Solids Liquids and Intermolecular forces Objective 1 Learn

Solids, Liquids, and Intermolecular forces

Objective • 1. Learn the concepts of intermolecular forces. • 2. Use the intermolecular forces to explain and predict the physical properties of substances.

Solids and Liquids • A substance’s state of matter depends on two factors: the average kinetic energy of the particles, and the intermolecular forces between the particles.
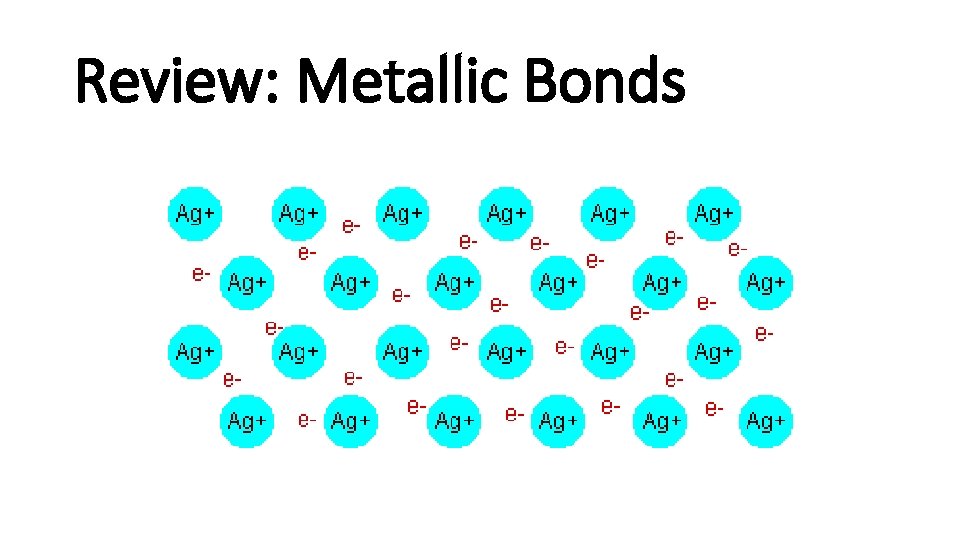
Review: Metallic Bonds
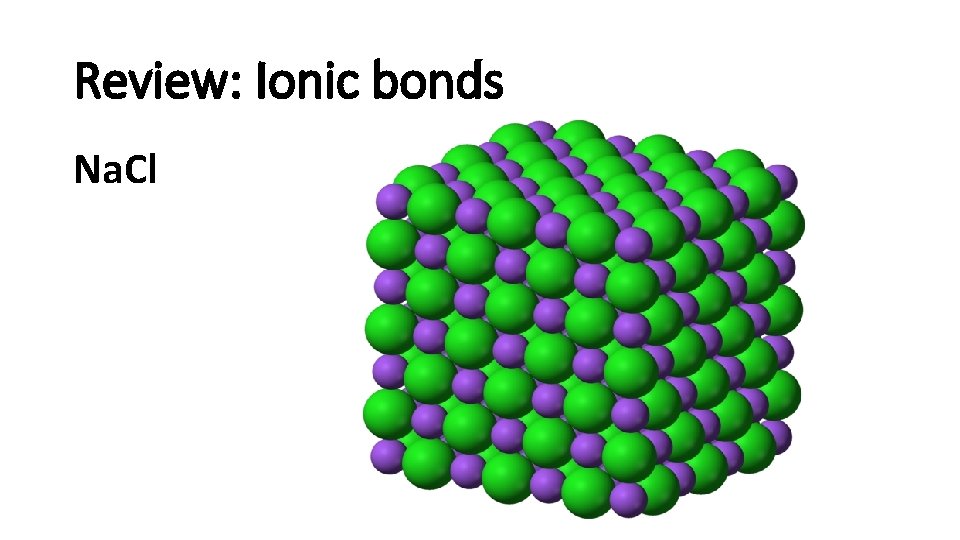
Review: Ionic bonds Na. Cl
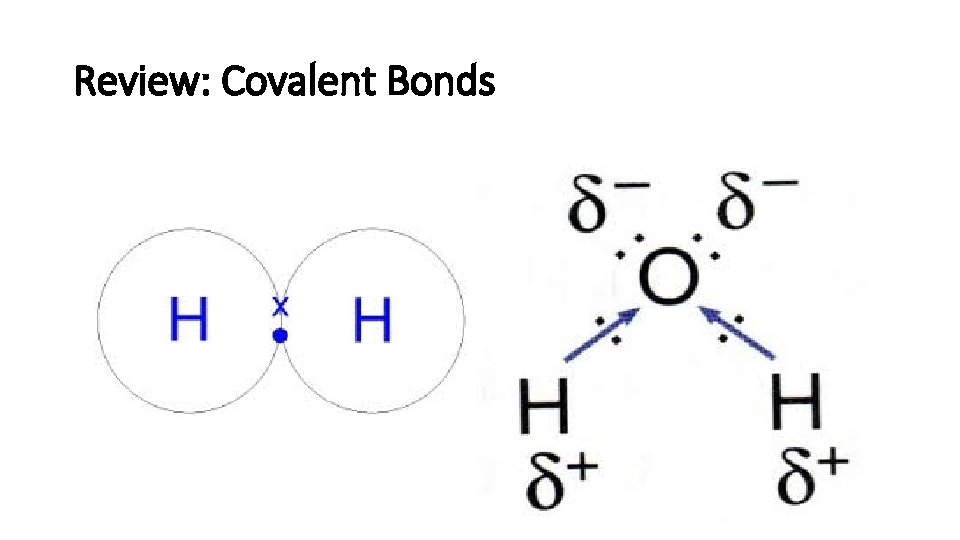
Review: Covalent Bonds

Review: Covalent bonds: • Polar molecule: H 2 O, NH 3 • Nonpolar molecule: O 2 , CO 2 , H 2 • Polarity: the dipole of a molecule. • Dipole moment: the measure of polarity.

