SOAR 2007 Energy and Temperature Energy Temperature Insolation



















- Slides: 56

SOAR 2007 Energy and Temperature

Energy & Temperature ü Insolation þ Spectrum þ Distribution with latitude þ Distribution with albedo ü Interaction with Air, Land & Water þ Reflected þ Absorbed by atmosphere þ Absorbed by land þ Absorbed by water ü Effect of insolation þ Heating & temperature

Temperature Scales (Again!) ü Celsius þ 0 - fresh water freezes þ 100 - fresh water boils at 1013. 2 h. Pa (1 atm) ü Kelvin Plots for different gases meet at -273 C Pressure þ Based on gases ý T C + 273 = TK -273 Temperature C üFahrenheit – used only in US þ 0 ~ salt water freezes þ 100 ~ human body temp. ý T C = (5/9)(T F - 32) ý T F = (9/5)T C + 32) Celsius – Fahrenheit touch points: -40 C = -40 F 16 C = 61 F 28 C = 82 F

Temperature Scales Celsius – Fahrenheit touch points: 28 C ≈ 82 F 16 C ≈ 61 F -40 C = -40 F “At 40 below it doesn’t matter!”

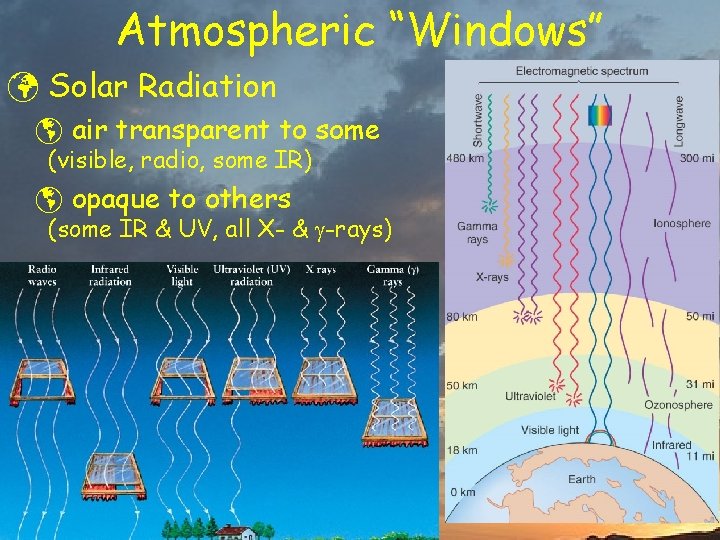
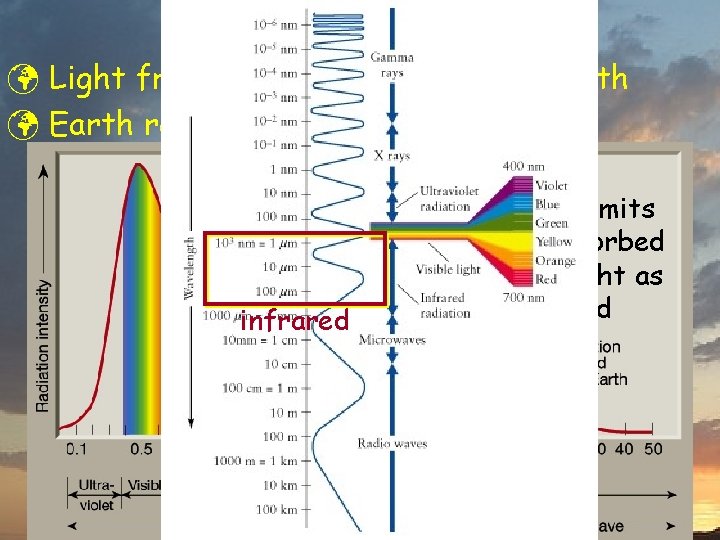
Atmospheric “Windows” ü Solar Radiation þ air transparent to some (visible, radio, some IR) þ opaque to others (some IR & UV, all X- & -rays)

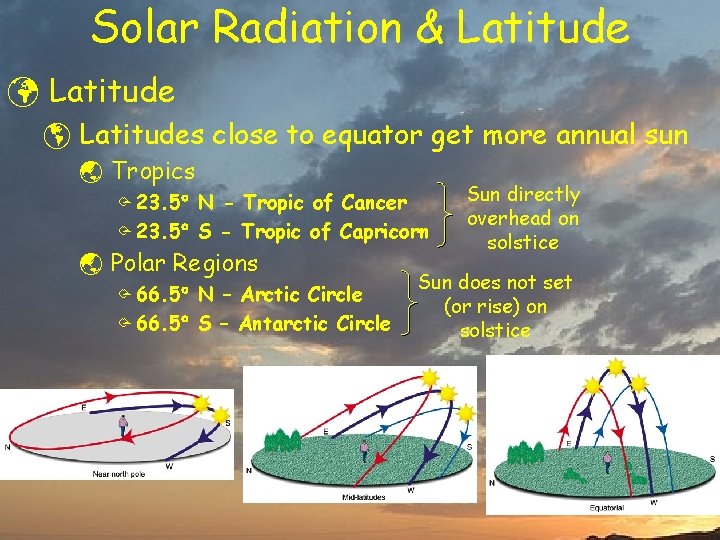
Solar Radiation & Latitude ü Latitude þ Latitudes close to equator get more annual sun ý Tropics Ö 23. 5 N - Tropic of Cancer Ö 23. 5 S - Tropic of Capricorn ý Polar Regions Ö 66. 5 N – Arctic Circle Ö 66. 5 S – Antarctic Circle Sun directly overhead on solstice Sun does not set (or rise) on solstice

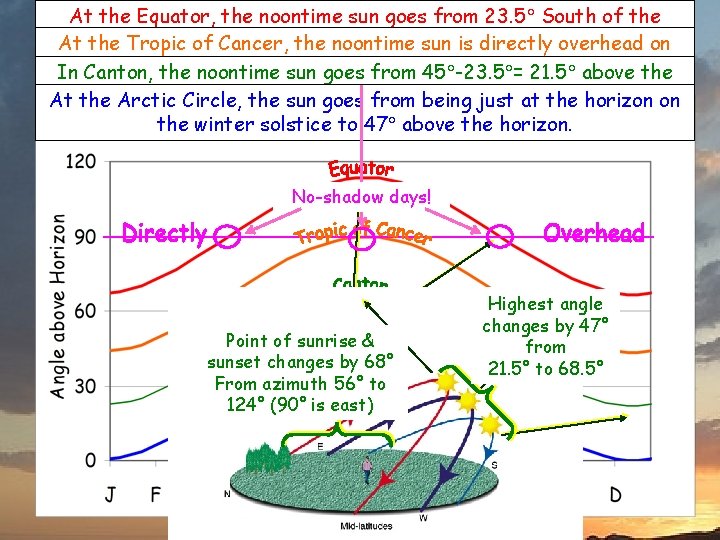
Sun Angle & Latitude At the Equator, the noontime sun goes from 23. 5 South of the zenith to 23. 5 North ofsun theiszenith. At the Tropic of Cancer, the noontime directly overhead on Allthe days have 12 summer daylight. 21. 5 above the northern solstice. In Insolation Canton, the noontime sun goes from 45 -23. 5 = ü depends onhours sunof angle. horizon to the 45 -23. 5 = At the Arctic Circle, sun goes 68. 5 fromabove beingthe justhorizon. at the horizon on the winter solstice to 47 above the horizon. No-shadow days! Point of sunrise & sunset changes by 68° From azimuth 56° to 124° (90° is east) Highest angle changes by 47° from 21. 5° to 68. 5°

