Solids From Bonds to Bands Atom E Molecule
Solids: From Bonds to Bands Atom E Molecule Levels Bond Band 1 -D Solid 1

• In the spirit of ‘bottom-up’ theory, we will identify minimal models to create metallic or semiconducting bands • A simple 1 -D chain of atoms with 1 electron/atom will yield a metal • A chain of dimers with 1 electron/atom will yield a semiconductor • A real 3 D solid will involve dimerization of atoms or orbitals 2
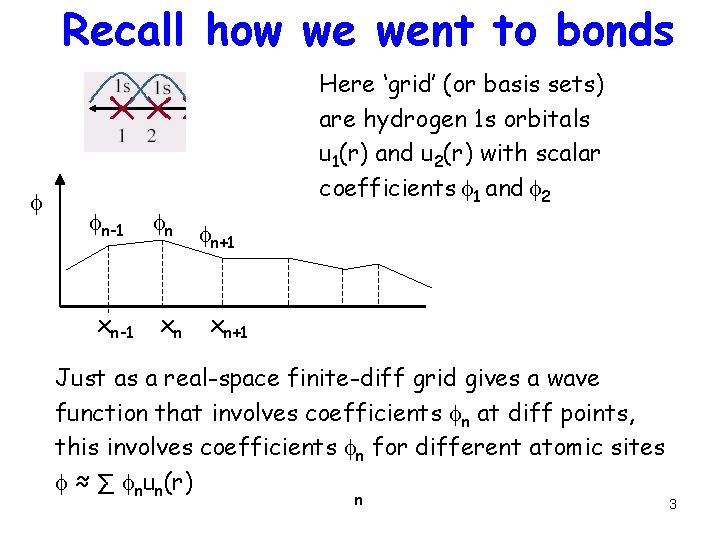
Recall how we went to bonds f fn-1 xn-1 fn xn Here ‘grid’ (or basis sets) are hydrogen 1 s orbitals u 1(r) and u 2(r) with scalar coefficients f 1 and f 2 fn+1 xn+1 Just as a real-space finite-diff grid gives a wave function that involves coefficients fn at diff points, this involves coefficients fn for different atomic sites f ≈ ∑ fnun(r) n 3

Recall how we went to bonds In this atomic basis set (rather than grid basis set), we calculate H, which becomes a matrix. The diagonal elements e = H 11 = ∫u 1(r)Hu 1(r)d. V, while the offdiagonal elements -t = ∫u 1(r)Hu 2(r)d. V H= e -t -t e in this atomic basis set Eigenvalues e ± t (ignoring non-orthogonality) Eigenvector coefficients f 1, 2 = ± 1/√ 2, ie, (u 1 u 2)/√ 2 Lower energy state occupied by electrons ± 4
