Solids Valentim M B Nunes ESTT IPT May

Solids Valentim M. B. Nunes ESTT - IPT May 2019

Unlike gases, solids have an order of short and long range (localized particles). In a monoatomic crystal every atom vibrates around the average equilibrium positions, according to each of the three axes.

Einstein model "The simple idea about thermal motion in solid bodies is to consider that atoms perform harmonic oscillations around their equilibrium positions" Crystal 3 N harmonic oscillators E

If g( ) is a frequency distribution function, such that g( )d is the number of vibration frequencies in the range and + d , then: In the Einstein’s solid: g( ) E is the characteristic frequency of Einstein. E

is the characteristic temperature of Einstein.

Considering the partition function for each of the oscillators, zvib: Expression deduced by Einstein: crystals with the same value of θE/T have the same heat capacity (corresponding states).

For high temperatures, T , At high temperatures the limit for heat capacity is:
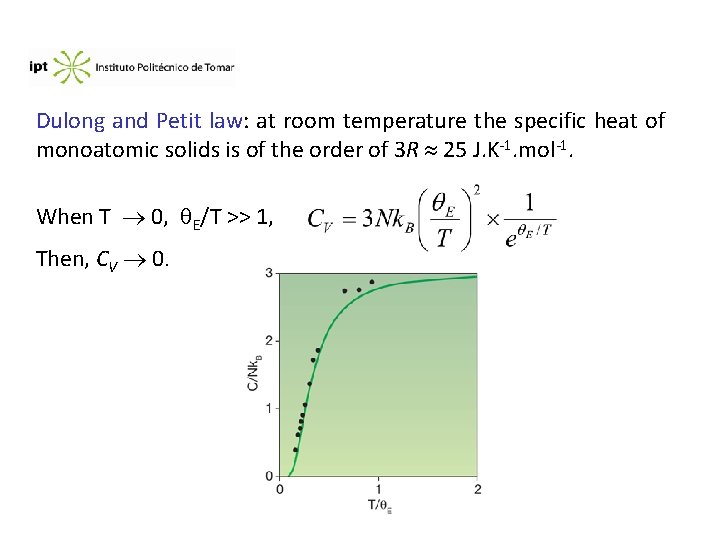
Dulong and Petit law: at room temperature the specific heat of monoatomic solids is of the order of 3 R 25 J. K-1. mol-1. When T 0, E/T >> 1, Then, CV 0.

Nernst – Lindeman model g( ) E/2 E Model predicts better CV values at low temperatures.

Debye model: Debye considers that the atoms of a crystal vibrate not all with the same frequency, but that g( ) = A 2, with a maximum frequency, D. g( ) 3 N D

where: D is the Debye characteristic temperature.

When T , then According to the Einstein model and the law of Dulong and Petit.

When T 0, Debye cubic law: at low temperatures the heat capacity varies proportionally to the cube of the temperature, i. e. CV T 3.
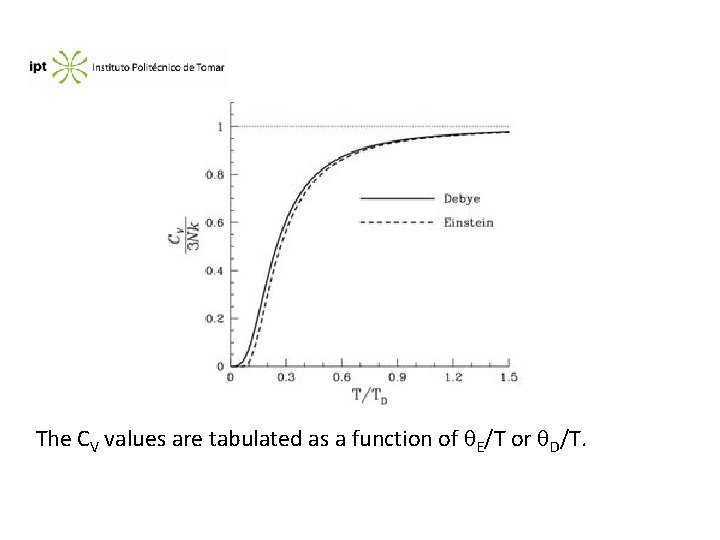
The CV values are tabulated as a function of E/T or D/T.

For diamond the experimental value for the heat capacity is CV = 2. 68 J. K-1. mol-1 at 207 K. Compare this value with the predicted by the models of a)Einstein e b) Debye. a) E/T = 1364/207 = 6. 59. By the tables, CV/3 R = 0. 05984, CV = 1. 4925 J. K-1. mol-1, much lower than the experimental value b) D/T = 1860/207 = 8. 9855. By the tables, CV/3 R = 0. 10208, CV = 2. 54 J. K-1. mol-1 , quite close to the experimental value.
- Slides: 15