Soaps and Detergents B Sc III Chemistry Dr

Soaps and Detergents B. Sc. III Chemistry Dr. S. R. Mane M. Sc. B. Ed. Ph. D. Associate Prof. & Head, Dept. of Chemistry, Smt. K. R. P. Kanya Mahavidyalaya, Islampur

Semester-V Paper-XII (Industrial Chemistry) 4. Soaps and Detergents Lectures 7 4. 1 Introduction 4. 2 Soaps i. Raw materials, ii. Types of soaps iii. Cleaning action of soap 4. 3 Manufacture of soap i. Cold Process ii. Semi-boiled Process iii. Boiled or Hot Process 4. 3 Detergents i. Raw Materials ii. Types of Detergents: Anionic, cationic and amphoteric 4. 4 Comparisons between soaps and detergents.

INTRODUCTION Soaps And Detergents are used for washing. It dissolves dirt, but some dirt will not dissolve in water. Soaps and detergents help water to remove dirt.



Saponification Process of making soap from animal fat or vegetable oil using a base. O CH 2 – O – C – (CH 2)14 CH 3 CH 2 – OH O O CH – O – C – (CH 2)14 CH 3 + 3 Na. OH CH – OH + 3 Na+ -OC – (CH 2)14 CH 3 O CH 2 – O – C – (CH 2)14 CH 3 Glyceryl Tripalmitate (Tripalmitin) CH 2 – OH Sodium hydroxide Glycerol 3 Sodium Palmitate (Soap)
