Solubility Concentration CREDIT D Scott CHS Concentration of

Solubility & Concentration CREDIT: D. Scott, CHS
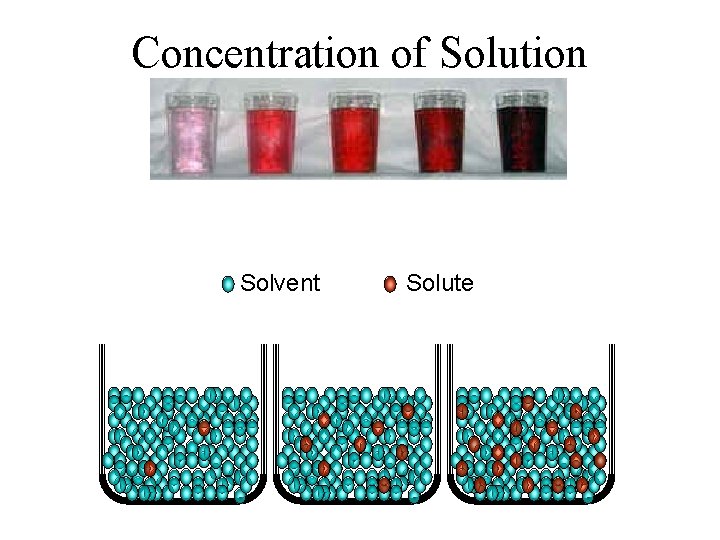
Concentration of Solution Solvent Solute

Concentration of Solution Moles of solute Mol • Molarity (M) = Liter of solution = L • Parts ratio amount of solute (g or ml) 2) or (106) or (109) (10 = amount of solution (g or ml) • Mole Fraction (c)= Total moles of solution Moles of solute
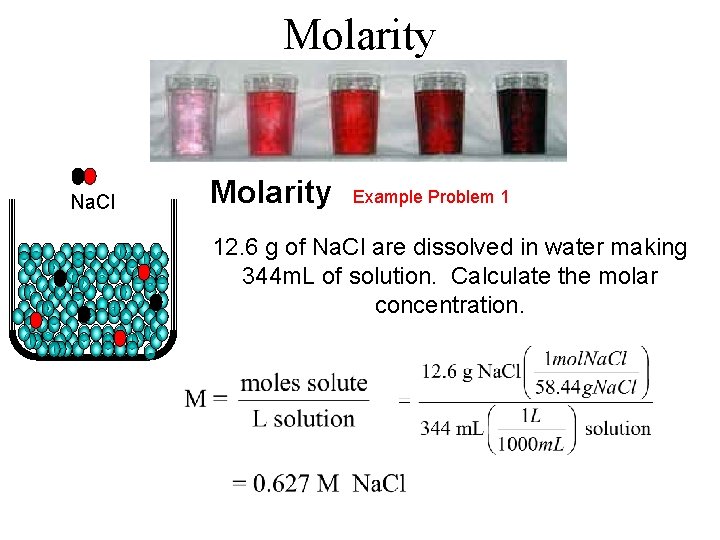
Molarity Na. Cl Molarity Example Problem 1 12. 6 g of Na. Cl are dissolved in water making 344 m. L of solution. Calculate the molar concentration.
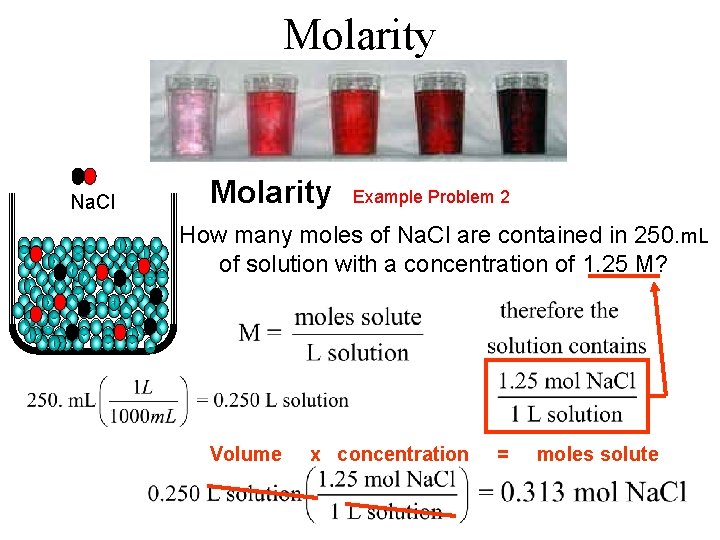
Molarity Na. Cl Molarity Example Problem 2 How many moles of Na. Cl are contained in 250. m. L of solution with a concentration of 1. 25 M? Volume x concentration = moles solute
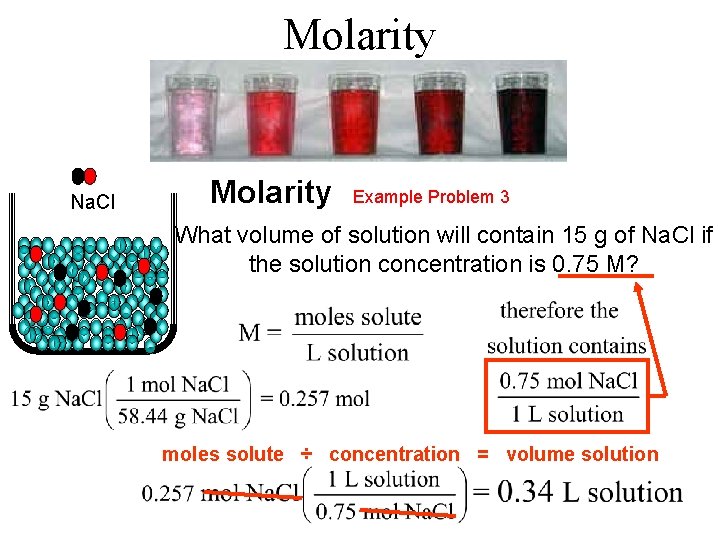
Molarity Na. Cl Molarity Example Problem 3 What volume of solution will contain 15 g of Na. Cl if the solution concentration is 0. 75 M? moles solute ÷ concentration = volume solution

