Solar energy and solar cells As another renewable

Solar energy and solar cells As another renewable energy

Photovoltaic power generation l Solar cells utilizes the photovoltaic effect with the related semiconductors. l Solar energy comes on the surface of the earth and the maximum flux density per 1 m 2 is 1. 366 k. W, which is called the solar constant. l Most of the materials to produce solar cells are abundant on the earth.
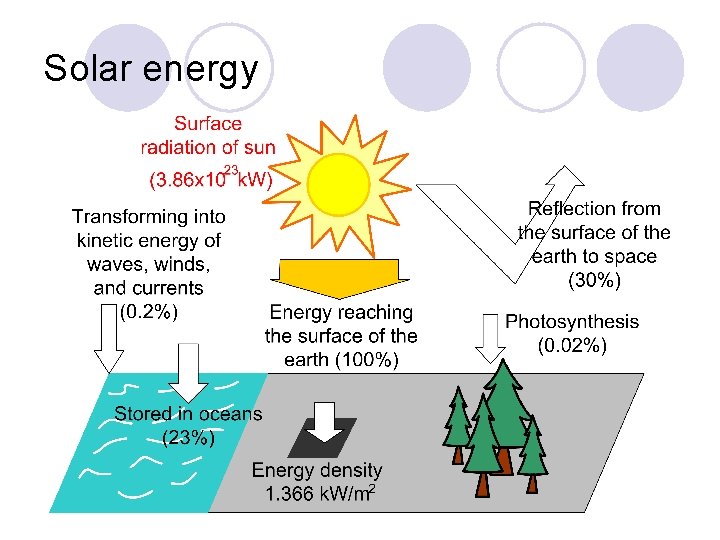
Solar energy

Disadvantages of photovoltaic power l The cost per kilowatt is relatively high. l The energy density is low. (It needs a large space. ) l It depends on hours of sunlight. l It requires an extra system to store electricity.
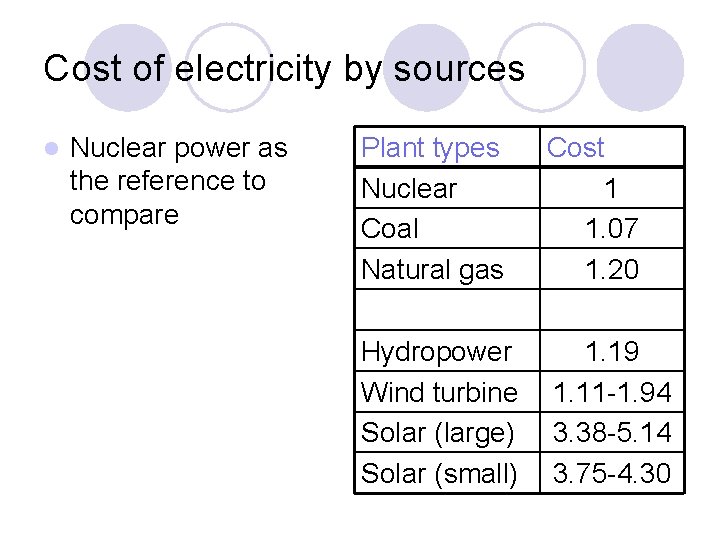
Cost of electricity by sources l Nuclear power as the reference to compare Plant types Nuclear Coal Natural gas Cost 1 1. 07 1. 20 Hydropower Wind turbine Solar (large) Solar (small) 1. 19 1. 11 -1. 94 3. 38 -5. 14 3. 75 -4. 30

Analysis of sunlight l Contents of spectra ¡ Visible rays (47%); Infrared rays (46%); and Ultraviolet rays (7%) l Solar cells and sunlight spectra silicon absorbs 0. 4 m – 1. 2 m. ¡ Amorphous silicon absorbs below 0. 8 m. ¡ Crystallized
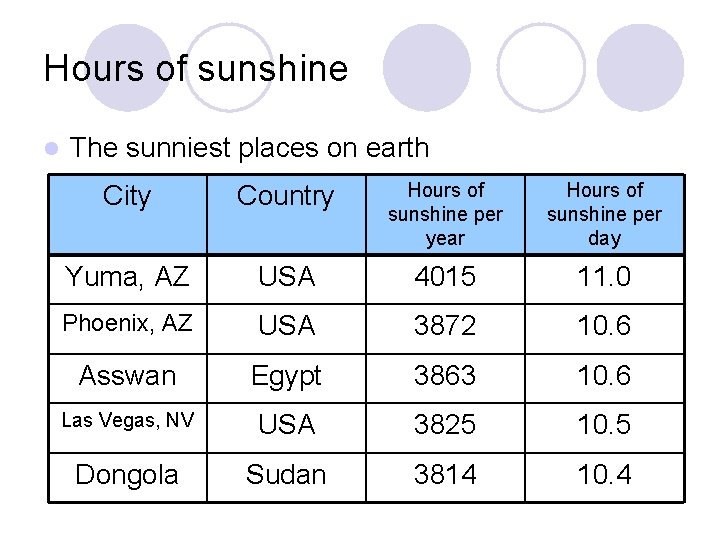
Hours of sunshine l The sunniest places on earth City Country Hours of sunshine per year Hours of sunshine per day Yuma, AZ USA 4015 11. 0 Phoenix, AZ USA 3872 10. 6 Asswan Egypt 3863 10. 6 Las Vegas, NV USA 3825 10. 5 Dongola Sudan 3814 10. 4