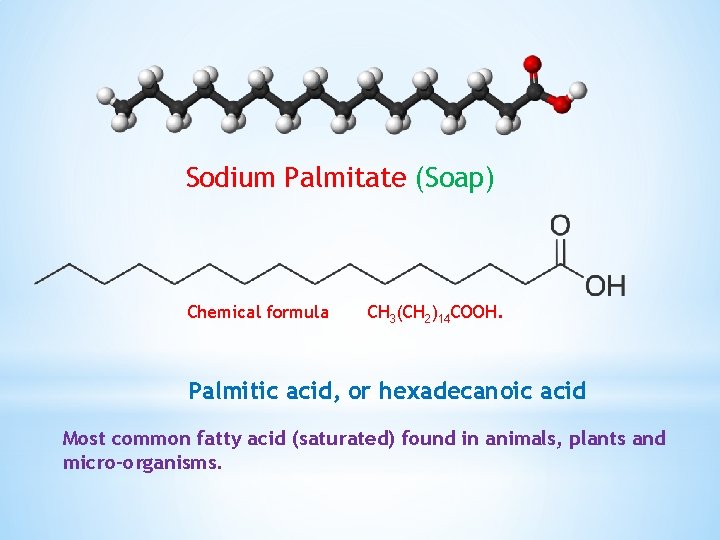
Sodium Palmitate (Soap) Chemical formula CH 3(CH 2)14 COOH. Palmitic acid, or hexadecanoic acid Most common fatty acid (saturated) found in animals, plants and micro-organisms.
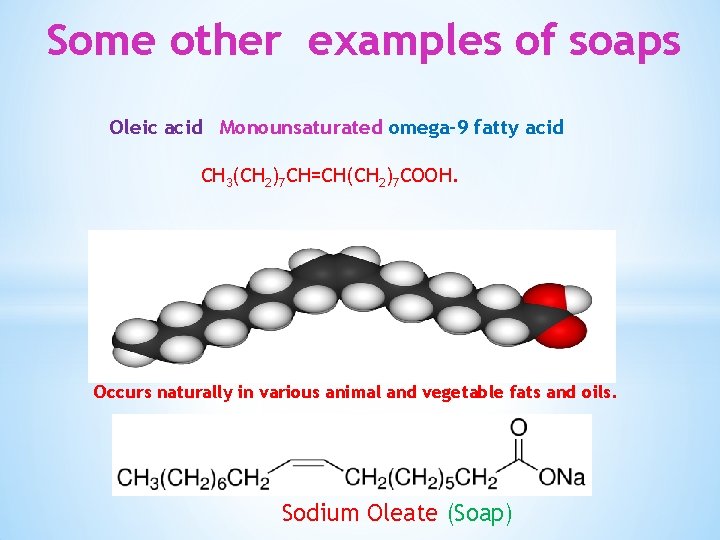
Some other examples of soaps Oleic acid Monounsaturated omega-9 fatty acid CH 3(CH 2)7 CH=CH(CH 2)7 COOH. Occurs naturally in various animal and vegetable fats and oils. Sodium Oleate (Soap)
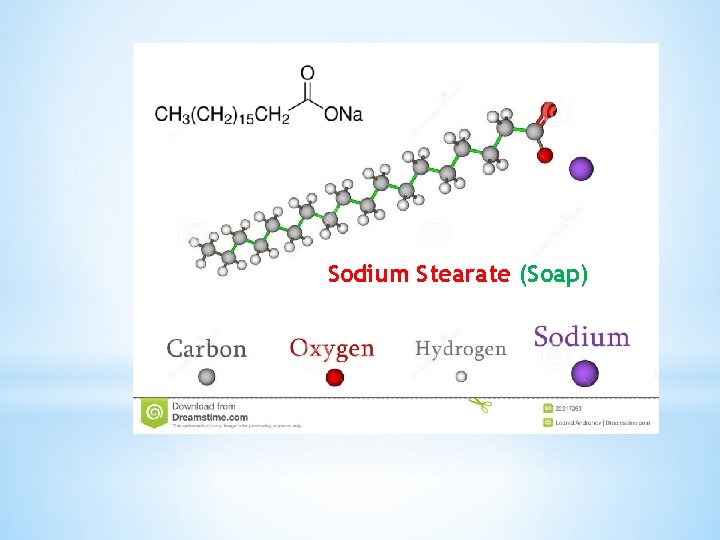
Sodium Stearate (Soap)
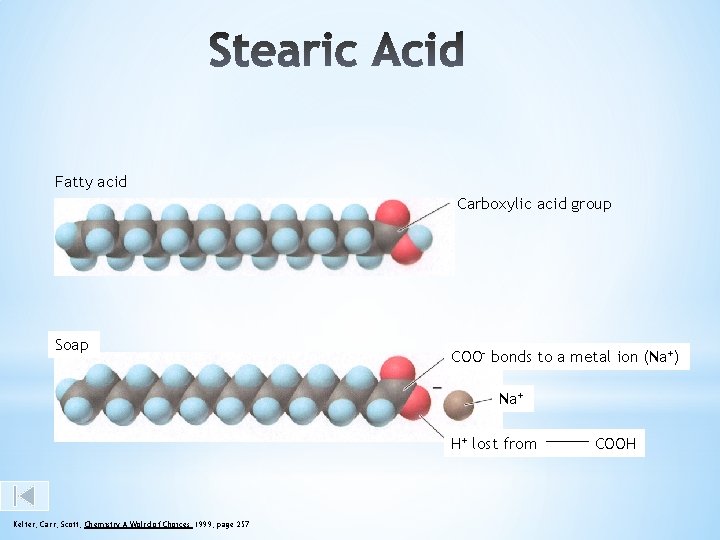
Fatty acid Carboxylic acid group Soap COO- bonds to a metal ion (Na+) Na+ H+ lost from Kelter, Carr, Scott, Chemistry A Wolrd of Choices 1999, page 257 COOH
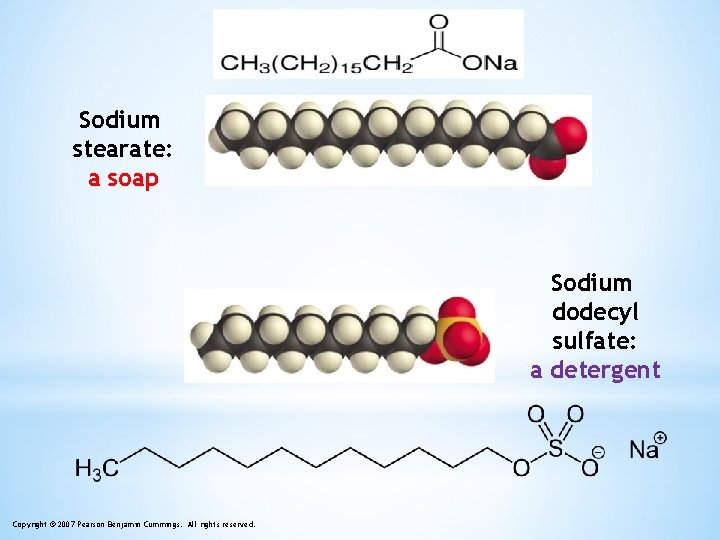
Sodium stearate: a soap Sodium dodecyl sulfate: a detergent Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.

