Solid Solutions Sometimes impurities are desirable example sterling




Solid Solutions Sometimes impurities are desirable, example, sterling • silver, 7. 5% Cu and 92. 5% Ag. The copper makes silver stronger and harder. • Zn is added to Cu in brass, which is stronger, harder and • more ductile than copper. Both alloys are solid solutions in which the original • structures of the metals are changed. Solid solutions consist of a solute and a solvent • Solute = small amount, solvent = large excess •
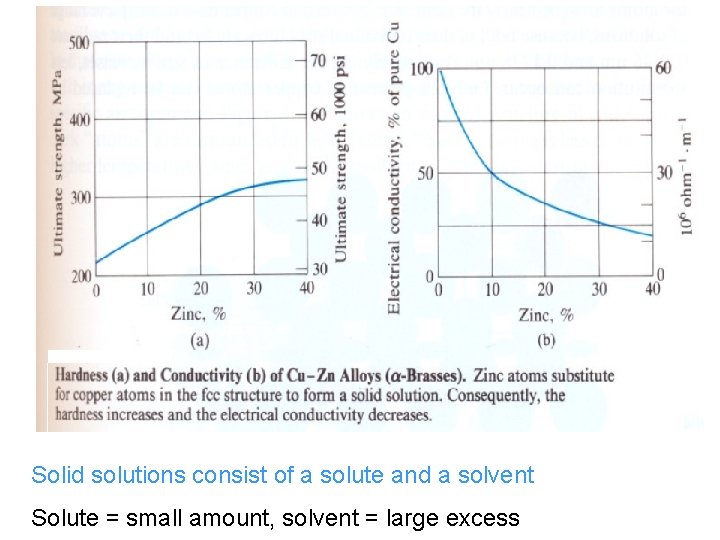
Solid solutions consist of a solute and a solvent Solute = small amount, solvent = large excess

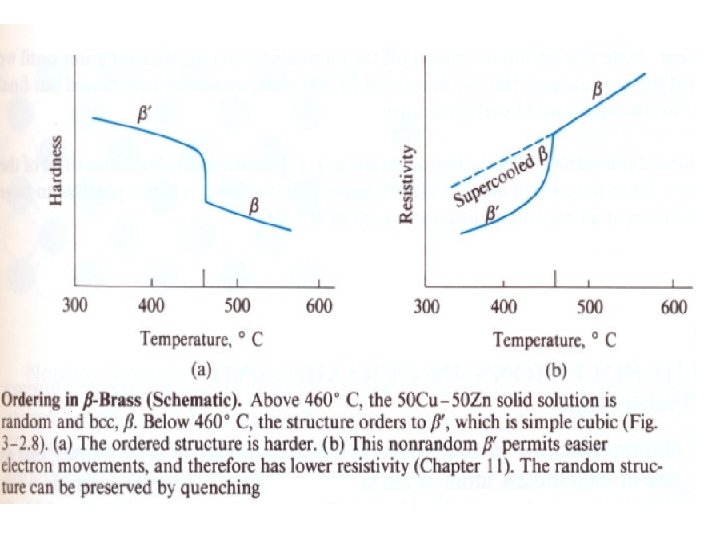
Types of solid solution Substitutional (1 Solute atoms replace solvent atoms. • It can be formed with limited solubility • e. g. Bronze (Sn in Cu) or unlimited solubility e. g. Brass (Zn in Cu), in this case both atoms have nearly the same size. 2) Interstitial solid solutions Small atoms located at the interstices between larger atoms. E. g. C in Fe 3) Ordered solid solutions The two types of atoms arranged in a specific order. Heating always destroys the ordering in the solution.

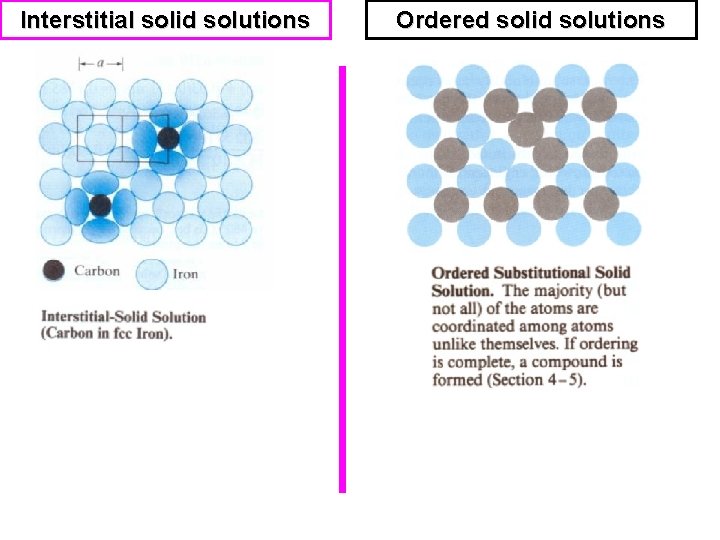
Interstitial solid solutions Ordered solid solutions

