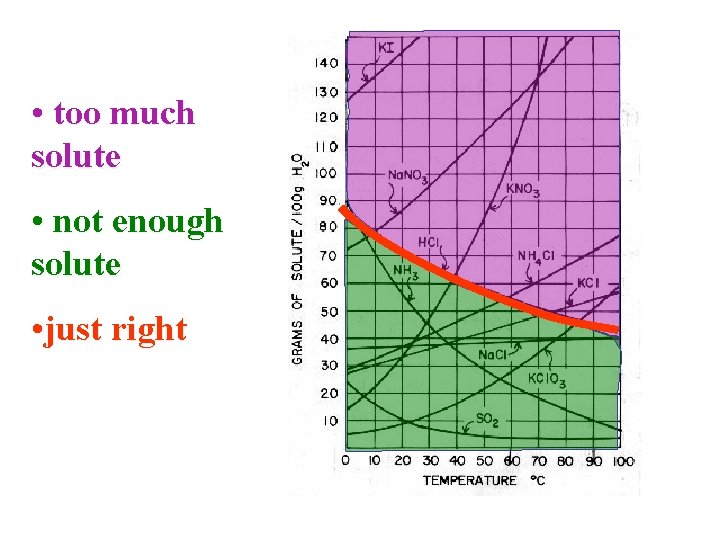
• too much solute • not enough solute • just right
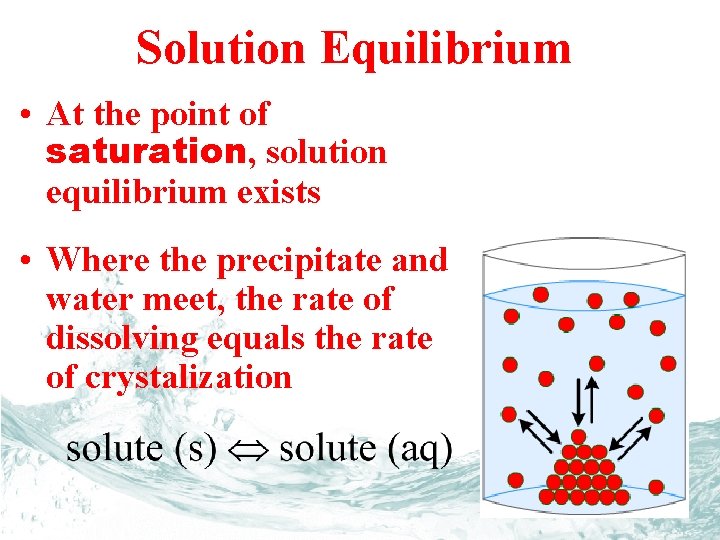
Solution Equilibrium • At the point of saturation, solution equilibrium exists • Where the precipitate and water meet, the rate of dissolving equals the rate of crystalization
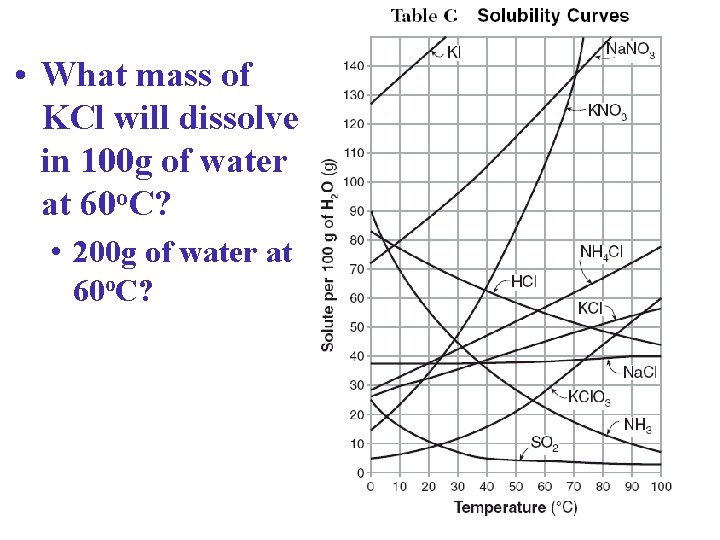
• What mass of KCl will dissolve in 100 g of water at 60 o. C? • 200 g of water at 60 o. C? KCl