Payback time l Energy Pay-Back Time (EPBT) ¡ EPT is defined as (entire energy spent through the life cycle) (the energy produced for one year). l CO 2 Pay-Back Time (CO 2 PBT) ¡ CO 2 PT is defined as (entire CO 2 produced through the life cycle) (the CO 2 reduced by introducing a renewable energy system for one year). Most of solar cells have 1. 4 to 2. 7 years for CO 2 PBT.
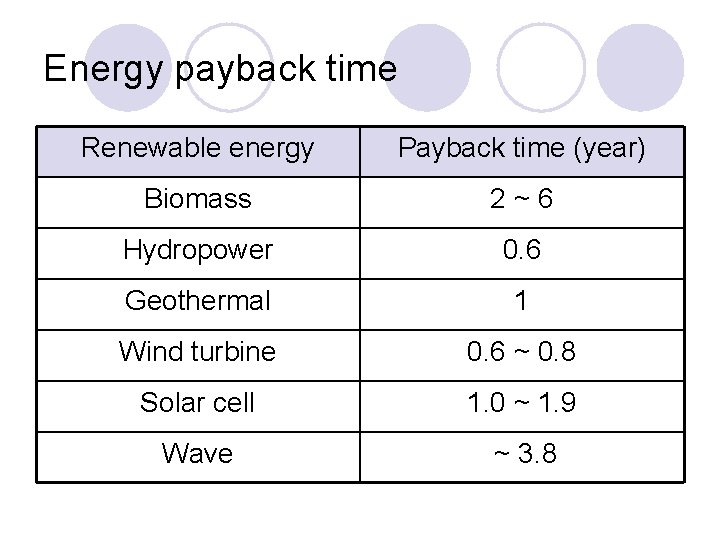
Energy payback time Renewable energy Payback time (year) Biomass 2~6 Hydropower 0. 6 Geothermal 1 Wind turbine 0. 6 ~ 0. 8 Solar cell 1. 0 ~ 1. 9 Wave ~ 3. 8

Solar cells and semiconductors 1 l Solar cells are made of semiconductors. ¡ N-type l Extra electrons become carriers to make current flows. ¡ P-type l semiconductor: Holes are become carriers for current. ¡ I-type l semiconductor: There is no extra electrons or holes. It needs certain heat or light energy to produce extra electrons from the covalent bonds.

Solar cells and semiconductors 2 l PN junction (single crystal) ¡A basic structure of solar cells – The efficiency is about 24. 2%. l PIN junction (thin film) ¡ I-type is sandwiched by p-type and n-type semiconductors. – The efficiency is about 10%, but it is inexpensive.

Materials for solar cells (semiconductors) l Silicon l Boron l Phosphorus l Titanium dioxide l Gallium – arsenate l Cadmium – tellurium, etc.
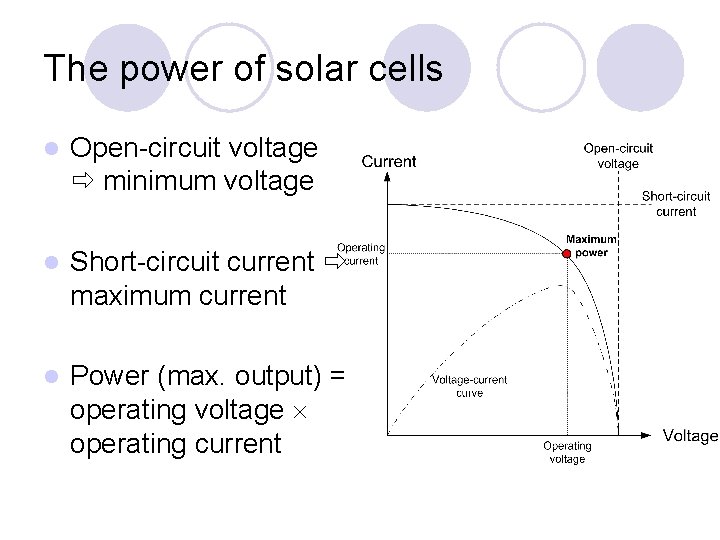
The power of solar cells l Open-circuit voltage minimum voltage l Short-circuit current maximum current l Power (max. output) = operating voltage operating current