Insolation in Canton ü Summer sun 21. 5 ~665 W falls on one m 2 in Canton in summer ~500 W falls on one m 2 in Canton in spring & fall ~700 W/m 2 from sun reaches surface ~260 W falls on one m 2 in Canton in winter

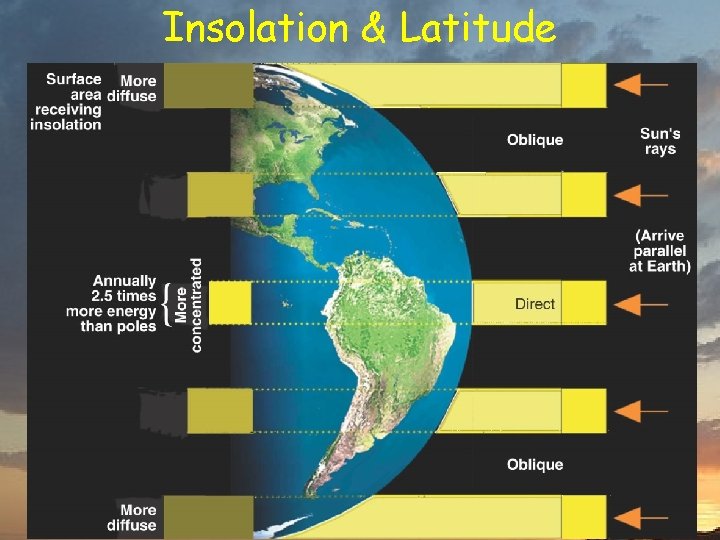
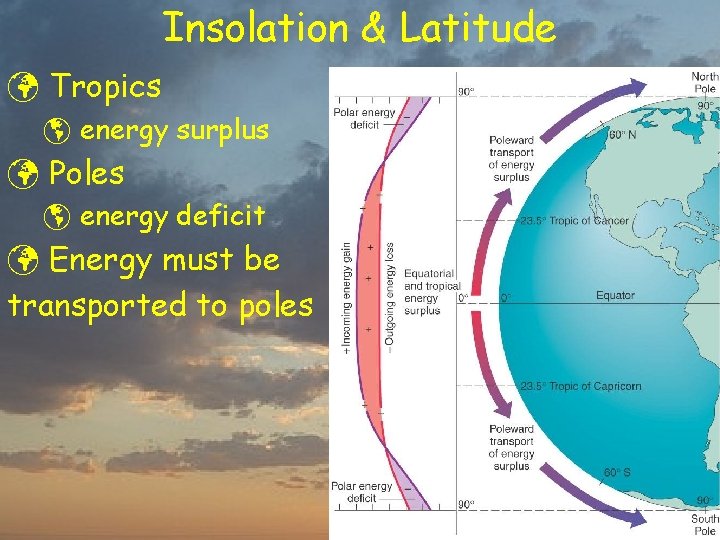
Insolation & Latitude

Insolation & Latitude ü Tropics þ energy surplus ü Poles þ energy deficit ü Energy must be transported to poles

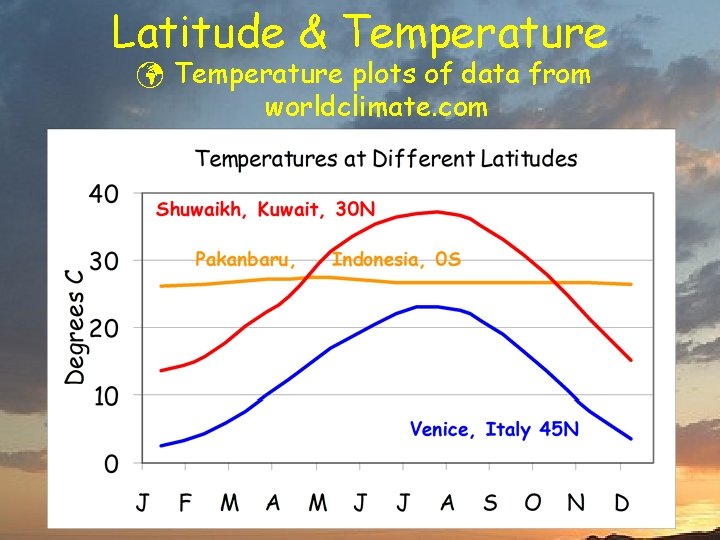
Latitude & Temperature ü Temperature plots of data from worldclimate. com

Reflected Radiation -- Albedo ü Different surfaces reflect differently Melting ice & snow increases absorption, enhances heating “Ice-Albedo Effect” Melting sea ice increases absorption, enhances heating

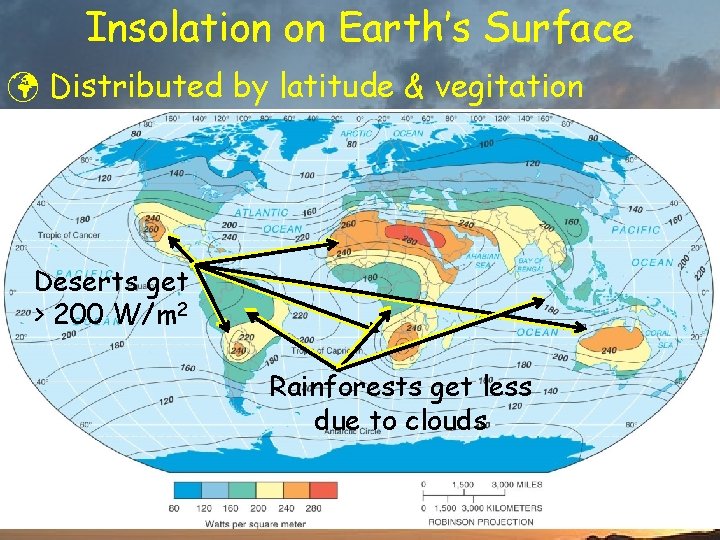
Insolation on Earth’s Surface ü Distributed by latitude & vegitation Deserts get > 200 W/m 2 Rainforests get less due to clouds

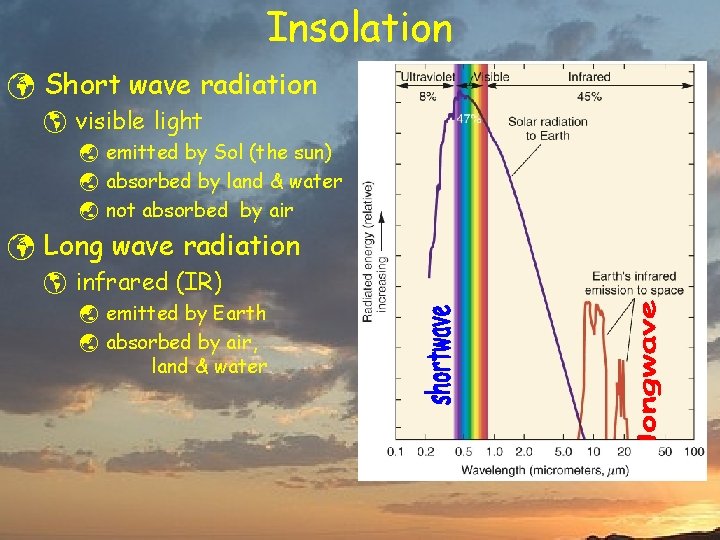
Insolation ü Short wave radiation þ visible light ý emitted by Sol (the sun) ý absorbed by land & water ý not absorbed by air ü Long wave radiation þ infrared (IR) ý emitted by Earth ý absorbed by air, land & water