Raw Materials for Soap Tallow- Obtained from cattle, sheep, goat etc. animals. Olein 40% Stearin 60%. Soap prepared from tallow alone is hard and water insoluble. In order to make it soft and water soluble it is mixed with coconut oil, palm oil, cotton seed oil. O O CH 2 – O – C – (CH 2)7 CH=CH-(CH 2)7 -CH 3 CH 2 – O – C – (CH 2)16 CH 3 O CH – O – C – (CH 2)7 CH=CH-(CH 2)7 -CH 3 O CH 2 – O – C – (CH 2)7 CH=CH-(CH 2)7 -CH 3 Olein Unsaturated O CH – O – C – (CH 2)16 CH 3 O CH 2 – O – C – (CH 2)16 CH 3 Stearin Saturated

Grease or Lard- Obtained from mail pig and smaller domestic animals. Olein 60% Stearin 40%. It is soft like butter. Used for making best quality soap Coconut oil- Rich in Lauric and Myristic acid CH 3(CH 2)10 -COOH. Lauric acid CH 3(CH 2)12 -COOH. Myristic acid Gives hard white soap lathers even in salty water. So used as a marine soap Palm oil- Toilet soaps Castor oil- Palmitin CH 3(CH 2)14 -COOH Olein CH 3(CH 2)7 CH=CH(CH 2)7 COOH. Trasperent Soaps Ricinolic acid (Hydroxy unsat. Acid) CH 3(CH 2)6 CH=CH(CH 2)7 -CH-COOH. OH

Linseed oil- soft soaps Linoleic acid C 17 H 31 -COOH CH 3(CH 2)4 CH=CH-CH 2 -CH=CH-(CH 2)7 -COOH. Di unsaturated Cotton seed oil- olein , stearin and palmitin Soap lathers freely Ground nut oil- Large proportion of unsat. Oleic acid so form very soft soap. Therefore it is saturated. Maize oil- Linoleic and oleic acid Fatty Acids. Alkaline Materials
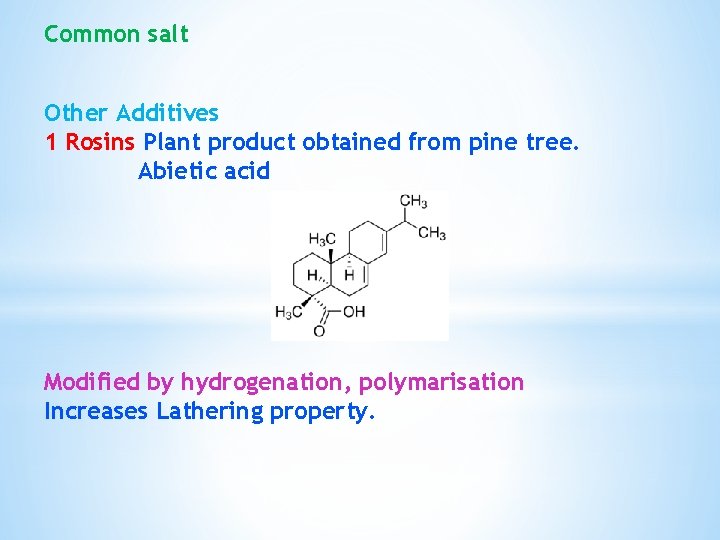
Common salt Other Additives 1 Rosins Plant product obtained from pine tree. Abietic acid Modified by hydrogenation, polymarisation Increases Lathering property.
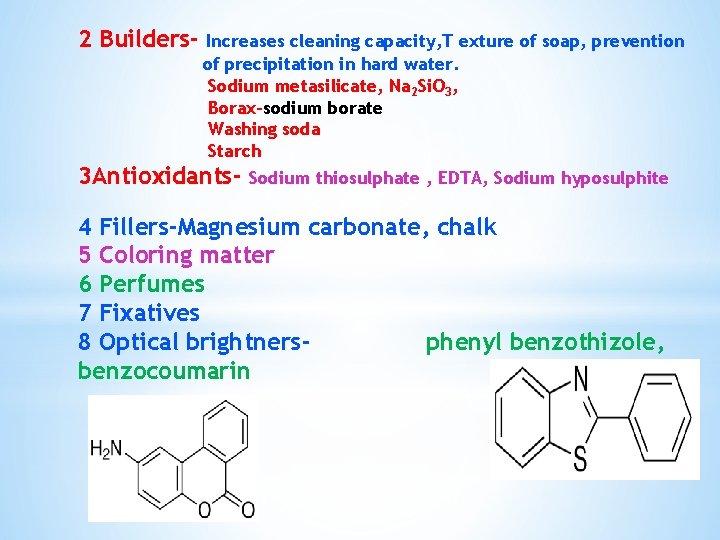
2 Builders- Increases cleaning capacity, T exture of soap, prevention of precipitation in hard water. Sodium metasilicate, Na 2 Si. O 3, Borax-sodium borate Washing soda Starch 3 Antioxidants- Sodium thiosulphate , EDTA, Sodium hyposulphite 4 Fillers-Magnesium carbonate, chalk 5 Coloring matter 6 Perfumes 7 Fixatives 8 Optical brightnersphenyl benzothizole, benzocoumarin