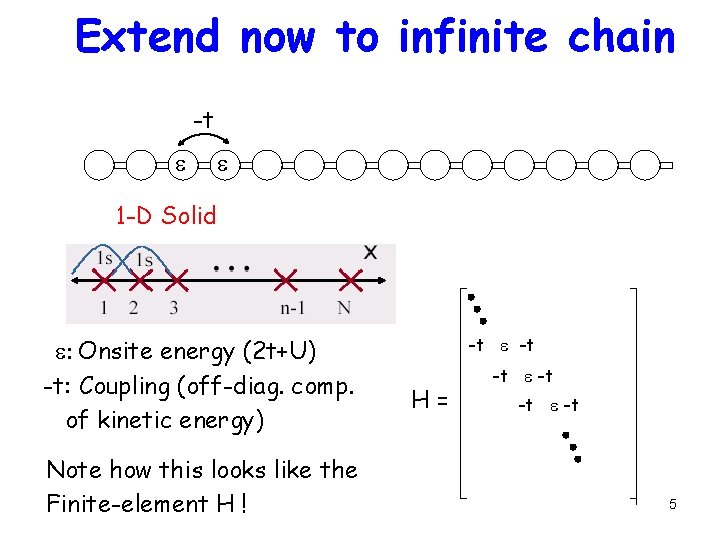
Extend now to infinite chain -t e e 1 -D Solid e: Onsite energy (2 t+U) -t: Coupling (off-diag. comp. of kinetic energy) Note how this looks like the Finite-element H ! -t e -t H= -t e -t 5
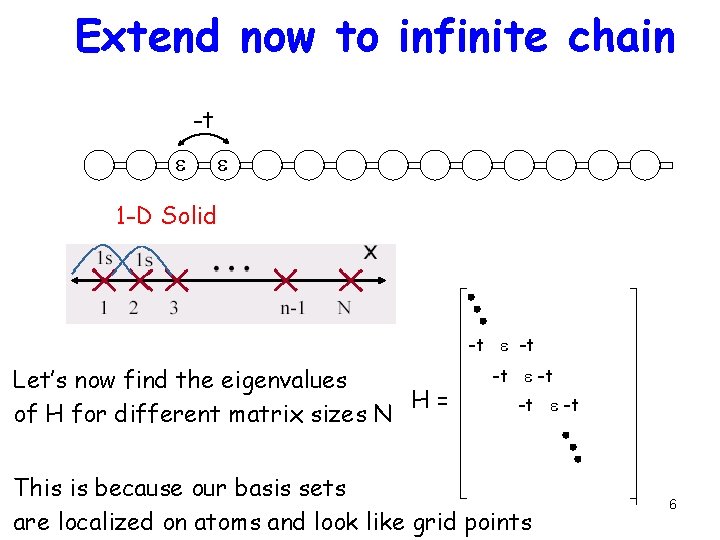
Extend now to infinite chain -t e e 1 -D Solid -t e -t Let’s now find the eigenvalues H= of H for different matrix sizes N -t e -t This is because our basis sets are localized on atoms and look like grid points 6
![Eigenspectra If we simply find eigenvalues of each Nx. N [H] and plot them Eigenspectra If we simply find eigenvalues of each Nx. N [H] and plot them](http://slidetodoc.com/presentation_image_h/2ddd725dc6af486558cd449ba3dcbc8d/image-7.jpg)
Eigenspectra If we simply find eigenvalues of each Nx. N [H] and plot them in a sorted fashion, a band emerges! Note that it extends over a band-width of 4 t (here t=1). The number of eigenvalues equals the size of [H] Note also that the energies bunch up near the edges, creating large DOS there N=2 4 6 8 10 20 50 500 7
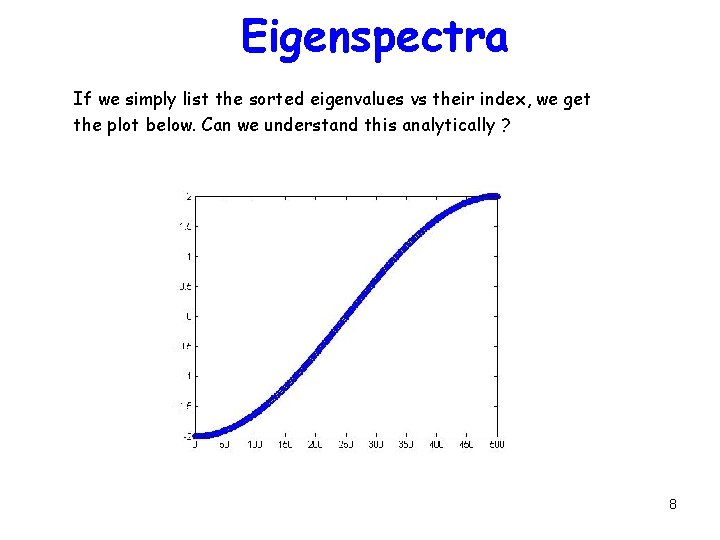
Eigenspectra If we simply list the sorted eigenvalues vs their index, we get the plot below. Can we understand this analytically ? 8