Simplified Energy Budget Shortwave radiation from sun (light) absorbed by Earth (land & water) Earth heats & emits longwave radiation Top of the atmosphere receives 1372 W/m 2 - solar constant 10 W = power to lift 1 kg 1 meter in 1 second

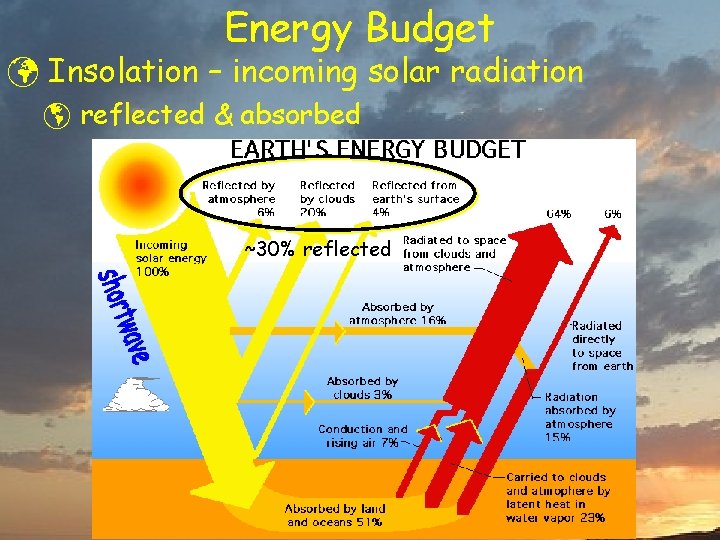
Energy Budget ü Insolation – incoming solar radiation þ reflected & absorbed ~30% reflected

Energy Budget ü Insolation – incoming solar radiation þ reflected & absorbed ~30% reflected Outgoing shortwave reflected from clouds and deserts, absorbed by ocean & forests

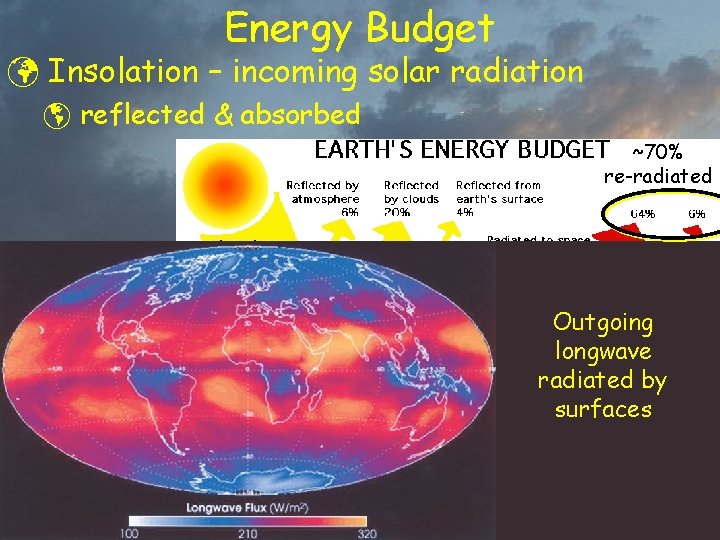
Energy Budget ü Insolation – incoming solar radiation þ reflected & absorbed ~70% re-radiated ~30% reflected ~19% absorbed by air & clouds ~51% absorbed by surface

Energy Budget ü Insolation – incoming solar radiation þ reflected & absorbed ~70% re-radiated Outgoing longwave radiated by surfaces ~51% absorbed by surface

Energy Budget ü Insolation þ Sun’s incident energy drives air motions (energy from deep interior adds a tiny bit) þ Distribution of Sunlight ý Reflection from clouds, landscape ý Absorption by atmosphere ý Absorption by surface þ Albedo = ratio of sunlight reflected ý Earth: 0. 367 ý Moon: 0. 113 ý Mars: 0. 15 ý Venus: 0. 84

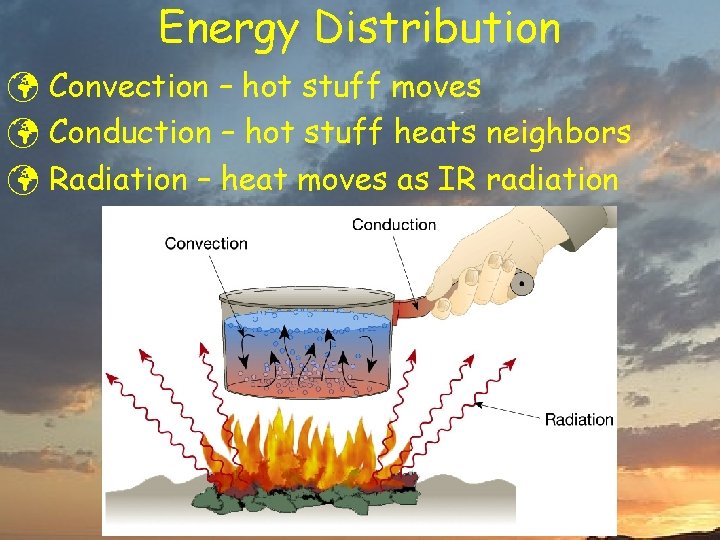
Energy Distribution ü Convection – hot stuff moves ü Conduction – hot stuff heats neighbors ü Radiation – heat moves as IR radiation

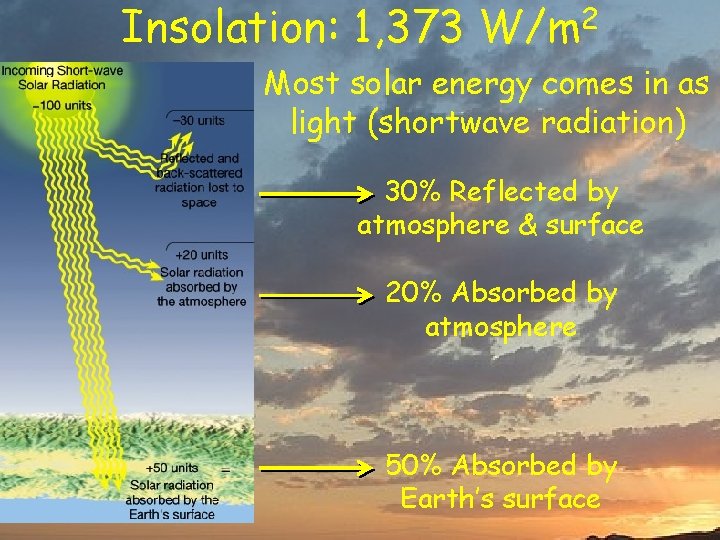
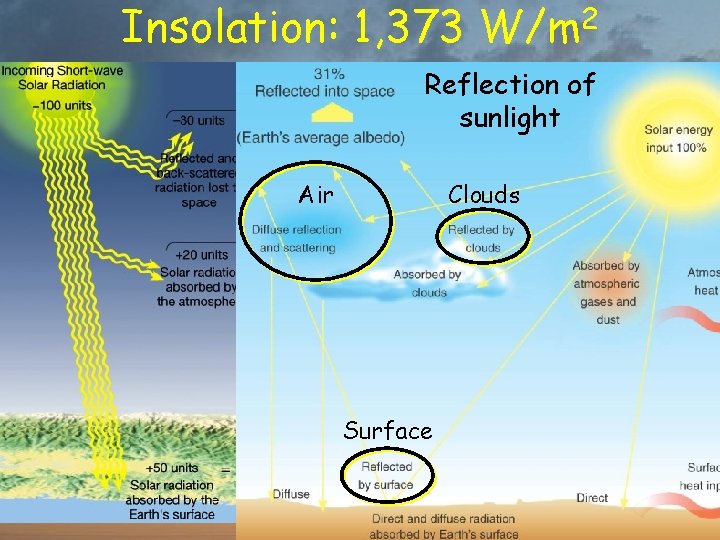
Insolation: 1, 373 W/m 2 Most solar energy comes in as light (shortwave radiation) 30% Reflected by atmosphere & surface 20% Absorbed by atmosphere 50% Absorbed by Earth’s surface