Example 4 -4. 1 Bronze is a solid-solution alloy of Cu and Sn in which 3%, more or less, of Cu atoms are replaced by tin atoms. The fcc unit cell of copper is retained, but is expanded a bit, because the RSn ~ 0. 151 nm. (a) What is the weight percent in a 3 a/o tin bronze? (b) Assuming that the lattice constant increases linearly with the atomic fraction of tin, what is the density of this bronze? Procedure Select a basis for calculation. 100 atoms (25 fcc unit cells) is a natural; thus, 97 Cu + 3 Sn. (a) Determine the masses. (b) Since = m/V, determine ā for V= a 3 and use the calculated mass.


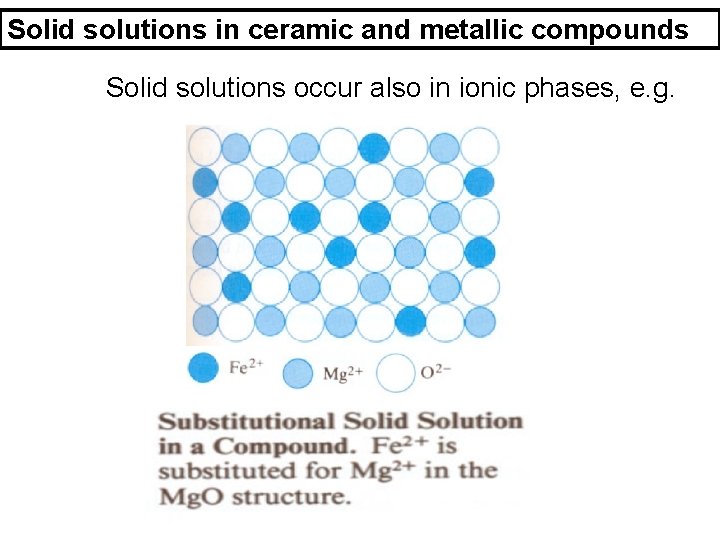
Solid solutions in ceramic and metallic compounds Solid solutions occur also in ionic phases, e. g.

Nonstoichiometric Compounds Simple compounds are stoichiometric • Other compounds deviate from the stoichio- • metric ratio, e. g. Cu 2 Al includes 31 a% to 37 a% Al (16 -20 w% Al), and not 33. 5 a% Al Fe. O is not 50% a% oxygen. • These compounds are present as solid solutions. •
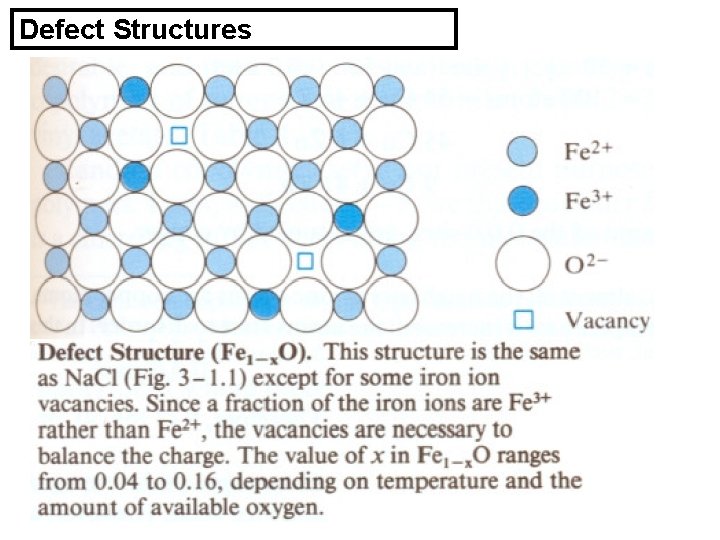
Defect Structures
- Slides: 17