% Concentration • % (w/w) = • % (w/v) = • % (v/v) = Mass and volume units must match. (g & m. L) or (Kg & L)

% Concentration Example Problem 1 (Solid in a Liquid) What is the concentration in %w/v of a solution containing 39. 2 g of potassium nitrate in 177 m. L of solution? % (w/v) = Example Problem 2 (Liquid in a Liquid) What is the concentration in %v/v of a solution containing 3. 2 L of ethanol in 6. 5 L of solution? % (v/v) =

% Concentration Example Problem 3 What volume of 1. 85 %w/v solution is needed to provide 5. 7 g of solute? % (w/v) = We know: g solute ÷ concentration We want to get: = volume solution

Parts per million/billion (ppm & ppb) • ppm = or = ppm • ppb = or = ppb Mass and volume units must match. (g & m. L) or (Kg & L) parts per trillion AND For very low concentrations: = ppt

ppm & ppb Example Problem 1 An Olympic sized swimming pool contains 2, 500, 000 L of water. If 1 tsp of salt (Na. Cl) is dissolved in the pool, what is the concentration in ppm? 1 teaspoon = 6. 75 g Na. Cl or

ppm & ppb Example Problem 2 An Olympic sized swimming pool contains 2, 500, 000 L of water. If 1 tsp of salt (Na. Cl) is dissolved in the pool, what is the concentration in ppb? 1 teaspoon = 6. 75 g Na. Cl or
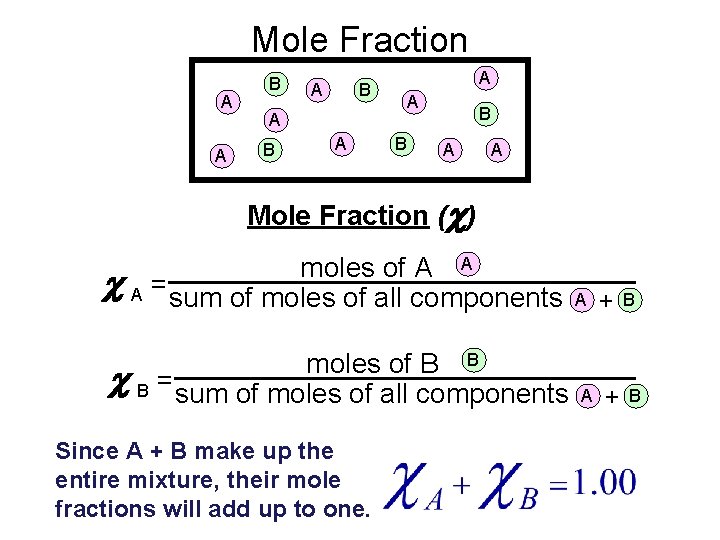
Mole Fraction A A B B A A Mole Fraction (c) moles of A A c A = sum of moles of all components moles of B B c B = sum of moles of all components Since A + B make up the entire mixture, their mole fractions will add up to one. A A + + B B

Mole Fraction Example Problem 1 In our glass of iced tea, we have added 3 tbsp of sugar (C 12 H 22 O 11). The volume of the tea (water) is 325 m. L. What is the mole fraction of the sugar in the tea solution? (1 tbsp sugar ≈ 25 g) First, we find the moles of both the solute and the solvent. Next, we substitute the moles of both into the mole fraction equation.

Mole Fraction Example Problem 2 Air is about 78% N 2, 21% O 2, and 0. 90% Ar. What is the mole fraction of each gas? First, we find the moles of each gas. We assume 100. grams total and change each % into grams. Next, we substitute the moles of each into the mole fraction equation.
- Slides: 15