Insolation: 1, 373 W/m 2 of Most solar Reflection energy comes in as sunlight (shortwave radiation) Air 30% Clouds Reflected by atmosphere & surface 20% Absorbed by atmosphere Surface 50% Absorbed by Earth’s surface

Energy Emitted by Planet Earth % of total insolation 30% reflected directly to space 70% emitted as IR

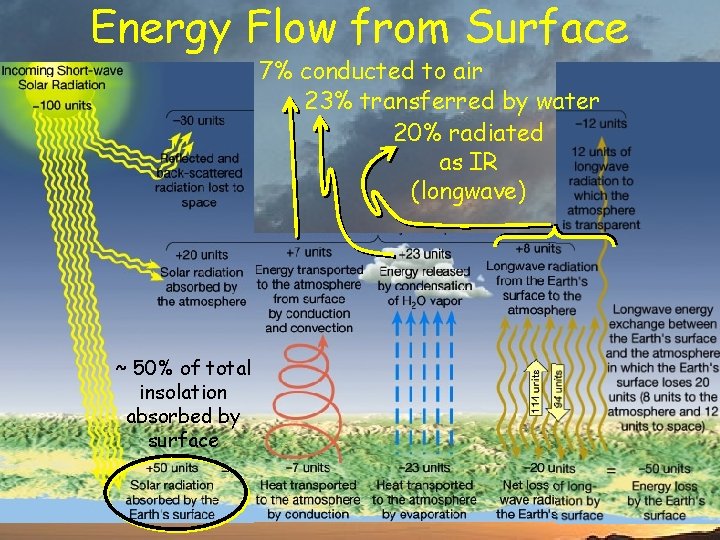
Energy Flow from Surface 7% conducted to air 23% transferred by water 20% radiated as IR (longwave) ~ 50% of total insolation absorbed by surface

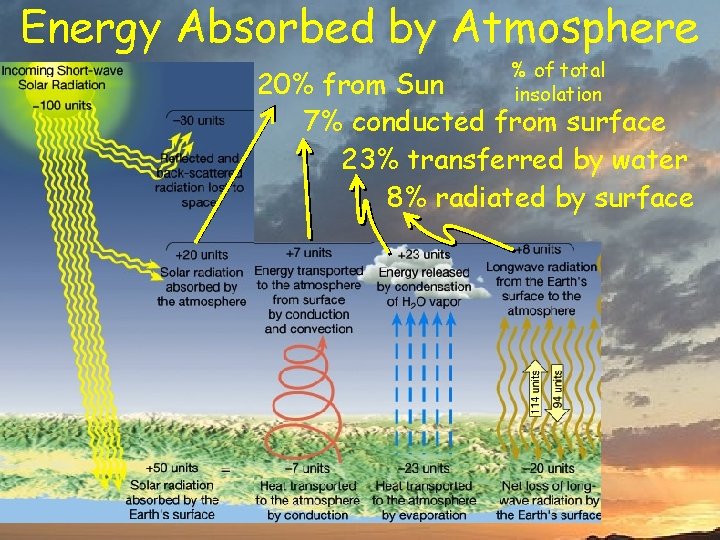
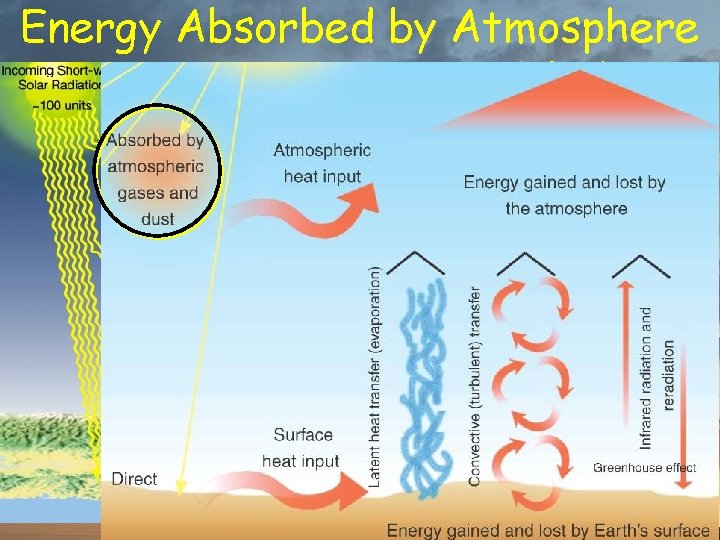
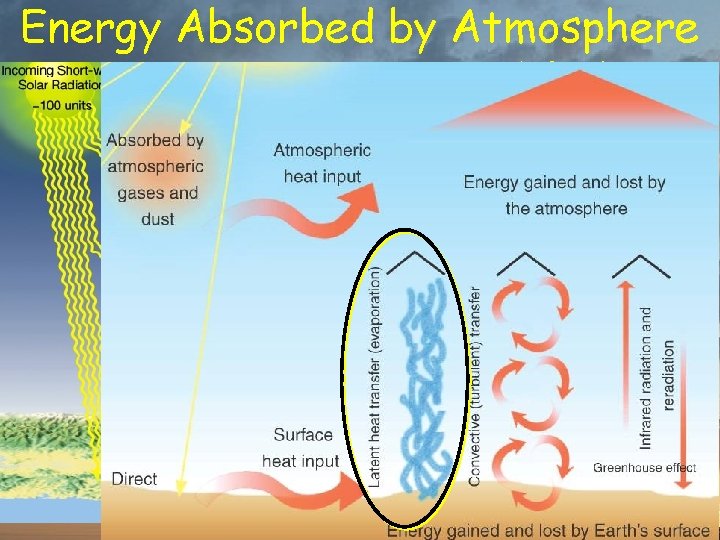
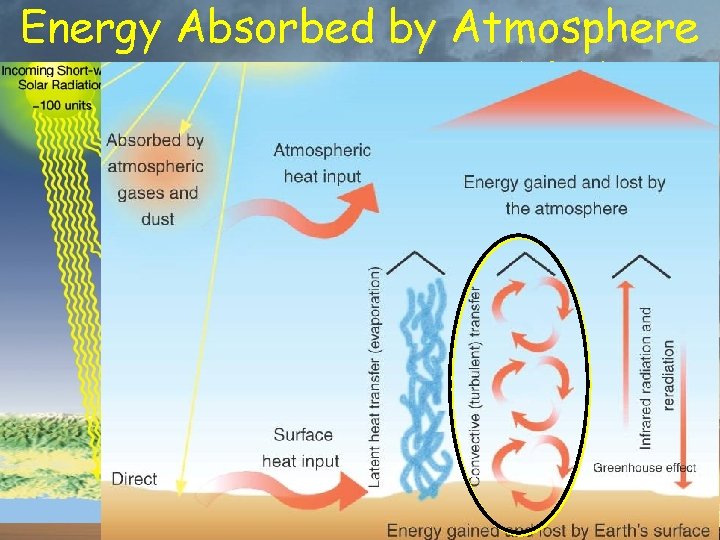
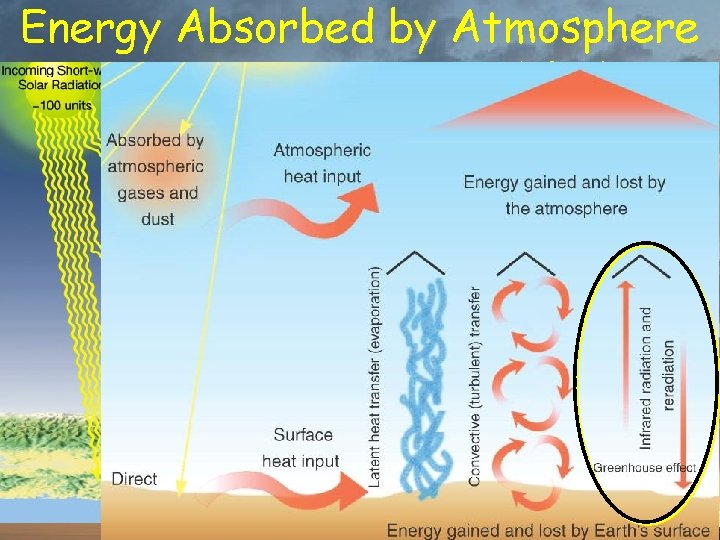
Energy Absorbed by Atmosphere % of total insolation 20% from Sun 7% conducted from surface 23% transferred by water 8% radiated by surface