Types of soaps 1. Washing Soap 2. Tiolet Soap 3. Shaving Soap 4. Industrial Soap

Ads’ and soaps Among the consumer products aggressively advertised, bath soap’s are in the top of the list. Although, a bath soaps basic function is to clean the body, the advertisements confer exotic qualities to it. Soaps are being advertised as - Beauty soaps - Cinema stars are their brand ambassadors - Health soaps - Sports person used to sponsor them - Complexion soaps

- Deodorant soaps - Freshness soaps - Baby soaps - Herbal or medicinal soaps

None of the advertisements speak of what exactly, goes into the making of such soaps. Can medicated soaps readily cure skin diseases? Are glycerine transparent soaps really gentle to the skin ? As mentioned earlier soaps are salts particularly sodium or potassium of long chain fatty acids. These carboxylic acids are derived from fatty oils. The principal active cleaning agent is sodium carboxylate.
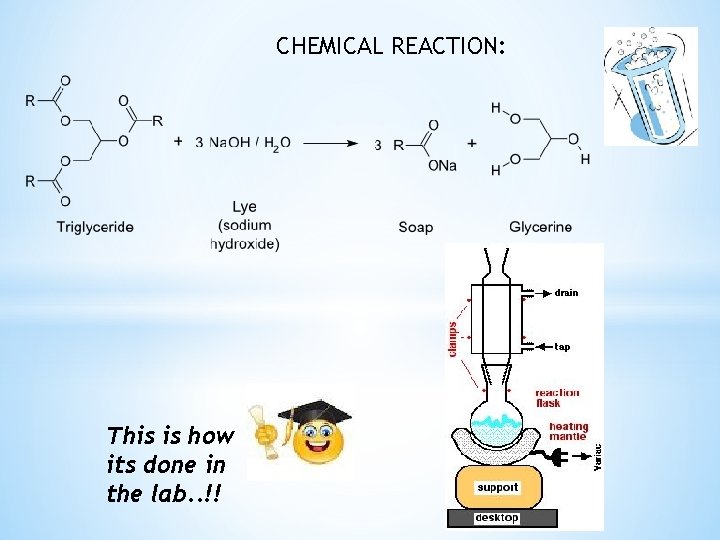
CHEMICAL REACTION: This is how its done in the lab. . !!