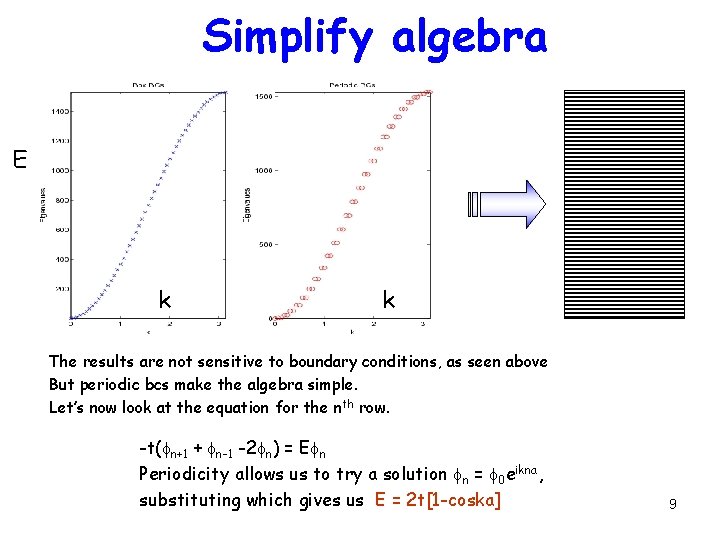
Simplify algebra E k k The results are not sensitive to boundary conditions, as seen above But periodic bcs make the algebra simple. Let’s now look at the equation for the nth row. -t(fn+1 + fn-1 -2 fn) = Efn Periodicity allows us to try a solution fn = f 0 eikna, substituting which gives us E = 2 t[1 -coska] 9
![Band properties E = 2 t[1 -coska] gives us a band of states. (a) Band properties E = 2 t[1 -coska] gives us a band of states. (a)](http://slidetodoc.com/presentation_image_h/2ddd725dc6af486558cd449ba3dcbc8d/image-10.jpg)
Band properties E = 2 t[1 -coska] gives us a band of states. (a) Need to go only upto ka = p (Brillouin Zone) f(x) f 1 f 3 f 2 2 p/a x 1 x 2 f 4 x 3 Equiv states as far as coeffs go x 4 (b) Band bottom at k=0 (value 0) and top at ka=p (value 4 t) (c) Bandwidth = 4 t depends on overlap between atoms 10
Band properties + + Periodic nuclear potential 11
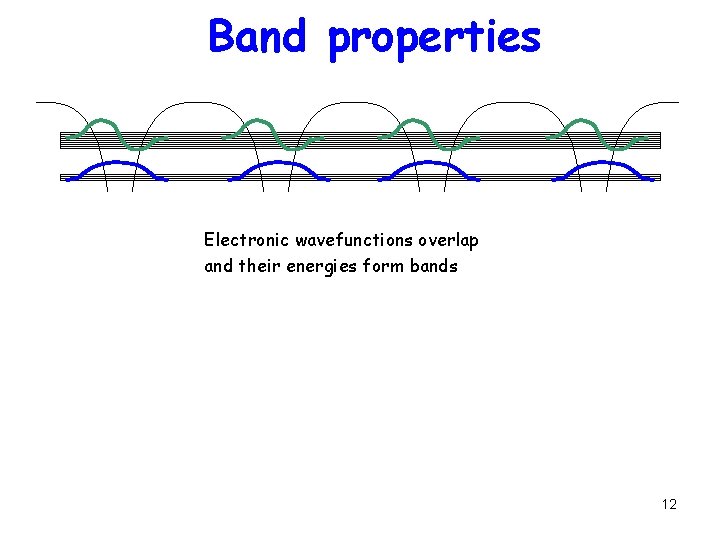
Band properties Electronic wavefunctions overlap and their energies form bands 12

Band properties Shallower potentials give bigger overlaps. Greater overlap creates greater bonding-antibonding splitting of each multiply degenerate level, creating wider bandwidths Since shallower potentials allow electrons to escape easier, they correspond to smaller effective mass Thus, effective mass ~ 1/bandwidth ~ 1/t (t: overlap) 13