Energy Absorbed by Atmosphere % of total insolation 20% from Sun 7% conducted from surface 23% transferred by water 8% radiated by surface

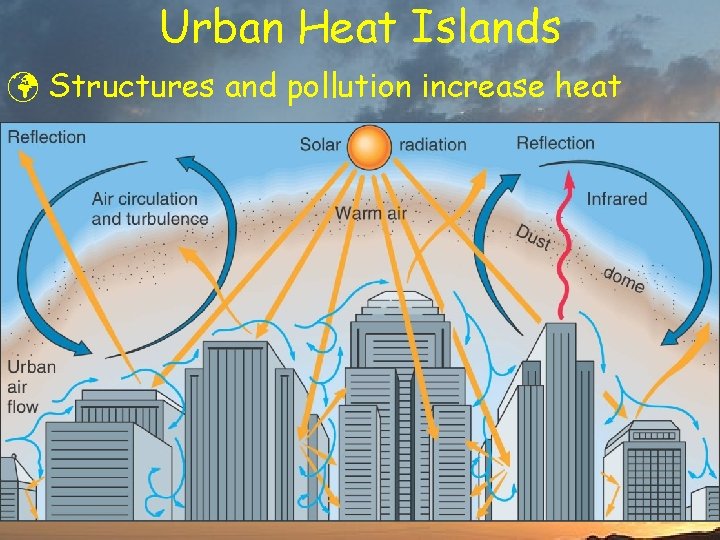
Urban Heat Islands ü Structures and pollution increase heat

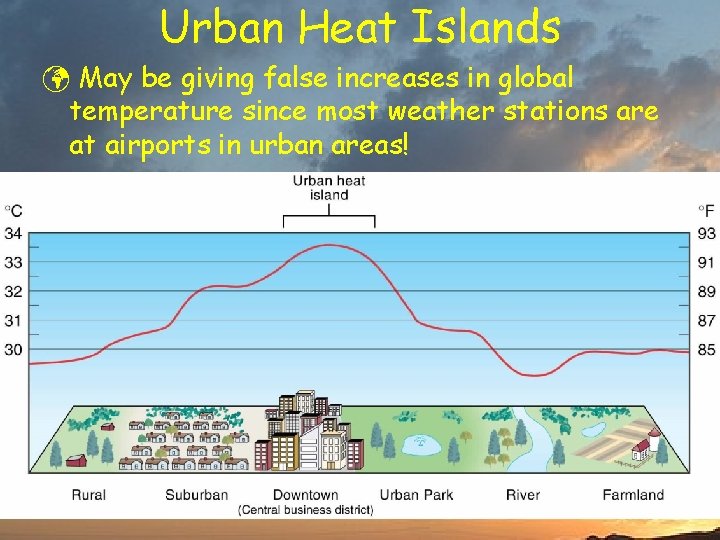
Urban Heat Islands ü May be giving false increases in global temperature since most weather stations are at airports in urban areas!

Energy Absorbed by Atmosphere % of total insolation 20% from Sun 7% conducted from surface 23% transferred by water 8% radiated by surface

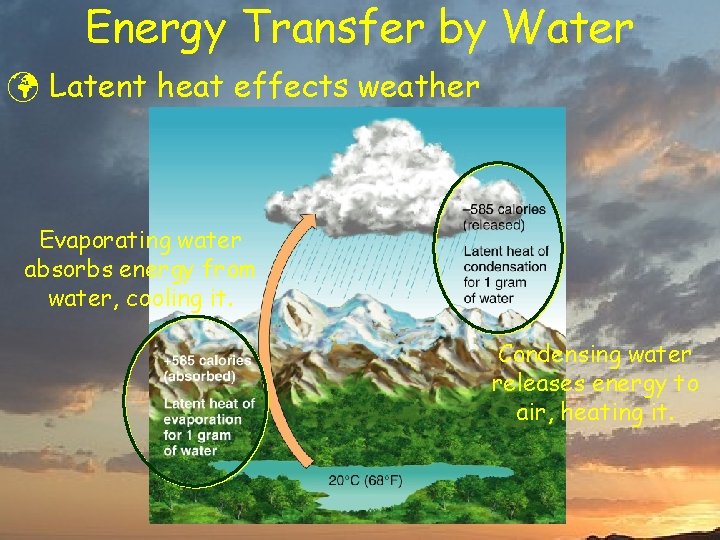
Energy Transfer by Water ü Latent heat effects weather Evaporating water absorbs energy from water, cooling it. Condensing water releases energy to air, heating it.

