Soap and Detergents Main groups in detergents 1














- Slides: 22

Soap and Detergents

Main groups in detergents 1. Surfactants 2. Builders 3. Bleaching agents 4. Additives

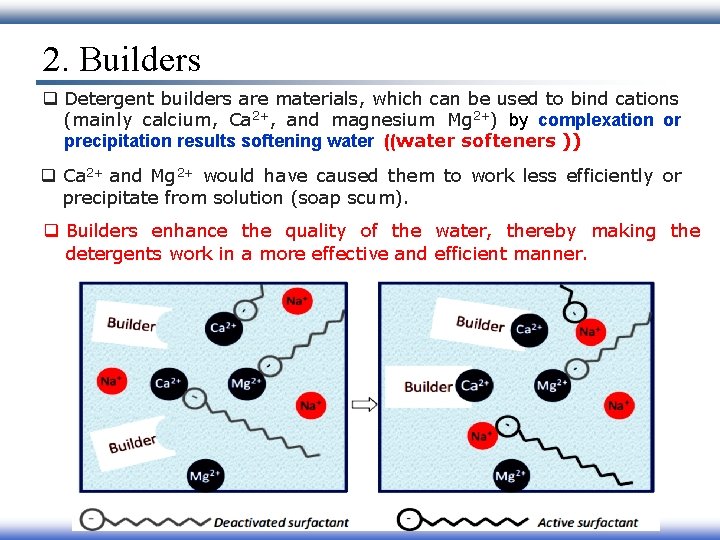
2. Builders q Detergent builders are materials, which can be used to bind cations (mainly calcium, Ca 2+, and magnesium Mg 2+) by complexation or precipitation results softening water ((water softeners )) q Ca 2+ and Mg 2+ would have caused them to work less efficiently or precipitate from solution (soap scum). q Builders enhance the quality of the water, thereby making the detergents work in a more effective and efficient manner.

Detergent builders types: Ø Sequestrating: These are water-soluble builders that form soluble complexes with Ca 2+ or Mg 2+. Ex. sodium tripolyphosphate (STPP) tetrasodium pyrophosphate, citrates, tartrates, succinates, gluconates, polycarboxylates, ethylenediamine and triethanolamine. Ø Precipitating: These builders are water-soluble until they form a complex with Ca 2+ or Mg 2+ and precipitate (fall out) of solution. The main example is sodium carbonate. Ø Ion exchange: These builders are insoluble in water and form insoluble complexes with Ca 2+ or Mg 2+. Examples are zeolites and sodium disilicate.

• Builders are basically water softeners Other benefits may be available: Ø Builders also provide a desirable level of alkalinity (increase p. H) that improves cleaning performance. Ø Help prevent the redeposition of soils after they have been removed from the fabric by surfactants Ø Carry absorbed liquid components in the products, like surfactants so as to produce a dry and free flowing powder Ø Typically improve the wetting and emulsification characteristics of detergent

Detergent builders can be categorized into two types: Organic detergent builders Inorganic detergent builders The inorganic constituents fall into the five following groups: 1. Phosphates 2. Silicates 3. Carbonates 4. Zeolites 5. Bleach-active compounds