Why the efficiency cannot be 100% l Light is reflected on the surface of the cell. l Some of light transmits through the cell. l It cannot absorb all of the wavelengths of sun light. l Part of carriers occur pair annihilation. l Inside solar cells contain electric resistance.
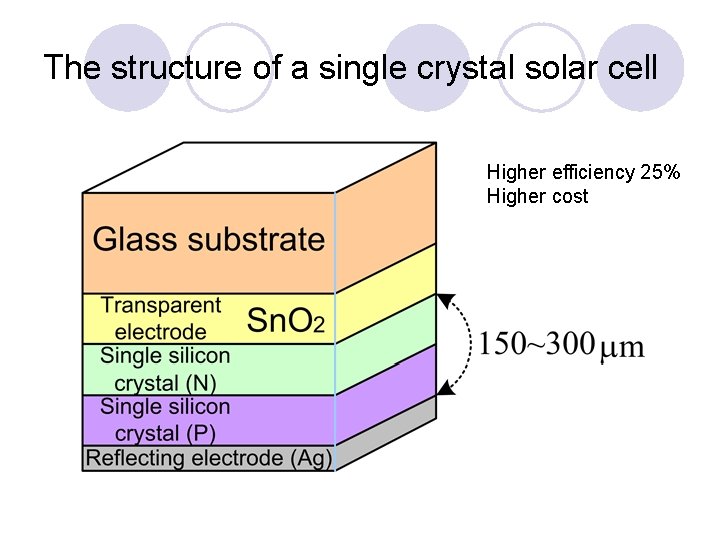
The structure of a single crystal solar cell Higher efficiency 25% Higher cost
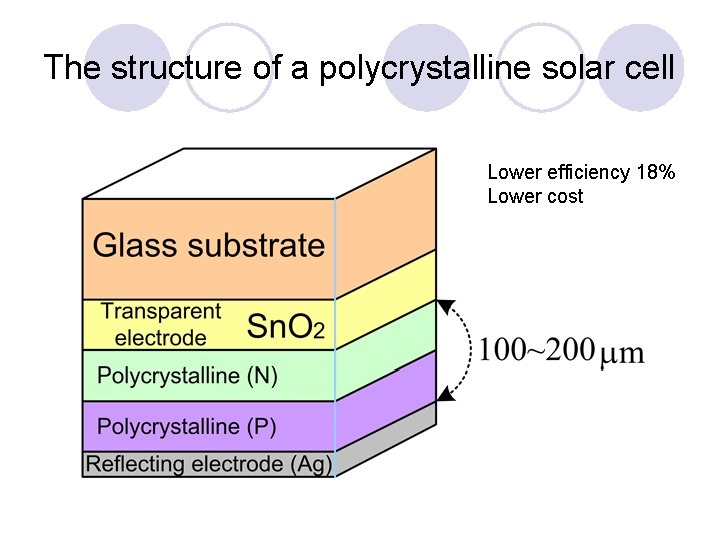
The structure of a polycrystalline solar cell Lower efficiency 18% Lower cost

Amorphous silicon (a-Si) solar cell Amorphous means non-crystallized. l Rate of absorption is large. l ¡ This is because of random configuration of atoms. One can use various substrates and produce thinner solar cells. l Photo deterioration of a-Si reduces the output by 10%. l However, the output under high temperatures is better than the others. l ¡ High temperature improves photodeterioration.
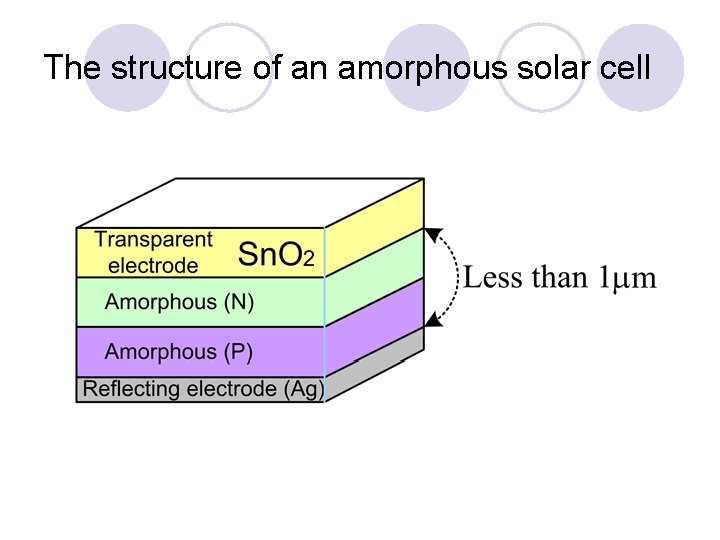
The structure of an amorphous solar cell