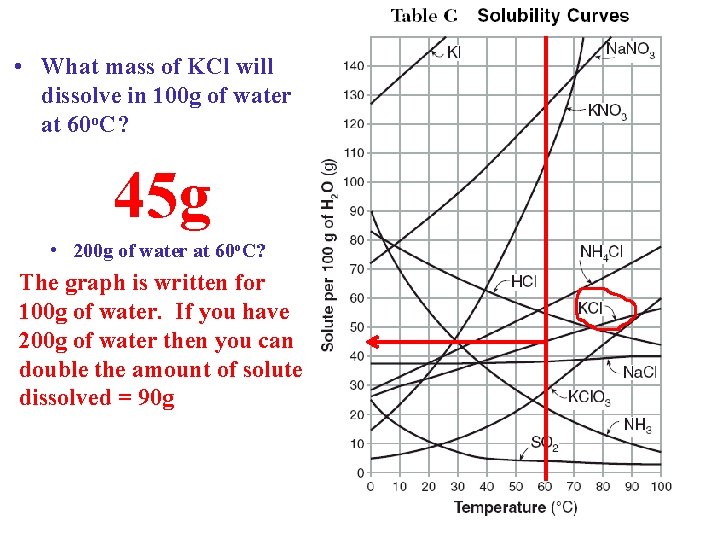
• What mass of KCl will dissolve in 100 g of water at 60 o. C? 45 g • 200 g of water at 60 o. C? The graph is written for 100 g of water. If you have 200 g of water then you can double the amount of solute dissolved = 90 g KCl
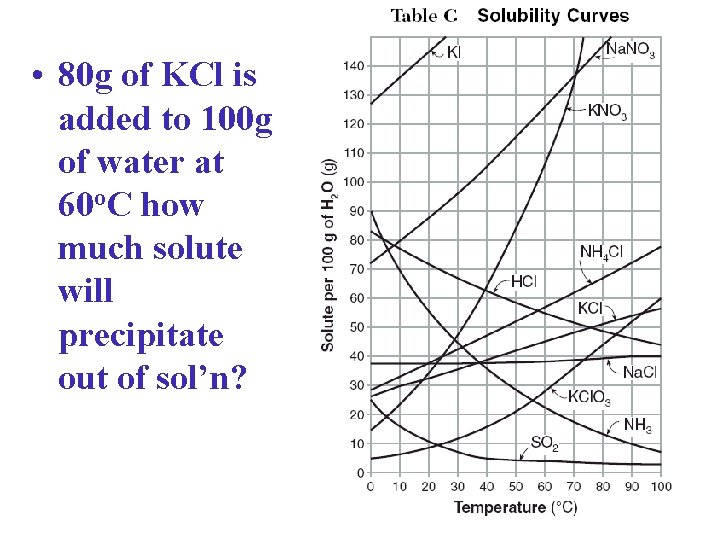
• 80 g of KCl is added to 100 g of water at 60 o. C how much solute will precipitate out of sol’n?
