Solubility and Solubility Curves What are Solutions Made
Solubility and Solubility Curves

What are Solutions Made Of? • A solution is a homogeneous mixture of 2 or more substances in a single phase. • The substance being dissolved (or is less concentrated) is the SOLUTE. • The substance doing the dissolving (or is more concentrated) is the SOLVENT.

“like dissolves like” Two substances with similar intermolecular forces are likely to be soluble in each other. • non-polar molecules are soluble in non-polar solvents CCl 4 in C 6 H 6 • polar molecules are soluble in polar solvents C 2 H 5 OH in H 2 O • ionic compounds are more soluble in polar solvents Na. Cl in H 2 O or NH 3 (l) 12. 2
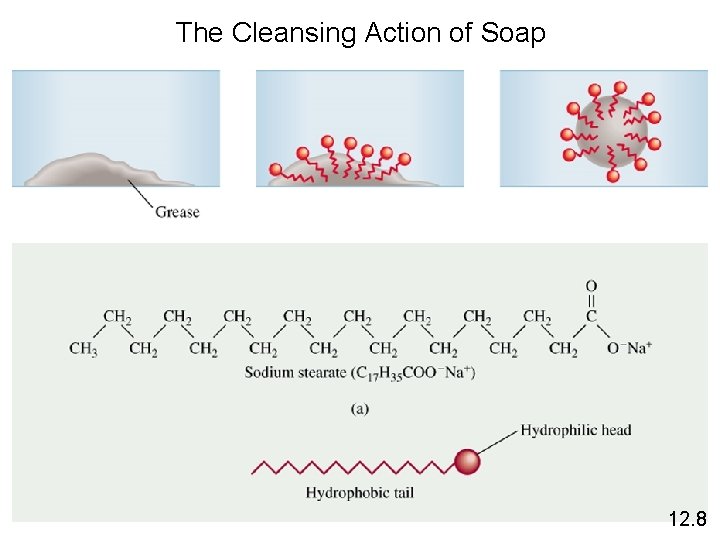
The Cleansing Action of Soap 12. 8

Not All Things Dissolve the Same… • When a solid dissolves into a liquid, the particles of solid break down into their individual parts and spread out throughout the liquid • BUT… we’ve got some different vocab words for the type of solid doing this – Ionic compounds will dissociate into their separate ions – Ex: Ca(OH)2 (s) Ca 2+(aq) + 2 OH-(aq)

Electrolytes in the Body Electrolytes are ionic solutions that conduct electricity. Ionic compounds that dissociate easily are called Strong Electrolytes Ionic compounds that don’t dissociate at all are called Non -electrolytes Carry messages to and from the brain as electrical signals. Important to maintain cellular function.

Covalent Compounds • Covalent compounds will still dissolve, but each molecule stays completely together, just spread further apart • This is called dispersion • Sugar is an excellent example of this • C 6 H 12 O 6 (s) C 6 H 12 O 6 (aq)

Acids – The in-between ones • Acids are a bit special because they are covalent compounds, but they break into ions when mixed with water. • So they get a special term… Ionization! • Ex: HCl (g) H+ (aq) + Cl- (aq)

Solutions are not just solids dissolved in liquids! Solute Solvent Example solid liquid gas gas Metal alloys 14 -K Gold Kool-Aid (Sugar in H 2 O) Wine or Vinegar Alcohol or Acid in H 2 O Carbonate Beverages (CO 2 in H 2 O) Air (O 2 and other gases in N 2)

Solubility is • The amount of solute that dissolves in a specific amount of solvent. • Expressed as grams of solute in 100 grams ( or ml) of solvent water. Grams of solute 100 g/ml water

