Solid and Gas Solubility What is Solubility Simply

Solid and Gas Solubility

What is Solubility? Simply put, solubility is dissolving one thing into something else. n Sugar in water, salt in water. Specifically, it is the maximum amount of solute that will dissolve in a certain amount of water.
Solubility Details Remember, solute is what you are dissolving and solvent is what is doing the dissolving. The amount of solute is expressed in grams. The amount of solute dissolved is dependant on the temperature of the solvent.

Lets say it again! If you change the temperature of the solvent, the amount of solute that will dissolve will also change. If you change the amount of solvent, the amount of solute that will dissolve will change.
How long does it take? Many factors affect the amount of time it takes for a solute to dissolve.
How long does it take to dissolve? 1. How soluble the solute is. 2. How big the crystals are. 3. How much you stir. 4. How long you stir the solution.

How long does it take to dissolve? These factors do not affect the total amount of solute that will dissolve, only the time it takes to dissolve. For example, stirring rapidly and for an extended time period does not change how much sugar you can dissolve in Kool-aid, only how long it takes for the sugar to dissolve.

A Specific Amount Because there are specific amounts of solutes that can be dissolved in solvents, there are 3 definitions that we can now use to describe solutions. 1. Unsaturated 2. Saturated 3. Supersaturated

Unsaturated Solutions If a solution is unsaturated, then the solvent can dissolve more solute at that temperature.

Saturated Solutions If a solution is saturated, then it has dissolved the exact amount it can at that given temperature.

Supersaturated Solutions If a solution is supersaturated, then it is has dissolved more solute than a saturated solution has. This is difficult to achieve. Usually requires heating and then slowly cooling the solution. Will eventually release some dissolved solute to return to a saturated state.

How can we tell the kind of solution? In order to know when our solutions are saturated or not, we have to know the solubility of the solute. To do this, we look at a solubility curve.

Solubility Curves A solubility curve graphs the temperature of the solvent versus the grams of solute dissolved in a given amount of solvent. This curve shows the solubility for a range of temperatures.

Solubility Curves In addition to showing us saturated solutions, they also can be used to determine if a solution is supersaturated and unsaturated.
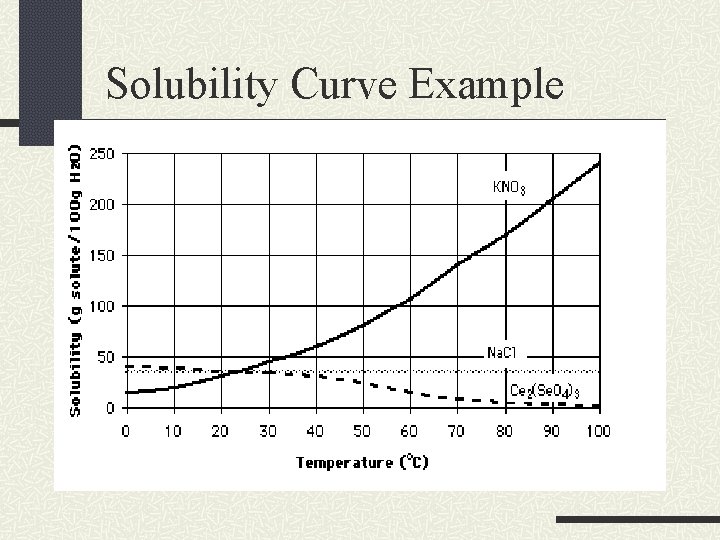
Solubility Curve Example
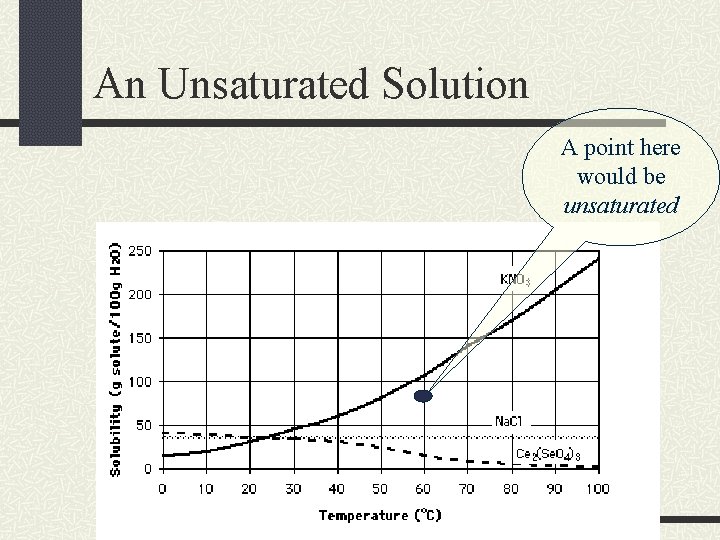
An Unsaturated Solution A point here would be unsaturated
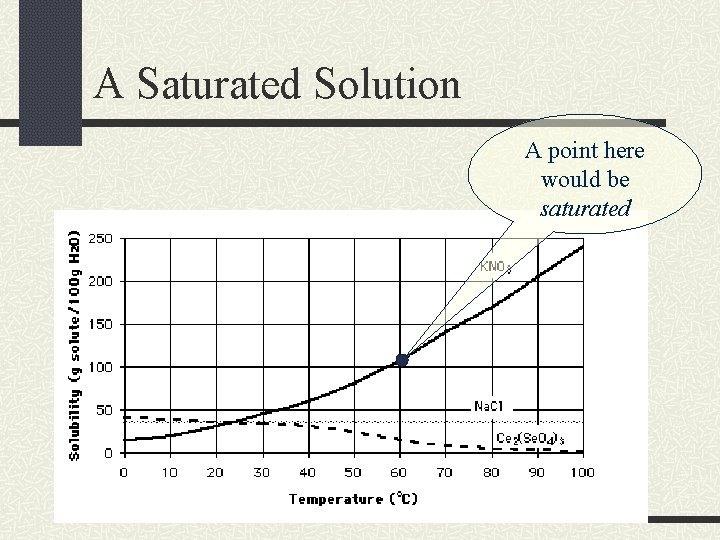
A Saturated Solution A point here would be saturated
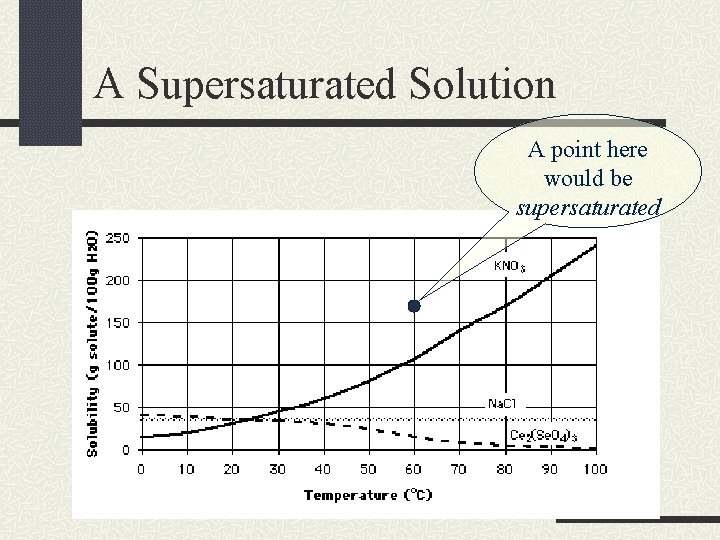
A Supersaturated Solution A point here would be supersaturated