Intermolecular force: London Dispersion Forces • It is the random polarity in a fleeting instant in nonpolar molecules. • It is very weak. • Example: gasoline, N 2, CO 2

Intermolecular Force: Dipole-Dipole • The intermolecular force between polar molecules: the positive end of one polar molecule is attracted to the negative end of another polar molecule. • Example: CH 3 CH 2 OH
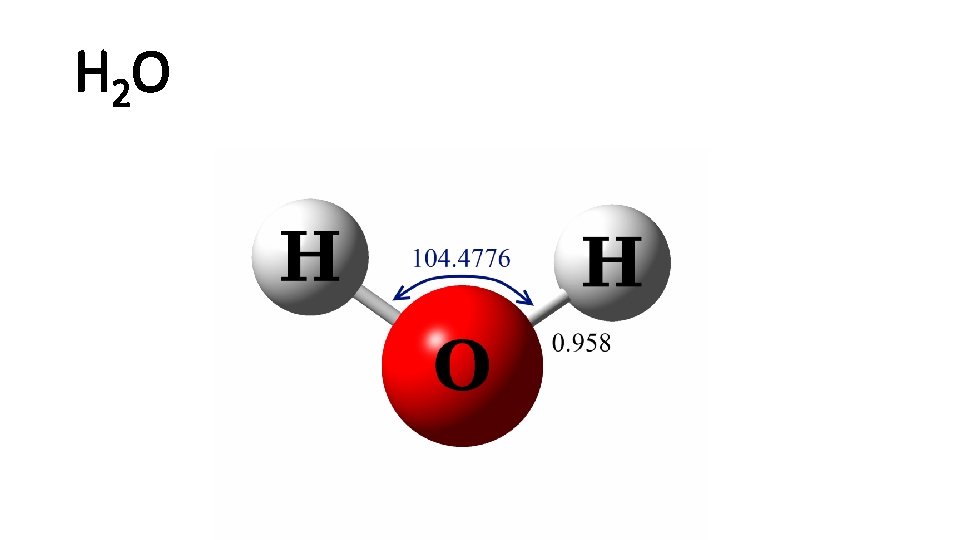
H 2 O
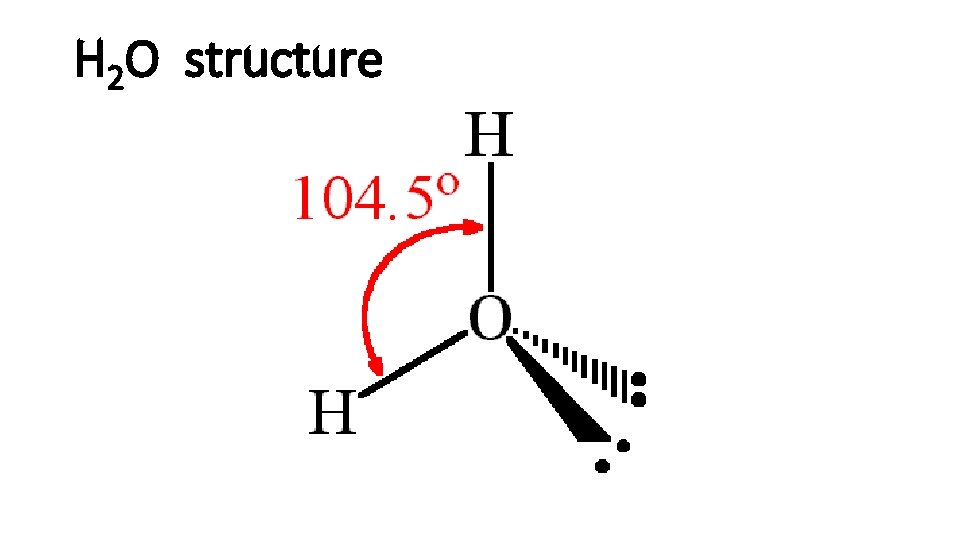