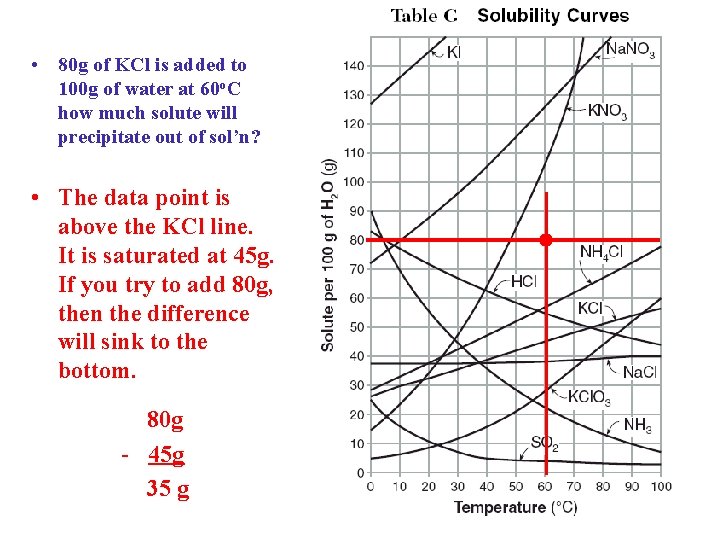
• 80 g of KCl is added to 100 g of water at 60 o. C how much solute will precipitate out of sol’n? • The data point is above the KCl line. It is saturated at 45 g. If you try to add 80 g, then the difference will sink to the bottom. 80 g - 45 g 35 g
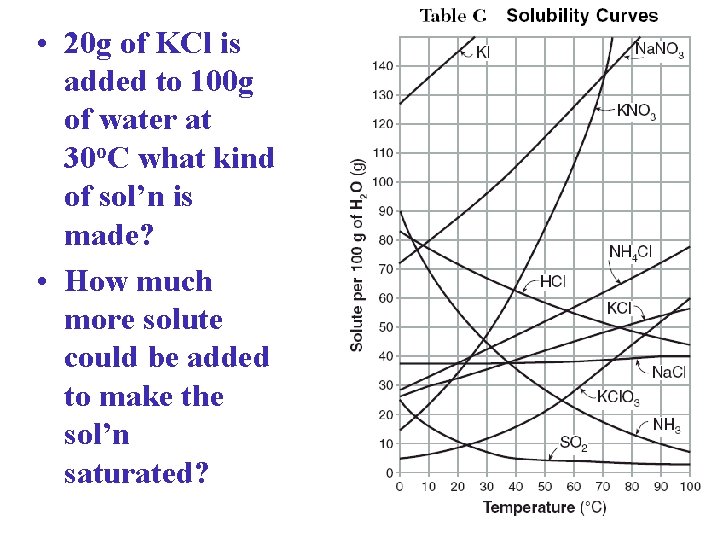
• 20 g of KCl is added to 100 g of water at 30 o. C what kind of sol’n is made? • How much more solute could be added to make the sol’n saturated?
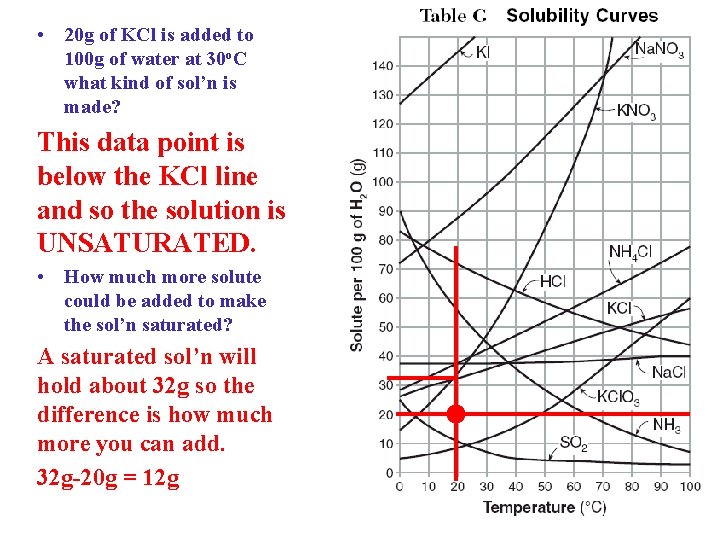
• 20 g of KCl is added to 100 g of water at 30 o. C what kind of sol’n is made? This data point is below the KCl line and so the solution is UNSATURATED. • How much more solute could be added to make the sol’n saturated? A saturated sol’n will hold about 32 g so the difference is how much more you can add. 32 g-20 g = 12 g

The End
- Slides: 13