Energy Absorbed by Atmosphere % of total insolation 20% from Sun 7% conducted from surface 23% transferred by water 8% radiated by surface

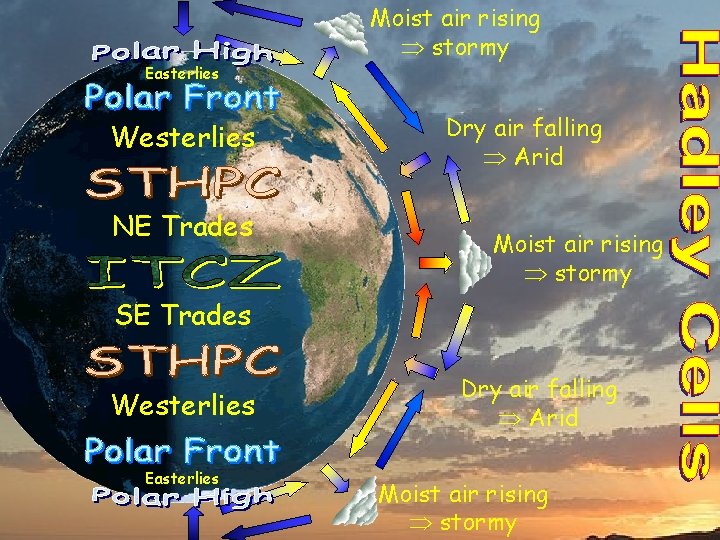
Easterlies Westerlies NE Trades Moist air rising stormy Dry air falling Arid Moist air rising stormy SE Trades Westerlies Easterlies Dry air falling Arid Moist air rising stormy

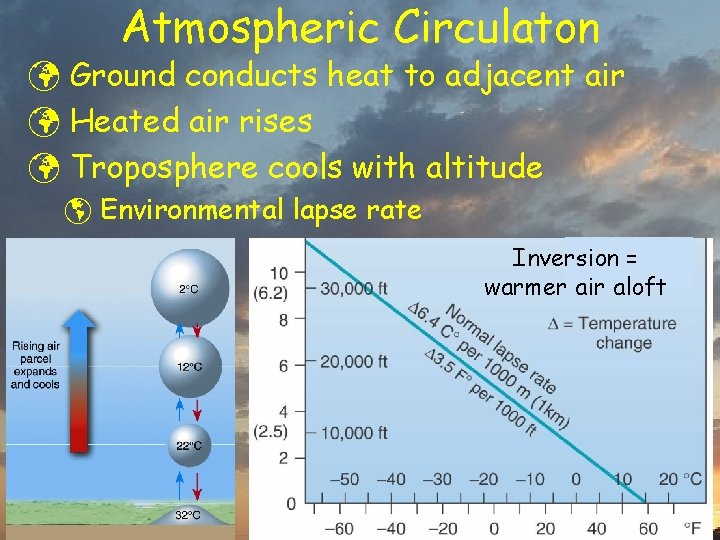
Atmospheric Circulaton ü Ground conducts heat to adjacent air ü Heated air rises ü Troposphere cools with altitude þ Environmental lapse rate Inversion = warmer air aloft

Energy Absorbed by Atmosphere % of total insolation 20% from Sun 7% conducted from surface 23% transferred by water 8% radiated by surface

Greenhouse Effect ü Light from sun gets absorbed by Earth ü Earth radiates infrared

Greenhouse Effect ü Light from sun gets absorbed by Earth ü Earth radiates infrared Earth re-emits energy absorbed from sunlight as infrared

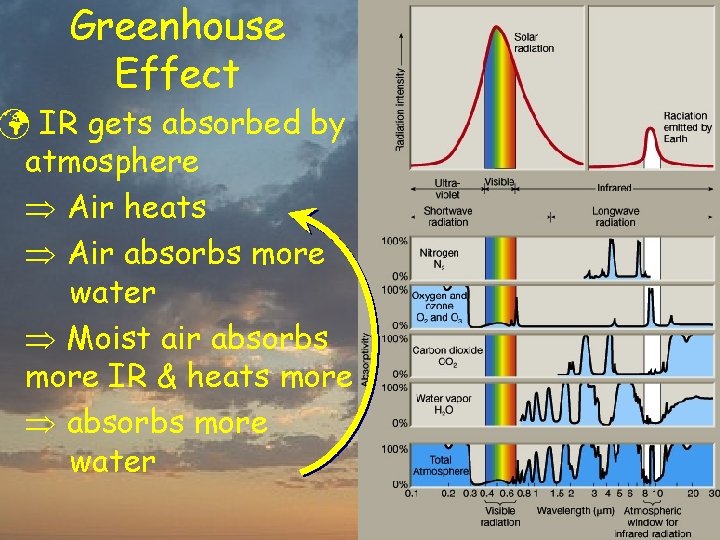
Greenhouse Effect ü IR gets absorbed by atmosphere Air heats Air absorbs more water Moist air absorbs more IR & heats more absorbs more water

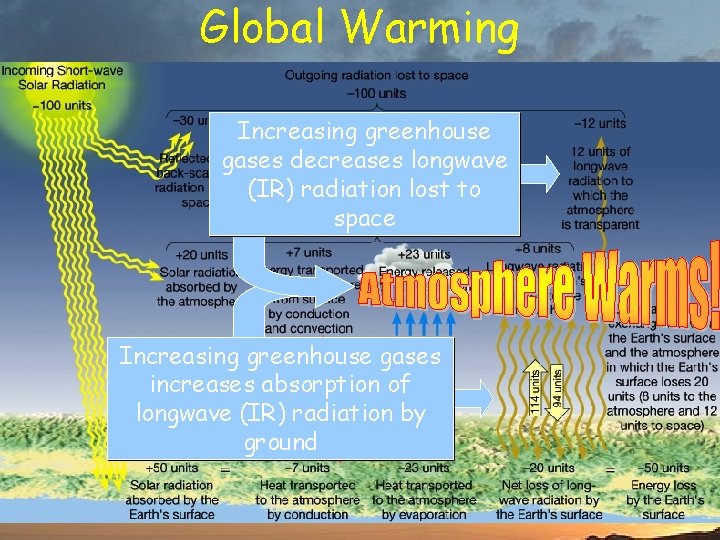
Global Warming Increasing greenhouse gases decreases longwave (IR) radiation lost to space Increasing greenhouse gases increases absorption of longwave (IR) radiation by ground

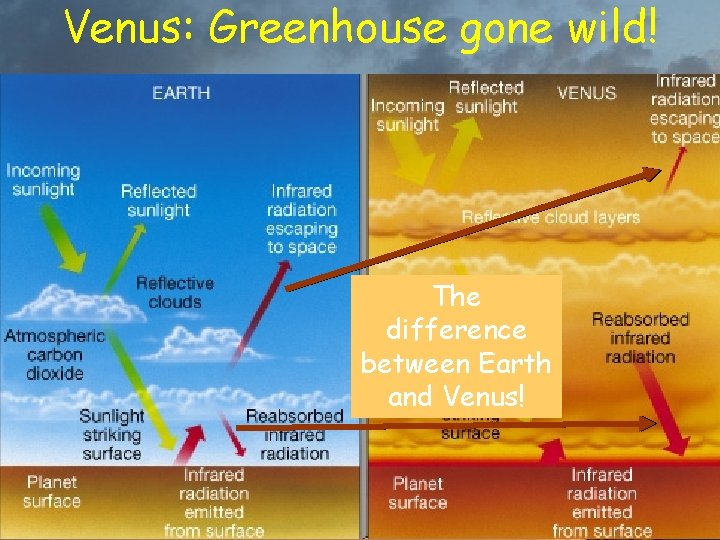
Venus: Greenhouse gone wild! The difference between Earth and Venus!