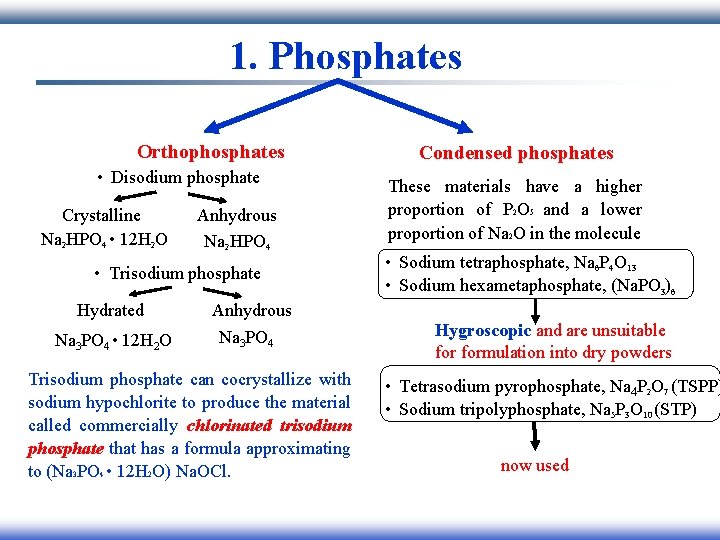
1. Phosphates Orthophosphates • Disodium phosphate Crystalline Na 2 HPO 4 • 12 H 2 O Anhydrous Na 2 HPO 4 • Trisodium phosphate Hydrated Na 3 PO 4 • 12 H 2 O Anhydrous Na 3 PO 4 Trisodium phosphate can cocrystallize with sodium hypochlorite to produce the material called commercially chlorinated trisodium phosphate that has a formula approximating to (Na 3 PO 4 • 12 H 2 O) Na. OCl. Condensed phosphates These materials have a higher proportion of P 2 O 5 and a lower proportion of Na 2 O in the molecule • Sodium tetraphosphate, Na 6 P 4 O 13 • Sodium hexametaphosphate, (Na. PO 3)6 Hygroscopic and are unsuitable formulation into dry powders • Tetrasodium pyrophosphate, Na 4 P 2 O 7 (TSPP) • Sodium tripolyphosphate, Na 5 P 3 O 10 (STP) now used

2. Silicates Sodium and potassium silicates important characteristics such as: Ø Alkalinity Ø Deflocculating, Ø Antiredeposition ability Ø Corrosion inhibitors of stainless steel and aluminumthat can be caused by synthetic detergents and complex phosphates • Sodium silicate is prepared by the fusion of sand that contains a high amount of silica with soda ash Na 2 CO 3 + Si. O 2 Na 2 Si. O 3 + CO 2 • (Na 2 O/Si. O 2 = 1/1) is used in dry blending • Liquid silicates Na 2 O/Si. O 2 ratio of 1: 2 or higher are used in laundry and automatic dishwashing applications

3. Carbonates • Carbonates are being used because of the restriction in the use of phosphates and silicate. • Na 2 CO 3 and zeolite has replaced sodium tripolyphosphate (STP) as a builder in granular laundry products High alkalinity Sodium carbonate provides Softens water by the precipitation of calcium and magnesium carbonates Can absorb large amounts of liquid material onto its surface and remains dry Sodium carbonate is produced by the Solvay process (burning limestone):

4. Zeolites Aluminosilicate • Zeolites occur naturally but are also produced industrially on a large scale. • Zeolites have a porous structure that can accommodate a wide variety of cations, such as Ca 2+, Mg 2+ and others. • The main advantage of zeolites compared to phosphates is nonsolubility in water, and they can remove heavy metal ions such as manganese and iron readily from the solution. • Magnesium ions are not totally removed by zeolites because of the size of magnesium; therefore, zeolites are then used in association with other builders such as sodium carbonate.

5. Bleach-active compounds • Oxygen-releasing compounds are added to detergent powders as bleach-active materials. • Hydrogen peroxide (H 2 O 2) is converted by alkaline medium into hydrogen peroxide anion as active intermediate • Active oxygen bleaches remove stains from fabric to improve fabric whiteness and brightness. • Suitable for use on coloured fabrics without damaging their colour.

Sodium perborate At elevated temperatures, active oxygen is released and has a bleaching effect Hypochlorite HOCl bleaching compound at normal temperature Hypochlorite reacts with an alkaline to produce hypochlorite anions (Na. OCl) is used as a source of active chlorine

q Advantages of sodium perborate over liquid sodium hypochlorite: • Na. OCl must be added separately whereas perborate can be included directly in the powder laundry product. • High dosage of Na. OCl may cause significant damage to laundry and clothes. • Na. OCl solutions have limited storage stability especially in the presence of impurities such as heavy metal ions. v However, hypochlorite bleach in either the wash or the rinse cycle is still a preferred bleaching agent in a large part of the world