Types of Solutions • Unsaturated • Contain less than the maximum amount of solute. • Can dissolve more solute. Dissolved solute
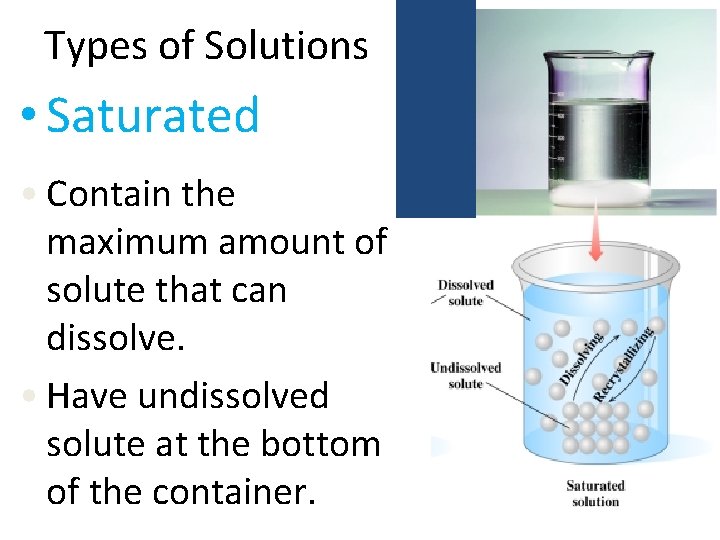
Types of Solutions • Saturated • Contain the maximum amount of solute that can dissolve. • Have undissolved solute at the bottom of the container.

Types of Solutions • Supersaturated • An unstable solution that contains an amount of solute greater than normal. • Will start out with no extra solute on bottom, but will eventually appear

• Simply adding in more solute DOES NOT create a supersaturated solution! • First, raise the temperature up to cause more solute to dissolve. • Next, lower the temperature back to where it was. NOW it’s a supersaturated solution!

Learning Check At 40 C, the solubility of KBr is 80 g/100 g H 2 O. Identify the following solutions as either 1) Saturated, (2) Unsaturated, or (3) Supersaturated A. 60 g KBr added to 100 g of water at 40 C. B. 200 g KBr added to 200 g of water at 40 C. C. 25 g KBr added to 50 g of water at 40 C. 15