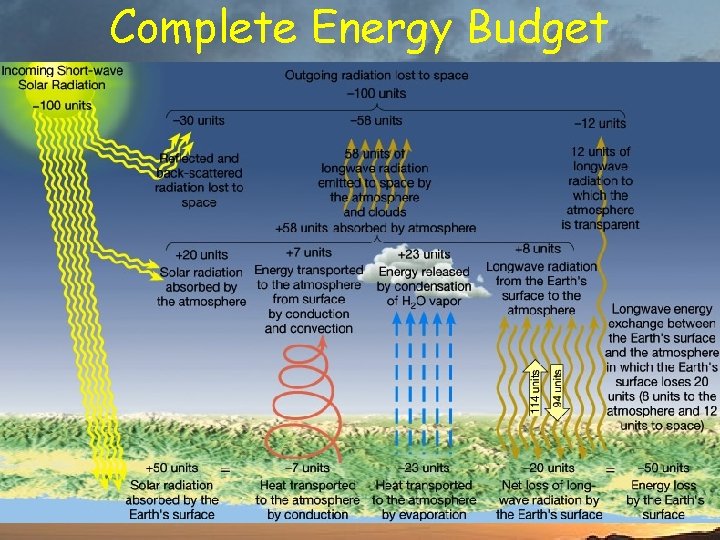
Complete Energy Budget

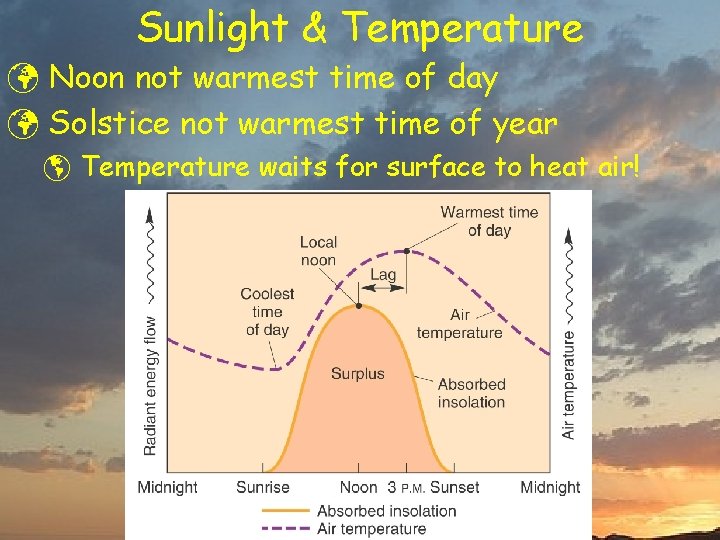
Sunlight & Temperature ü Noon not warmest time of day ü Solstice not warmest time of year þ Temperature waits for surface to heat air!

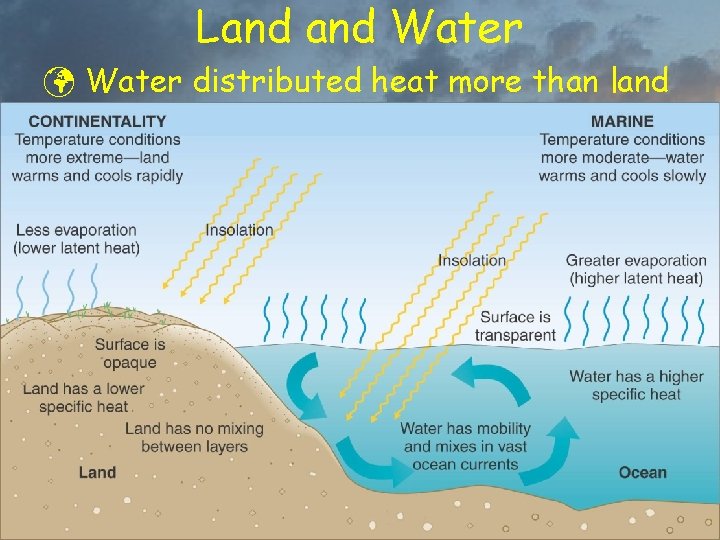
Insolation on Land Water ü Land þ Light heats surface, some downward conduction ý no downward transmission or convection þ Conduction to subsurface very slow ü Water þ Surface molecules evaporate, cooling surface þ Light penetrates to depths, heats subsurface þ Heats slowly due to high specific heat þ Convection moves heat across surface and beneath surface … currents

Land Water ü Water distributed heat more than land

Energy Absorbed by Water ü Specific Heat þ Energy absorbed/released to change temp. ü Latent Heat þ Energy needed to change phase (substance remains at same temperature)

Energy Absorbed by Water ü Specific Heat þ Energy absorbed or released to change temp. Raising 1 kg of water 1°C absorbs 4, 168 Joules 1 kg 4000 Joules 10 cm square cube of water Substance Specific Heat (Joule/K/kg) Air (50 C) 1050 Iron or Steel 460 Lead 130 Glass 840 Quartz 762 Granite 804 Sandstone 1088 Shale 712 Soil (average) 1050 Wood (average) 1680 Ice 2100 Steam 2050 Water 4168 ≈ energy to lift 400 kg or 900 lb 1 m

Energy Absorbed by Water ü Latent Heat Specific Heat (Joule/kg) þ Energy absorbed or Substance released to change phase vaporization Evaporating 1 kg of water absorbs 2, 257, 000 Joules 1 kg 2, 257, 000 Joules fusion Alcohol 879, 000 109, 000 Water 2, 257, 000 333, 500 10 cm square cube of water ≈ energy to lift 225, 700 kg or 507, 000 lb 1 m

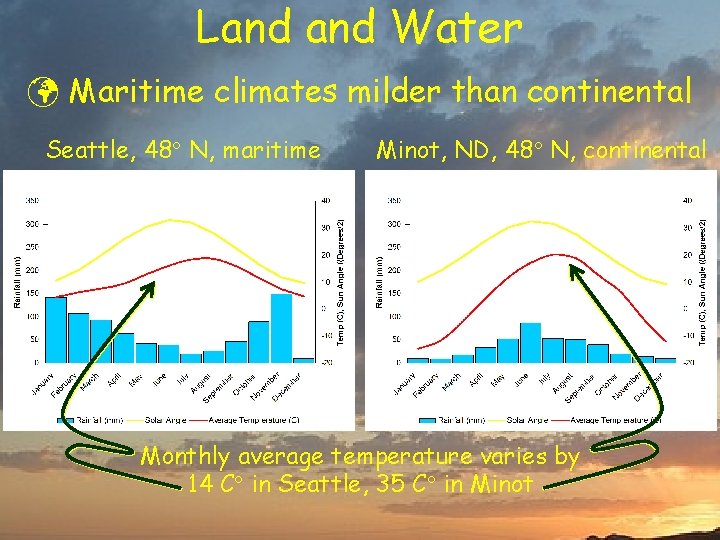
Land Water ü Maritime climates milder than continental Seattle, 48 N, maritime Minot, ND, 48 N, continental Monthly average temperature varies by 14 C in Seattle, 35 C in Minot

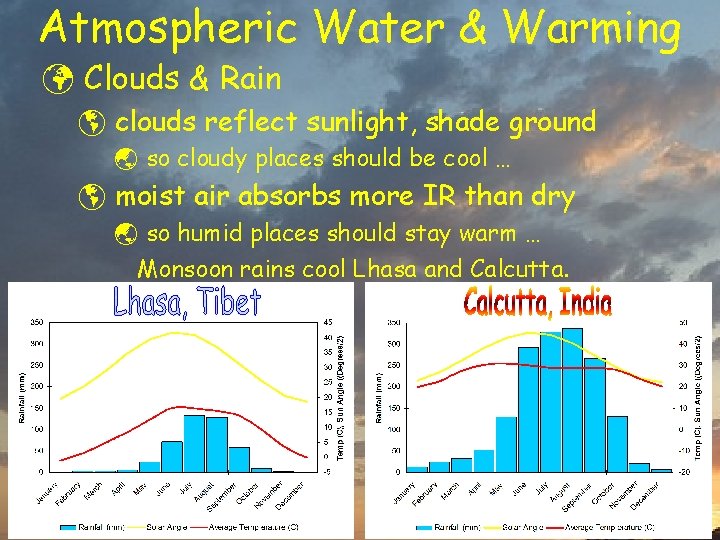
Atmospheric Water & Warming ü Clouds & Rain þ clouds reflect sunlight, shade ground ý so cloudy places should be cool … þ moist air absorbs more IR than dry ý so humid places should stay warm … Monsoon rains cool Lhasa and Calcutta.