H 2 O structure
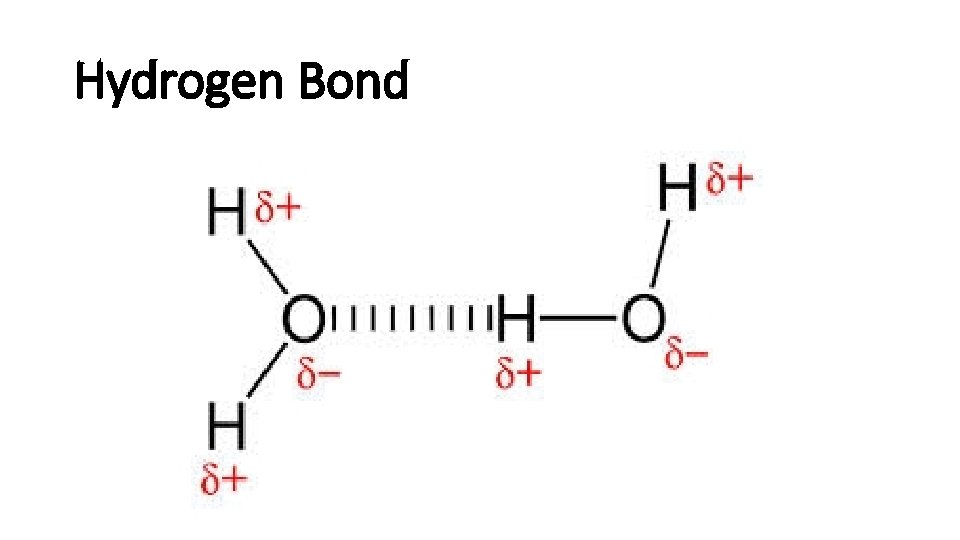
Hydrogen Bond
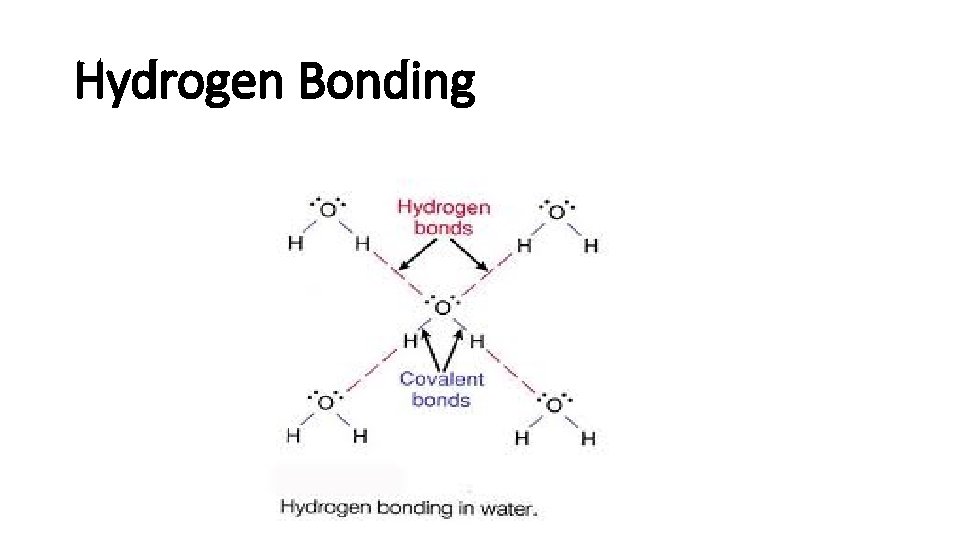
Hydrogen Bonding
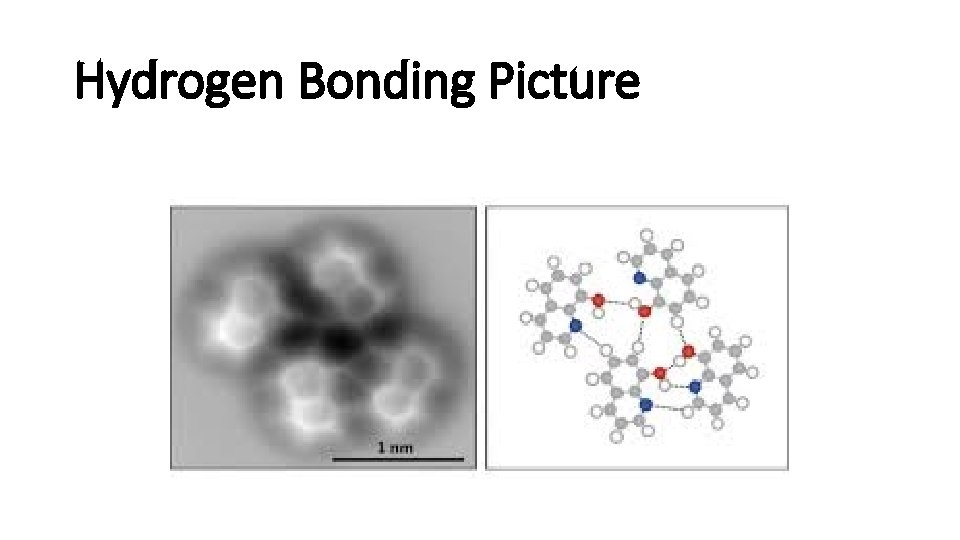
Hydrogen Bonding Picture

Hydrogen bonds • H is a nucleus, positive charged. • The other extremely electronegative elements like O, N, and F has lone pair electrons. • H and a electronegative element form hydrogen bond. • Hydrogen bond is strong.