A. 2 Amount of 60 g KBr/100 g water is less than the solubility of 80 g KBr/100 g water. B. 1 In 100 g of water, 100 g KBr exceeds the solubility of 80 g KBr water at 40 C. 2 This is the same as 50 g KBr in 100 g of water, which is less than the solubility of 80 g KBr/100 g water at 40 C. 16
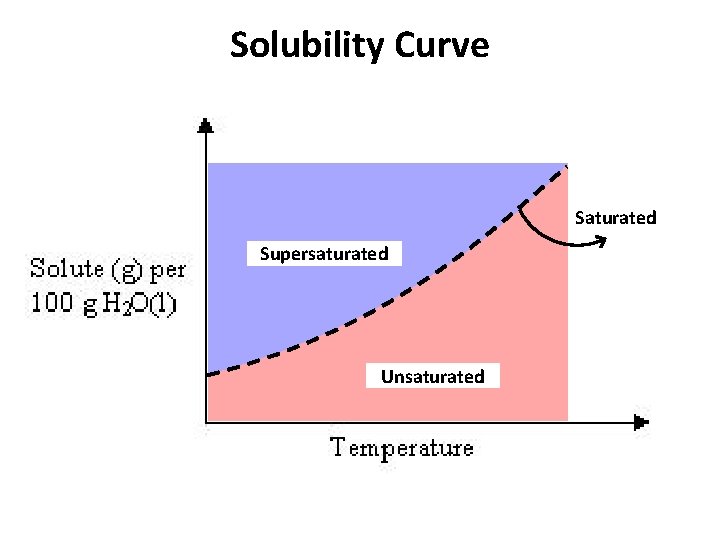
Solubility Curve Saturated Supersaturated Unsaturated
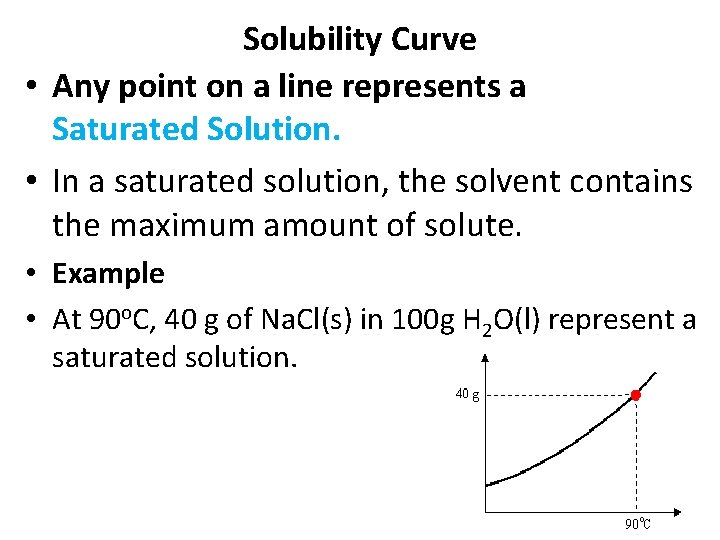
Solubility Curve • Any point on a line represents a Saturated Solution. • In a saturated solution, the solvent contains the maximum amount of solute. • Example • At 90 o. C, 40 g of Na. Cl(s) in 100 g H 2 O(l) represent a saturated solution.
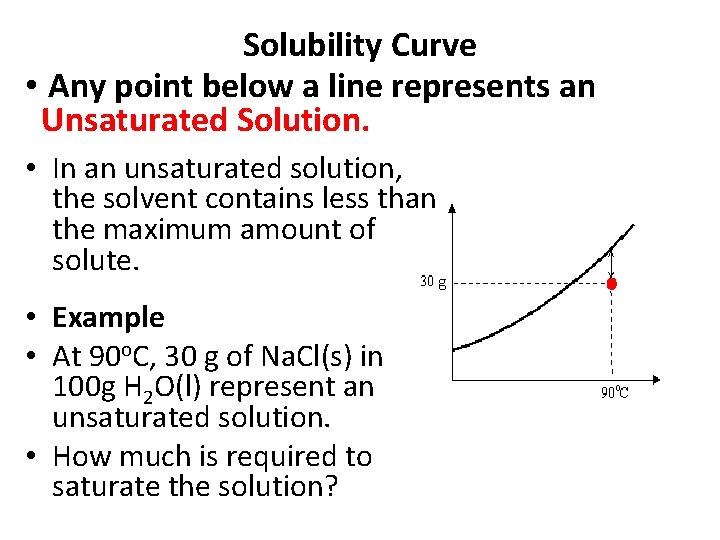
Solubility Curve • Any point below a line represents an Unsaturated Solution. • In an unsaturated solution, the solvent contains less than the maximum amount of solute. • Example • At 90 o. C, 30 g of Na. Cl(s) in 100 g H 2 O(l) represent an unsaturated solution. • How much is required to saturate the solution?
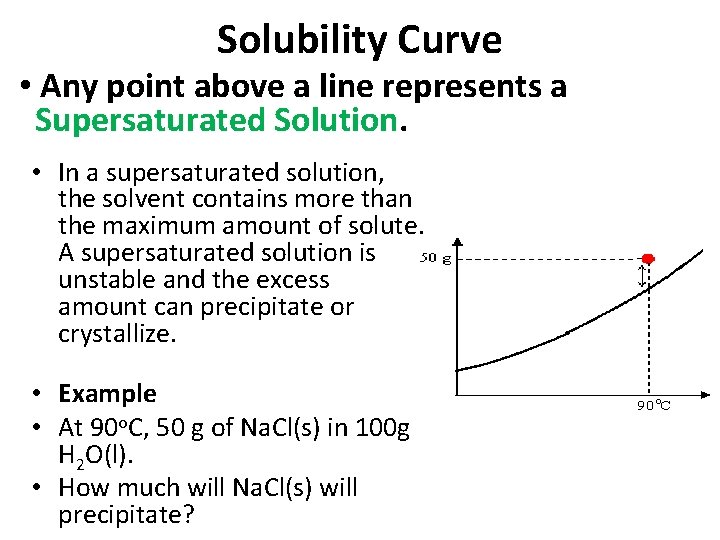
Solubility Curve • Any point above a line represents a Supersaturated Solution. • In a supersaturated solution, the solvent contains more than the maximum amount of solute. A supersaturated solution is unstable and the excess amount can precipitate or crystallize. • Example • At 90 o. C, 50 g of Na. Cl(s) in 100 g H 2 O(l). • How much will Na. Cl(s) will precipitate?
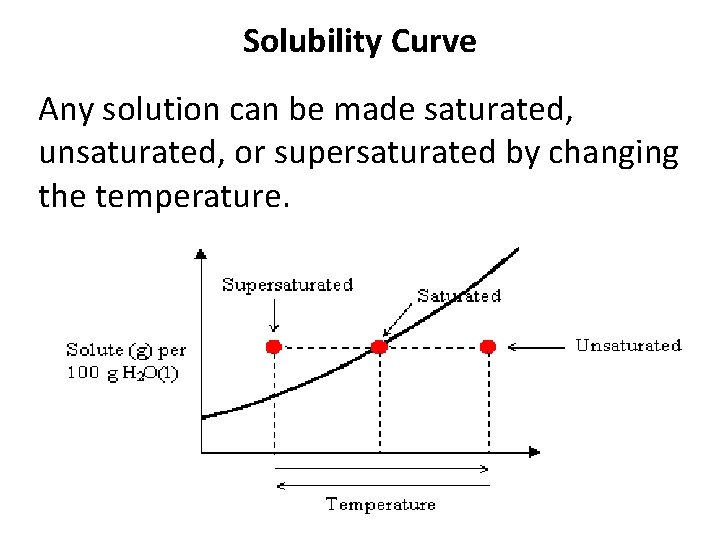
Solubility Curve Any solution can be made saturated, unsaturated, or supersaturated by changing the temperature.
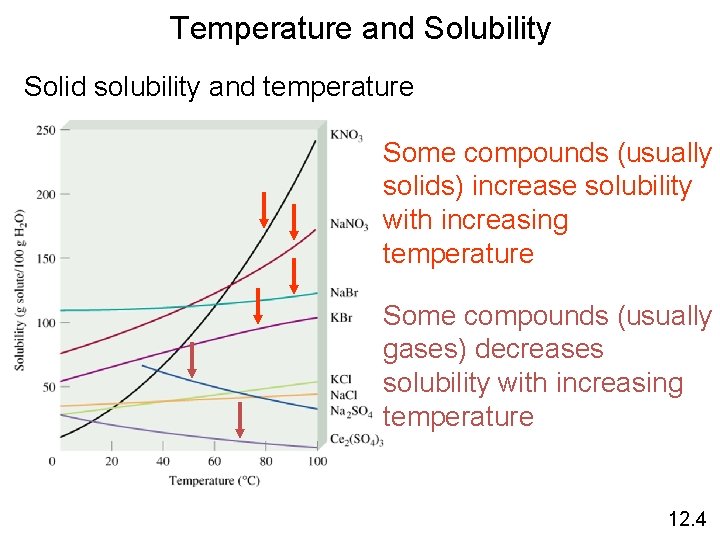
Temperature and Solubility Solid solubility and temperature Some compounds (usually solids) increase solubility with increasing temperature Some compounds (usually gases) decreases solubility with increasing temperature 12. 4