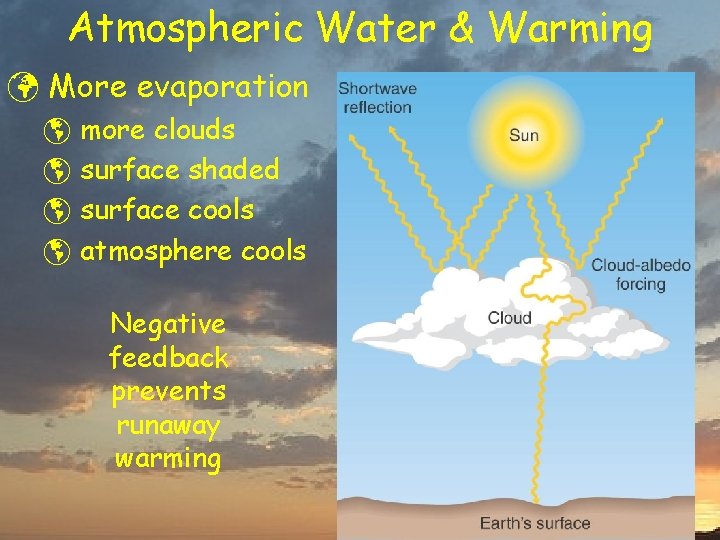
Atmospheric Water & Warming ü More evaporation þ more clouds þ surface shaded þ surface cools þ atmosphere cools Negative feedback prevents runaway warming

Atmospheric Water & Warming ü More evaporation þ more clouds þ more IR absorption þ atmosphere warms Positive feedback enhances runaway warming

Ocean Currents Surface currents move warm water toward poles Gulf Stream warms east coast of US and Europe

Ocean Currents Sea Surface Temperature monitored by satellites, augmented by buoy array. Hurricanes Equator 9/26/07 http: //www. ssec. wisc. edu/data/sst. html

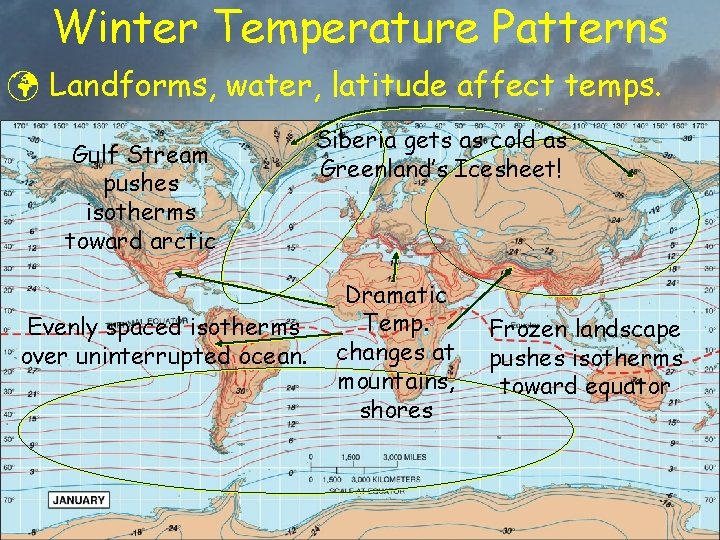
Winter Temperature Patterns ü Landforms, water, latitude affect temps. Gulf Stream pushes isotherms toward arctic Evenly spaced isotherms over uninterrupted ocean. Siberia gets as cold as Greenland’s Icesheet! Dramatic Temp. changes at mountains, shores Frozen landscape pushes isotherms toward equator

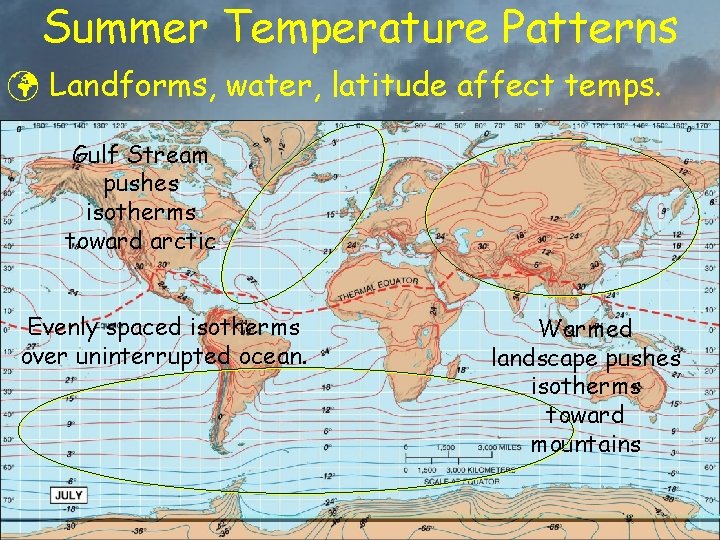
Summer Temperature Patterns ü Landforms, water, latitude affect temps. Gulf Stream pushes isotherms toward arctic Evenly spaced isotherms over uninterrupted ocean. Warmed landscape pushes isotherms toward mountains

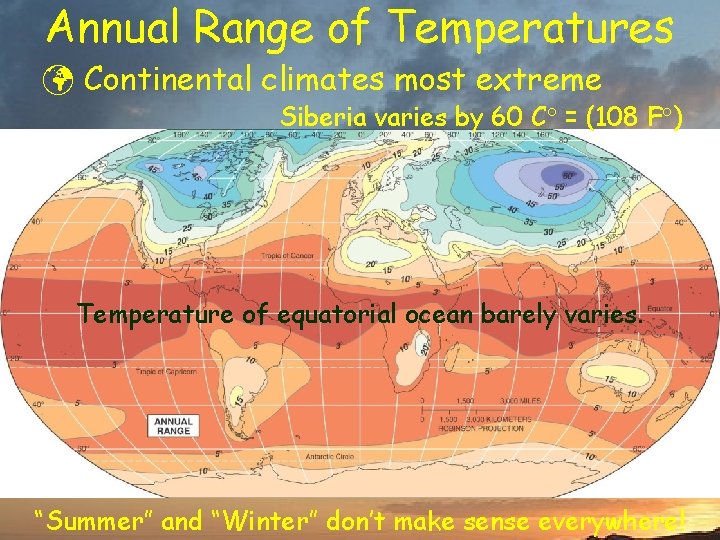
Annual Range of Temperatures ü Continental climates most extreme Siberia varies by 60 C = (108 F ) Temperature of equatorial ocean barely varies. “Summer” and “Winter” don’t make sense everywhere!