Microcrystalline silicon ( -Si) cell l Photo deterioration is small. l Absorption rate is higher within wavelengths of sun light. l The efficiency is about 10%. l The thickness is 2 ~ 3 m. l The overall properties of -Si are between crystallized and amorphous Si.

Multi-junction silicon solar cell l These are more efficient. ¡ Silicon-type: 20% or more; Compound-type: 35% or more l These can absorb more wavelengths due to multiple materials. l Multiple structure makes it complicated and increases internal losses of various properties. l Multiple junction makes it bulky and heavy.
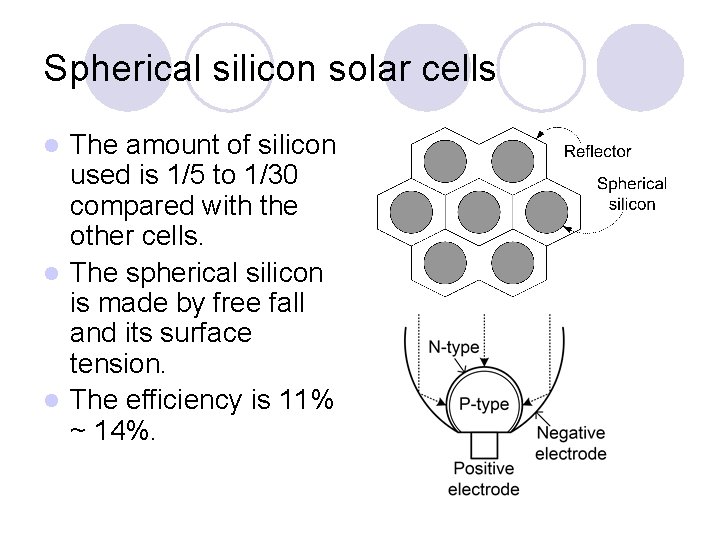
Spherical silicon solar cells The amount of silicon used is 1/5 to 1/30 compared with the other cells. l The spherical silicon is made by free fall and its surface tension. l The efficiency is 11% ~ 14%. l
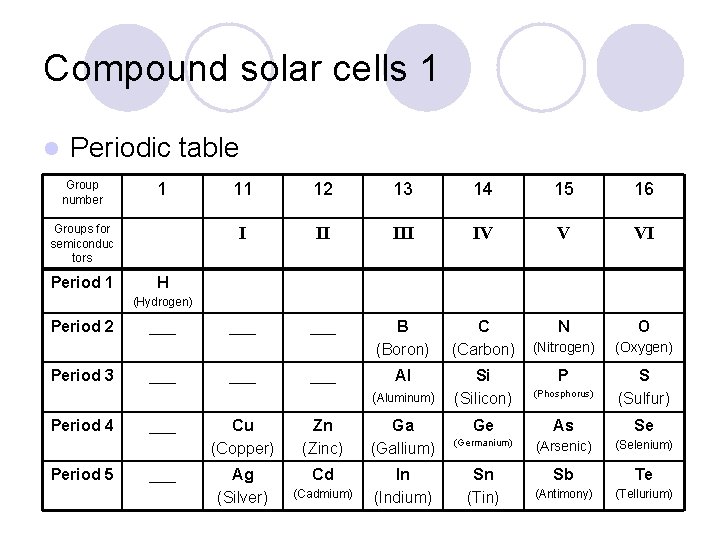
Compound solar cells 1 l Periodic table Group number 1 Groups for semiconduc tors Period 1 11 12 13 14 15 16 I II IV V VI ___ B (Boron) C (Carbon) N O (Nitrogen) (Oxygen) Al Si (Silicon) P S (Sulfur) H (Hydrogen) Period 2 Period 3 ___ ___ (Aluminum) Period 4 Period 5 ___ Cu (Copper) Zn (Zinc) Ga (Gallium) Ag (Silver) Cd In (Indium) (Cadmium) (Phosphorus) Ge As Se (Germanium) (Arsenic) (Selenium) Sn (Tin) Sb Te (Antimony) (Tellurium)