How does soap clean. . Soap dissolves in water. A soap molecule has a long hydrocarbon End a short ionic end containing COONa+. The non-ionic hydrocarbon end has the property to repel water. (hydrophobic tail). The ionic end has the property to attract water. This end attaches itself to water. (hydrophilic head). The hydrocarbon end attaches to the dirt or grease to form structures called ‘micelles’

See how the dirt is removed. . !!
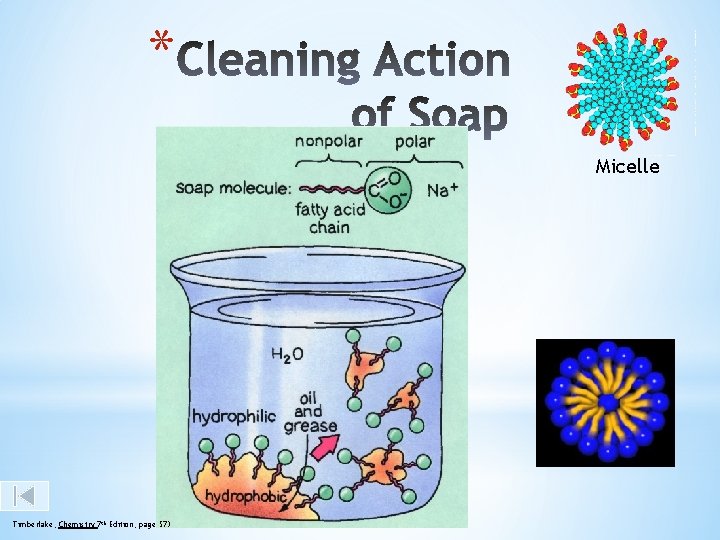
* Micelle Timberlake, Chemistry 7 th Edition, page 573

PRINCIPLE GROUPS OF SYNTHETIC DETERGENTS (SURFACTANTS) Four main groups of surfactants: anionic, cationic, non-cationic and amphoteric. 1. Anionic Surfactants are compounds in which the detergency is realized in the anion. R-SO 3 - Na+ Alkylsulfonates (anionic surfactants) 2. Cationic Surfactants are compounds in which the detergency is in the cation. R 2 N+(CH 3)2 Cl- Dialkyl dimethylammonium chlorides (cationic surfactants) 3. Non-ionic Surfactants contain non-ionic constituents. RO-(CH 2 -O)n. H Alkyl poly(ethylene glycol)ethers (non-ionic surfactants) 4. Amphoteric Surfactants includes both acidic and basic groups in the same molecule.
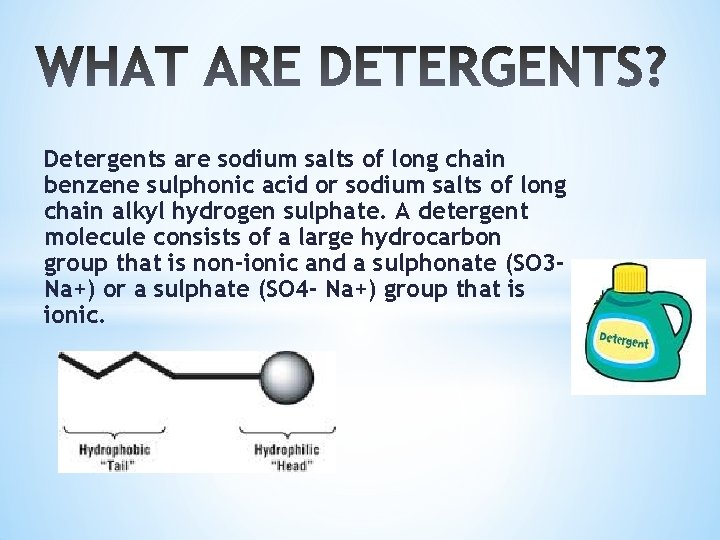
Detergents are sodium salts of long chain benzene sulphonic acid or sodium salts of long chain alkyl hydrogen sulphate. A detergent molecule consists of a large hydrocarbon group that is non-ionic and a sulphonate (SO 3 Na+) or a sulphate (SO 4 - Na+) group that is ionic.

DETERGENT, The king and DIRT, The Slave. . !!

SOAPS V/S DETERGENTS


DISADVANTAGES Detergents are not biodegradable. They pollute water and soil.

Thanks
- Slides: 31