SOCIETY PERSPECTIVE PELVIC ORGAN PROLAPSE REGISTRIES Catherine Bradley

SOCIETY PERSPECTIVE: PELVIC ORGAN PROLAPSE REGISTRIES Catherine Bradley, MD September 7, 2018

Outline • Background • Pelvic Organ Prolapse – Background • Mesh in POP Surgery • AUGS Registry Experience – the Pelvic Floor Disorders Registry (PFDR) • Accomplishments • Lessons learned • POP research questions

What are pelvic floor disorders (PFDs)? • Interrelated clinical conditions: • Pelvic organ prolapse (POP) • Urinary incontinence (UI) • Fecal incontinence (FI) • Emptying abnormalities of the lower urinary and GI tracts • Almost 25% of all women and more than 1 of 3 older women report symptoms of at least one PFD (Nygaard, JAMA 2008) • 1 in 7 women will undergo POP surgery by age 80 (Wu, Ob. Gyn, 2014)

Pelvic Organ Prolapse (POP) • Descent of the pelvic organs into or through the vagina • Often accompanied by: • Vaginal bulging • Urinary symptoms • Bowel symptoms • Sexual dysfunction • 3%-6% of women have POP (vaginal/uterine descent) beyond the vaginal opening • 15 -17% of all hysterectomies done for POP; most common indication for hysterectomy in postmenopausal women

POP Treatment • Non-surgical • Observation, Behavioral modifications • Pelvic floor muscle exercises • Pessary • Surgical • Transvaginal • Native tissue repair • Transvaginal Mesh • Abdominal or Laparoscopic/Robotic • Sacrocolpopexy (includes mesh) • Obliterative (vaginal closure or colpocleisis)
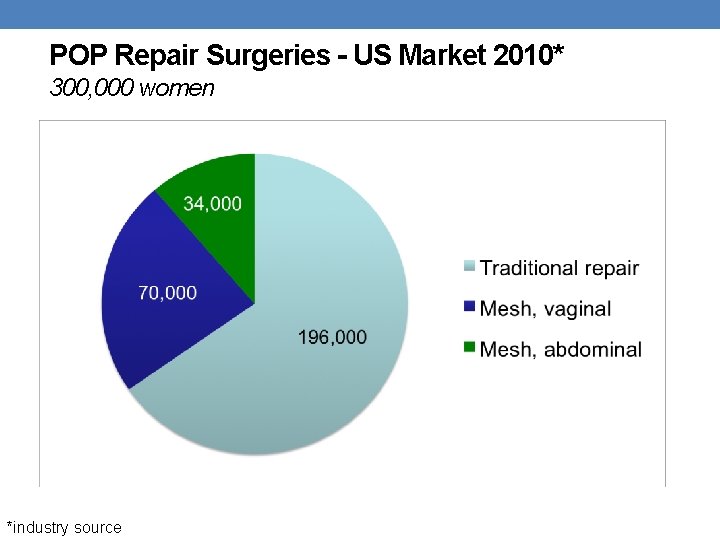
POP Repair Surgeries - US Market 2010* 300, 000 women *industry source

Transvaginal Mesh - Events • 2008 FDA Public Health Notification • 2008 AUGS began exploring development of POP registry • July 2011 FDA Updated Safety Communication on Transvaginal Mesh for POP • Sept 2011 FDA Advisory Panel convened • Nov 2011 AUGS Strategic Planning Meeting – POP registry development high priority for Society • Jan 2012 522 Orders

• Surgeons performing transvaginal placement of mesh for POP should undergo training specific to each device and have experience with reconstructive surgical procedures and a thorough understanding of pelvic anatomy. • Transvaginal placement of mesh for POP should be reserved for highrisk individuals in whom the benefits of mesh placement may justify the risk. . . • ACOG & AUGS strongly support continued audit and review of outcomes, as well as the development of a registry for surveillance for all current and future vaginal mesh implants.

Transvaginal Mesh/FDA • Jan 2012: FDA ordered postmarket surveillance studies (“ 522 studies”) by manufacturers of urogynecologic surgical mesh devices • RCT or prospective cohort recommended with comparison to transvaginal surgery without mesh (“native tissue”) • Amenable to facilitating multi-sponsor registry

POP Outcomes Registry - Development • Collaborative effort • AUGS registry leadership and experts on content, research design, and implementation • Industry partners, FDA, NICHD, ACOG, Women’s Health Registry Alliance, PFDN Advisory Panel, SUFU, AUA • Consensus on data collected • Outcomes • Shared native tissue control group • Shared non-surgical group

What is the PFDR? • Multiple POP treatment registries/databases • Designed to collect prospective data from patients undergoing treatment for pelvic organ prolapse (POP) • Intended to collect both provider and patient reported outcomes

PFDR Objectives • Evaluate effectiveness, QOL and safety of POP surgery • Assess effectiveness and QOL associated with non- surgical management (pessary) of POP • Provide a framework for clinical studies to be conducted within the registry, including industry-sponsored studies required to fulfill the FDA’s request for postmarketing surveillance for transvaginal mesh for POP • Allow surgeons to track patients, outcomes, adverse events, quality measures

What did we accomplish? • Development • Data Access/Legal Agreements • Common core dataset and protocol • Shared native tissue group • Common primary outcome, adverse outcomes • ~700 common data elements • Functioning database • Scope • 4 Industry Partners: 7 protocols (522) • AUGS Quality and Research protocols

PFD Registry - Timeline • Legal agreements - 2012 • Data collection and database requirements 2012 -2013 • User acceptance testing 2013 • Launch • Industry protocols 2014 • AUGS quality/research protocols 2015

PFDR – Current Stats • 4 active Industry protocols • 1 active AUGS Research protocol • Enrollment ended, 3 -year follow-up ongoing • > 700 total PFDR sites • > 3500 total PFDR participants • AUGS Research Registry • 11 active sites • 1, 159 participants • 820 Surgery • 339 Pessary

PFDR Challenges - Development • Collaboration of “Rivals” • Making a Registry Platform also function/meet requirements for a pre-market approval study • 9 protocols (7 industry-sponsored, AUGS research, AUGS quality) • Thousands of volunteer hours

PFDR Challenges – Use, Longevity • Double data entry • Difficult/costly to make changes in platform • Maximizing patient participation • Long-term patient follow-up • Long-term funding

POP Registry Planning • More focused objective for POP core minimum dataset • Device effectiveness, safety • Quality measure requirements • Opportunity for modular expansion • Research questions that require more detailed data collection • Changes in device indications • Direct data collection from electronic medical record • User-friendly platform

High Priority POP Research Questions Surgical factors: • Does a concomitant hysterectomy impact outcomes? • Are native tissue repairs really equivalent to mesh repairs in terms of durability? • When is an apical prolapse repair absolutely necessary? Patient factors: • What patient factors will modify risks and recurrence rates? • What outcomes best match the patient experience? Surgeon factors: • Does fellowship training make a difference in outcomes?
- Slides: 19