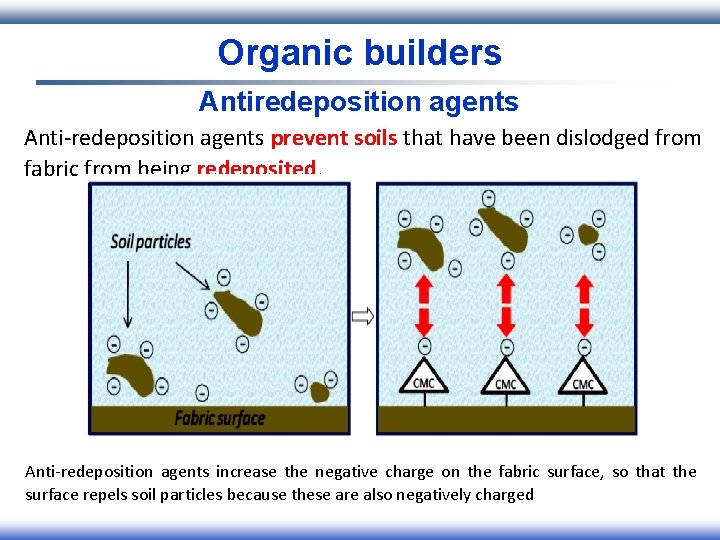
Organic builders Antiredeposition agents Anti-redeposition agents prevent soils that have been dislodged from fabric from being redeposited. Anti-redeposition agents increase the negative charge on the fabric surface, so that the surface repels soil particles because these are also negatively charged

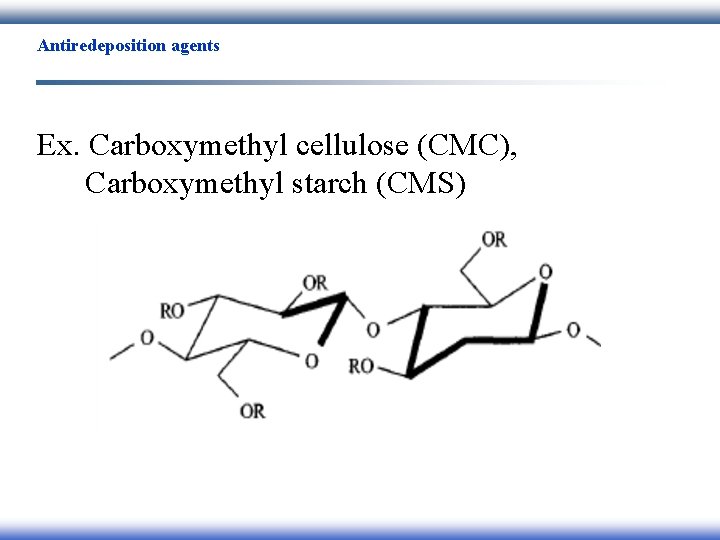
Antiredeposition agents Ex. Carboxymethyl cellulose (CMC), Carboxymethyl starch (CMS)

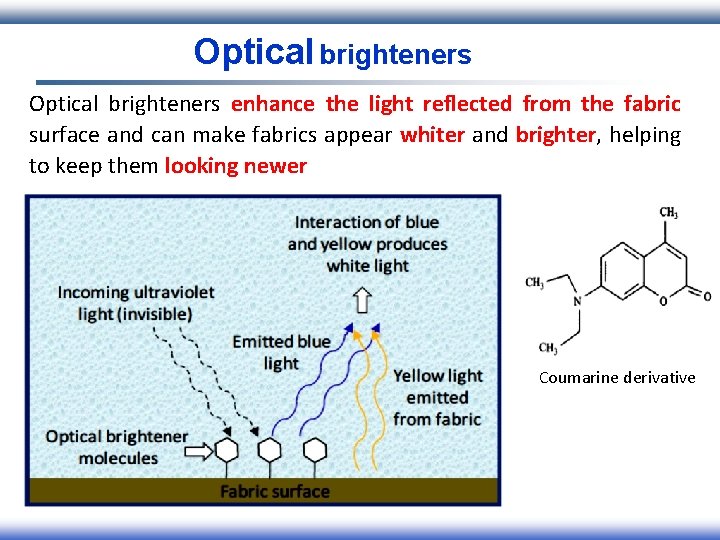
Optical brighteners enhance the light reflected from the fabric surface and can make fabrics appear whiter and brighter, helping to keep them looking newer Coumarine derivative