Effective Mass Near bottom of band, can fit a parabola But instead of using a free electron mass to calculate t (chapter 2), we are using a differently computed t (overlap of basis sets un) to extract m*. This does not need to match the free electron mass!! In fact it incorporates nuclear attraction terms effectively. • • So Effective Mass m* = ħ 2/(∂2 E/∂k 2) ~ 1/curvature of E-k In case of our 1 -D band E=2 t(1 -coska), m* = ħ 2/2 ta 2 (~ 1/t, as argued earlier) Electronic properties at low bias depend on these effective masses Eg. Ga. As m* = 0. 067 me, Si ml*=0. 91 me, mt* = 0. 19 me 14

Effective Mass m* = ħ 2/(∂2 E/∂k 2) Larger E-k curvature means smaller m* (Steeper bowl in k-space gives mobile electrons) Is that sensible? Recall that x and k are Fourier transforms This means that a steeper parabola in k-space actually corresponds to a shallower bowl-shaped potential in real space, which an electron can easily get out of. This corresponds to a more mobile electron with a smaller m* and a larger band-width t. 15
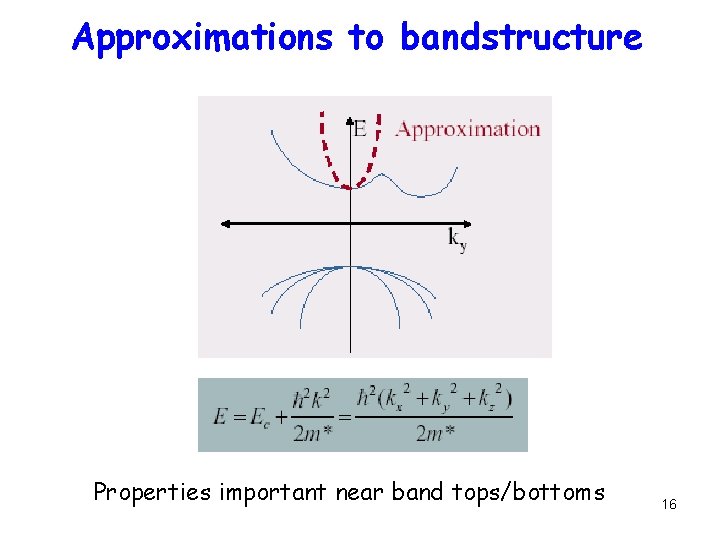