Chemistry In Action: The Killer Lake Nyos – formed from a former volcano that deposited large quantities of CO 2 at the bottom of the lake. It is not sure of the exact cause, but at some instant, 1. 6 million tons of CO 2 was released from the bottom of the lake. As the gas erupted and covered the land, all oxygen was displaced and over 1, 700 individuals and over 3, 500 livestock were killed due to asphyxiation. Lake Nyos, West Africa
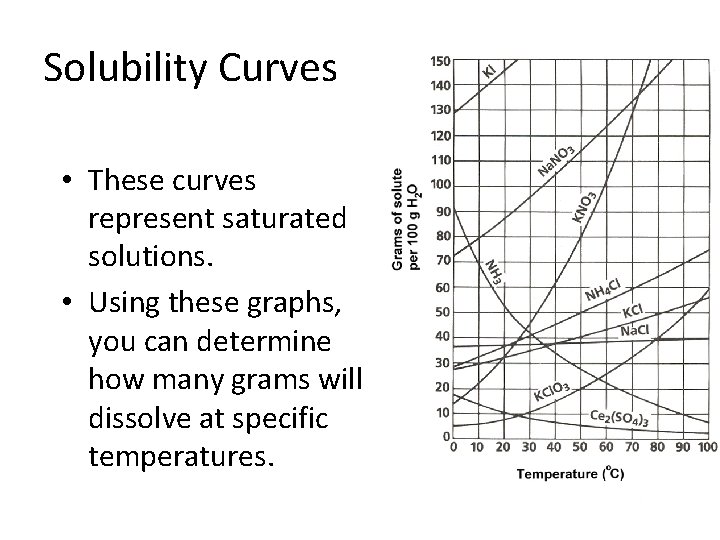
Solubility Curves • These curves represent saturated solutions. • Using these graphs, you can determine how many grams will dissolve at specific temperatures.
- Slides: 24