Solid and Gas Solubility The 3 phases of matter all behave differently with solubility. We will discuss how solids and gases differ in solubility.

Solid Solubility By studying solubility graphs, a general trend can be seen. As you increase temperature, the solubility also increases. At higher temps, you can dissolve more.
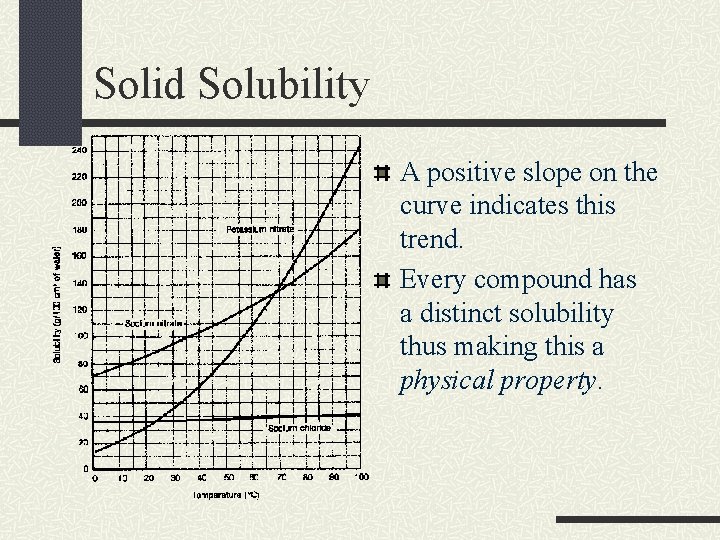
Solid Solubility A positive slope on the curve indicates this trend. Every compound has a distinct solubility thus making this a physical property.

Gas Solubility As with solids, temperature affects the solubility of gases. But, the trend is opposite, as you increase the temperature, the solubility decreases.

Gas Solubility In addition to temperature affecting gas solubility, gas pressure also determines the solubility of a gas. Gas solubility is directly proportional to the gas pressure. The higher pressure, the more gas that will dissolve.

A Good Example Carbonated beverages contain dissolved CO 2. When you open a can of soda, the pressure is decreased and the amount of CO 2 dissolved also decreases. That’s why you see bubbles coming up.

Solution Concentration
- Slides: 25