Compound solar cells 2 l If the total of the number of electrons in the valence band is multiples of 8, it becomes a stable crystal. Si-Si has: 4 + 4 = 8. l Ga-As has: 3 + 5 = 8. l Cu(In, Ga)Se 2 has: 1+3+6 2=16. l Cu 2 Zn. S 4 has: 1 2+2+4+6 4=32 l

Compound solar cells 3 (Classification) l Silicon-diamond structure l III-V: Zincblende structure (In, Ga, As, P) l II-VI: Zincblende structure (Cd, Te, S) l I-III-VI: Chalcopyrite structure CIS (Cu, In, Se) and CIGS (Cu. In, Ga, Se) l I-II-IV-VI: (Cu, Zn, S, Se)

Compound solar cells 4 (Example) l Examples of compound semiconductors III-V group II-VI group Binary compound Al. As Ga. As In. P In. As Al. Sb Ga. P Al. P Zn. S ZSe Cd. Te Cd. S Zn. Te Ternary compound Al. Ga. As. P Ga. In. P Al. Ga. Sb Zn. SSe Cd. Zn. Te Quaternary compound In. Ga. As. P In. Ga. Al. P In. Al. Ga. As In. Ga. As. Sb I-III-VI group CIS: Cu. In. S 2 CIGS: Cu(In, Ga)Se 2

Compound solar cells 5 l The most efficient ones are Ga. As and In. Ga. As solar cells. (35. 8%) l The theoretical efficiency of compound multi-junction solar cells: one layer = 37%; two layers = 50%; three layers = 56%; and 36 layers = 72%

Concentrator Photo Voltaic System l When the collection efficiency is n, the area of solar cell required is 1/n. l This system is used for more expensive semiconductors. l The current efficiency is about 40%.

Cadmium-tellurium solar cell l Lower cost l This type of solar cell reached grid parity in 2009. (Namely, the cost is equal to the electric power generation. ) l The amount of cadmium is very small and it will not harm the environment and human.

Organic-type solar cell 1 l OSC (Organic Solar Cell) ¡ Conductive polymer and fullerene are used for the surface layer. l DSC (Dye-sensitized Solar Cell) ¡ The mechanism is based on Grätzel cell. ¡ The pigments in electrolyte are positively ionized to absorb electrons with light. ¡ Fluorine-doped tin oxide (FTO) and Indium tin oxide (ITO) are used.

Organic-type solar cell 2 l This type can be printed (OSC) ¡ Evaporation method ¡ Print-on method l The efficiency is about 7%. l DSC has achieved about 10% of efficiency.

Quantum dot solar cell 1 l Confine electrons 3 dimensionally with the diameter of dozens of nanometers. l When photons hit the quantum dots, excitons are generated by the energy absorption. l Therefore, emerged. electrons and holes are
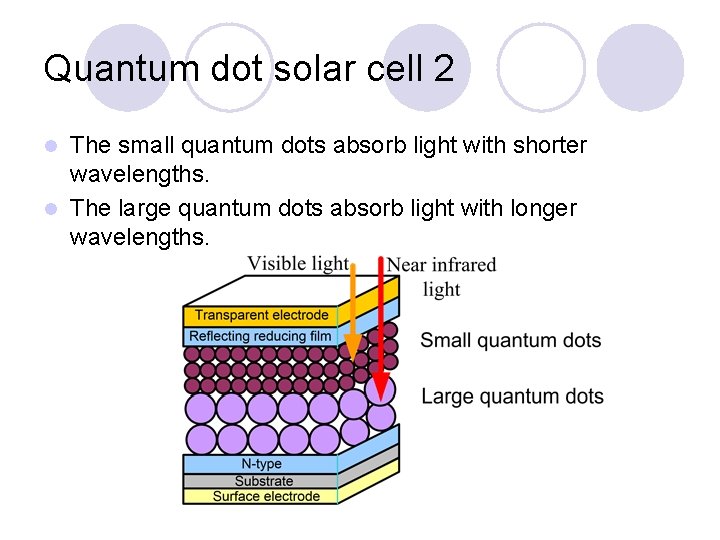
Quantum dot solar cell 2 The small quantum dots absorb light with shorter wavelengths. l The large quantum dots absorb light with longer wavelengths. l

Quantum dot solar cell 3 l This solar cell can utilizes wide range of light. l This absorbs various wavelengths by using different sizes of quantum dots to laminate the cell. l In theory, the efficiency can go up as the technique improves.
- Slides: 33