Approximations to bandstructure Properties important near band tops/bottoms 16

Filling these band states Periodic bcs mean exp(ik. L) = exp(ik. Na) = 1 k. L = 2 pn (n=0, 1, 2, …, N-1) N sites give N states, and N electrons 17
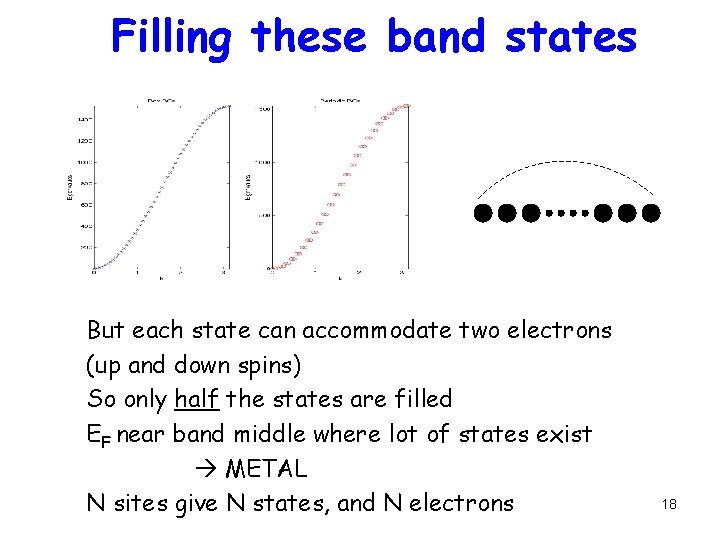
Filling these band states But each state can accommodate two electrons (up and down spins) So only half the states are filled EF near band middle where lot of states exist METAL N sites give N states, and N electrons 18
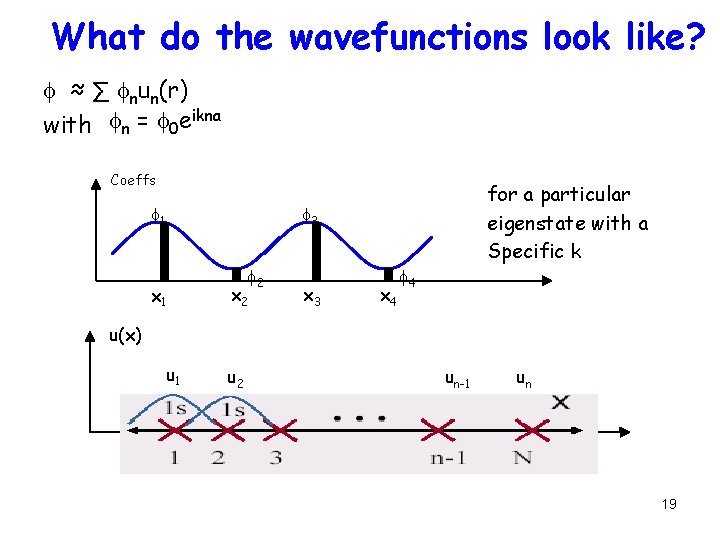
What do the wavefunctions look like? f ≈ ∑ fnun(r) with fn = f 0 eikna Coeffs f 1 for a particular eigenstate with a Specific k f 3 x 1 f 2 x 3 x 4 f 4 u(x) u 1 x 1 u 2 un-1 x 2 f 2 x 3 un x 4 f 4 19
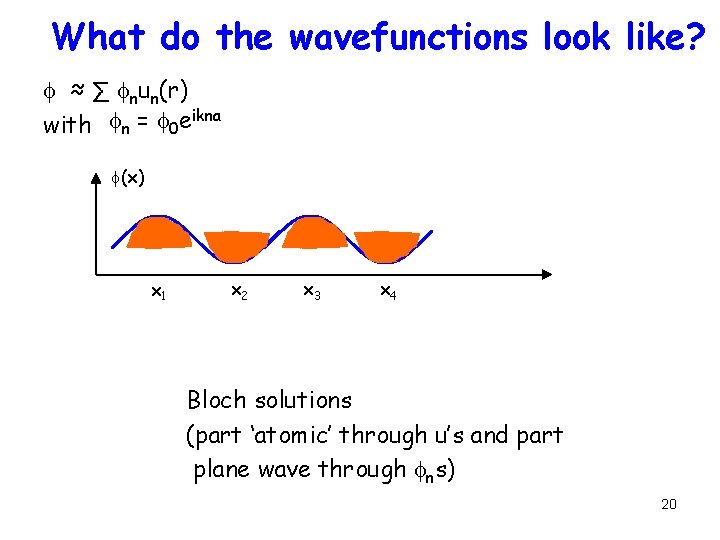
What do the wavefunctions look like? f ≈ ∑ fnun(r) with fn = f 0 eikna f(x) x 1 x 2 x 3 x 4 Bloch solutions (part ‘atomic’ through u’s and part plane wave through fns) 20
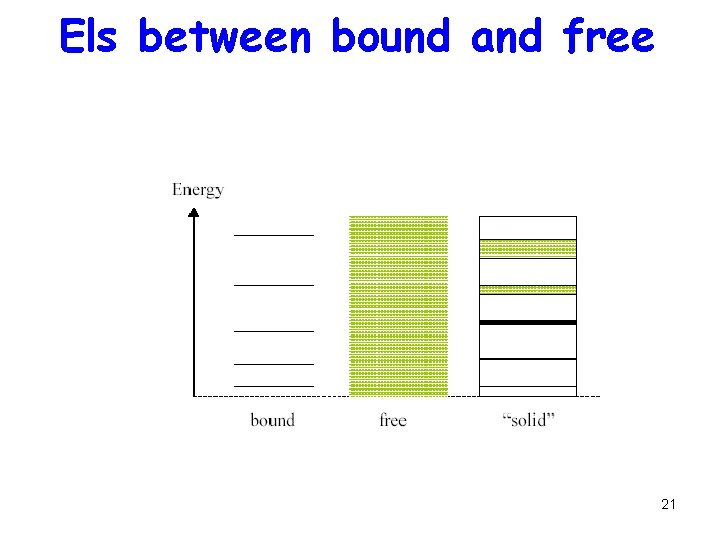
Els between bound and free 21
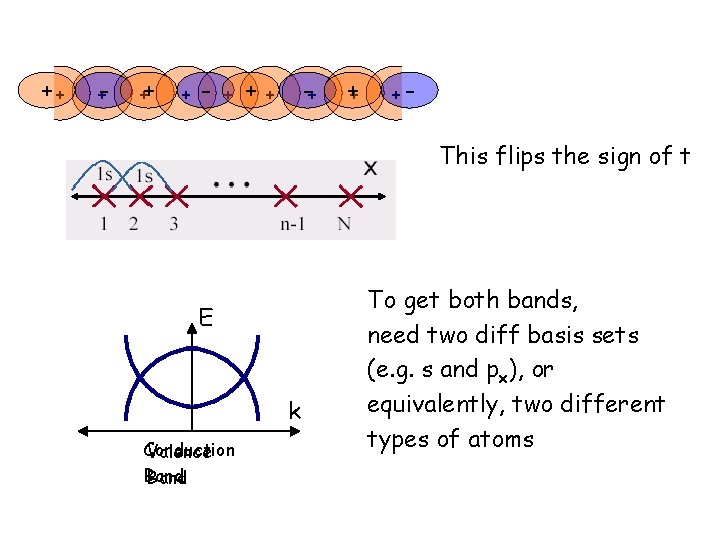
++ +- ++ + - + ++ -+ ++ +- This flips the sign of t E k Conduction Valence Band To get both bands, need two diff basis sets (e. g. s and px), or equivalently, two different types of atoms
- Slides: 22