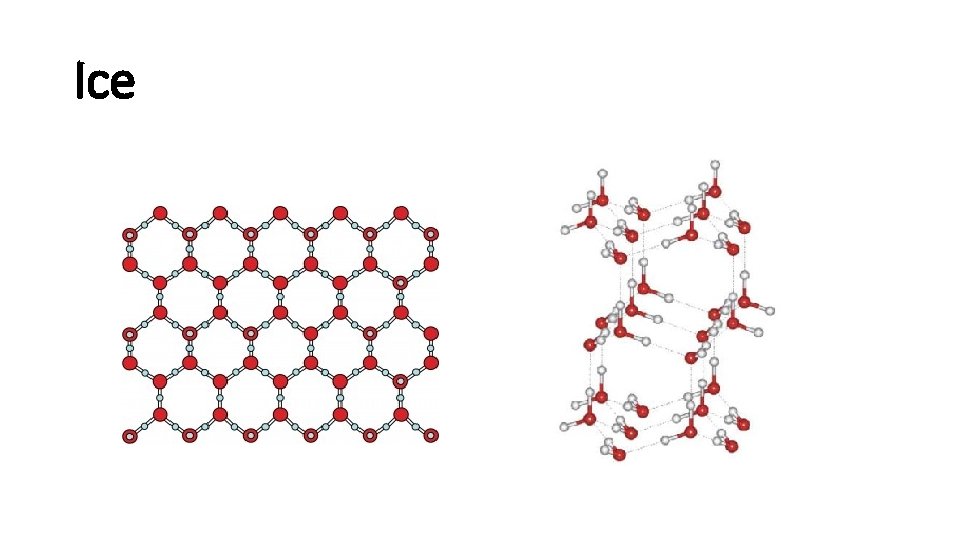
Ice
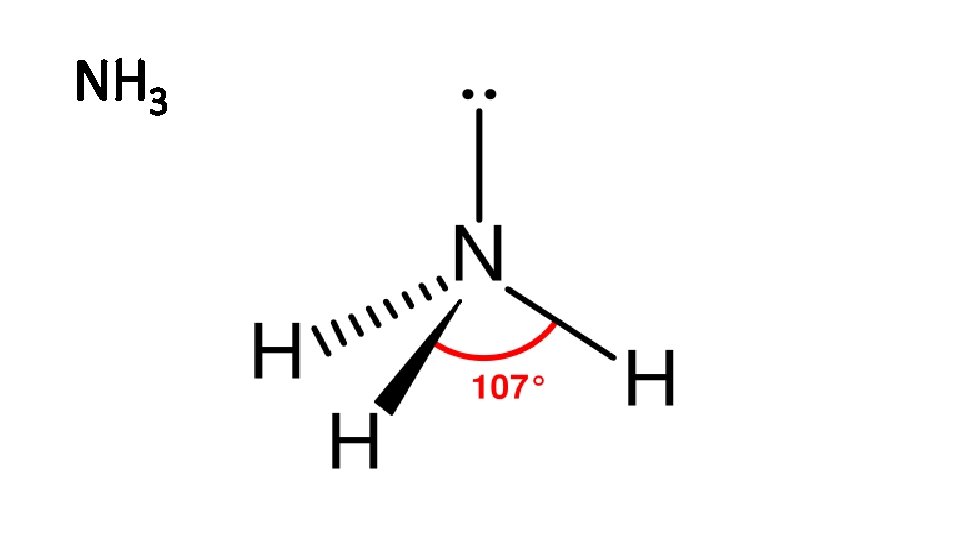
NH 3
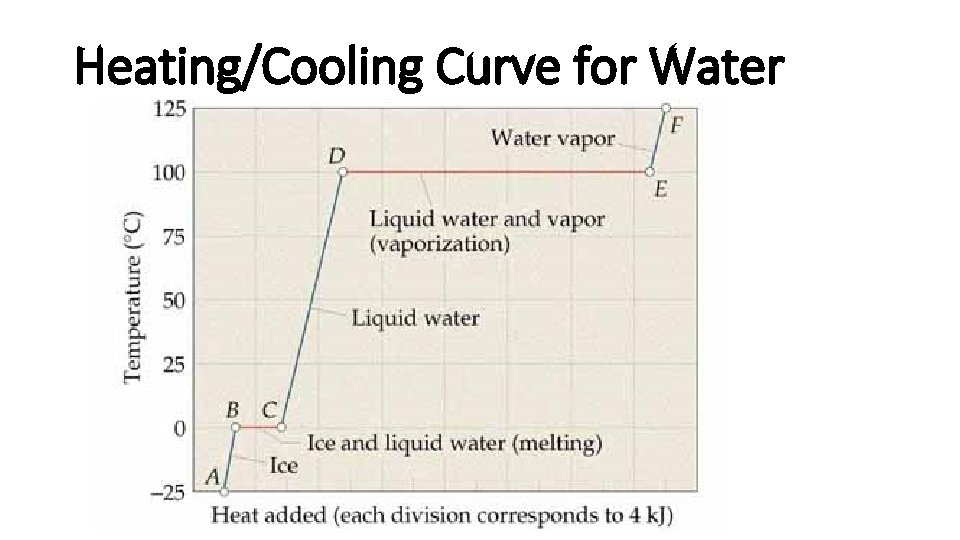
Heating/Cooling Curve for Water

What is the strongest intermolecular force present for each of the following compounds? • H 2 O • CH 3 CH 2 OH • CH 3 OH • CH 4 • NH 3 • N 2,
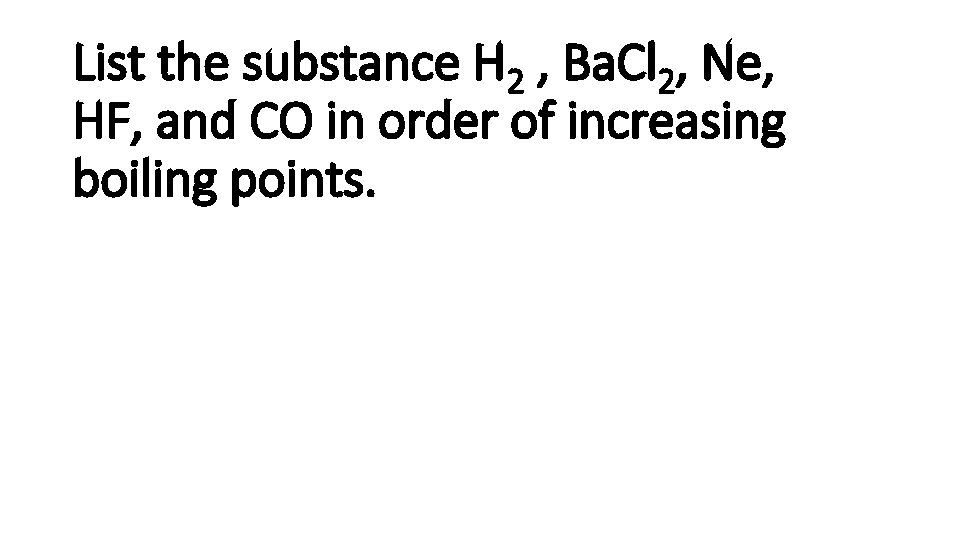
List the substance H 2 , Ba. Cl 2, Ne, HF, and CO in order of increasing boiling points.

CH 3, CH 3 CH 2 OH, CH 3 OH. • What is the intermolecular forces in each substance? • Which has the highest boiling point?

Learn With Technology • http: //www. chem. umass. edu/genchem/chem 112/MCQ_Intermolecu lar_Forces. htm http: //alpha. chem. umb. edu/chemistry/ch 115/Mridula/CHEM%20116 /documents/chapter_11 au. pdf
- Slides: 22