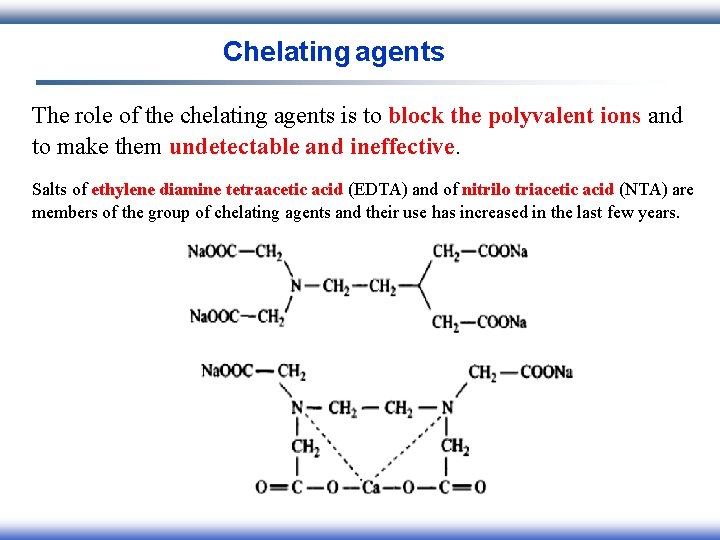
Chelating agents The role of the chelating agents is to block the polyvalent ions and to make them undetectable and ineffective. Salts of ethylene diamine tetraacetic acid (EDTA) and of nitrilo triacetic acid (NTA) are members of the group of chelating agents and their use has increased in the last few years.

ENZYMES Enzymes can perform two main roles in a laundry detergent: • Effect stain removal • Provide colour and fabric care Enzymes break down large molecules such as proteins, carbohydrates and fats into smaller segments. These smaller segments are either water-soluble, or are of size and polarity compatible with surfactants meaning that they can be suspended in solution. There are four types of enzymes of interest to the detergent industry: 1. Proteases: act on protein to form amino acids 2. Amyloses: convert starches into dextrins 3. Lipases: attack fats and oils 4. Celluloses: hydrolyze cellulose of broken surface fibers and remove micro-pills from cotton and restore color

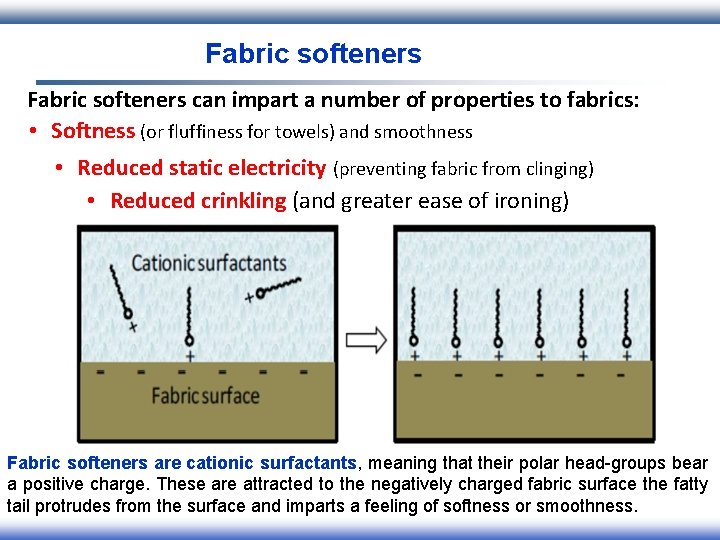
Fabric softeners can impart a number of properties to fabrics: • Softness (or fluffiness for towels) and smoothness • Reduced static electricity (preventing fabric from clinging) • Reduced crinkling (and greater ease of ironing) Fabric softeners are cationic surfactants, meaning that their polar head-groups bear a positive charge. These are attracted to the negatively charged fabric surface the fatty tail protrudes from the surface and imparts a feeling of softness or smoothness.

PRESERVATIVES Preservatives can prevent detergent spoilage during storage. Generally, preservatives are only required in laundry liquids. FOAM REGULATORS Foam regulators either prevent the formation of foam by disrupting the surfactants at the air-water interface of the forming bubble, or cause foam bubbles to collapse by forming. hydrophobic bridges across multiple bubbles CORROSION INHIBITORS Corrosion inhibitors inhibit corrosion of metallic washing machine interior parts. Sodium silicate is a common corrosion inhibitor.

• Kent, J. , Riegel's Handbook of Industrial Chemistry, Van Notrand Reinhold, New York, N. Y. , 1992. • Austin, G. , Shreve's Chemical Process Industries, 5 th ed. , Mc. Graw-Hill Book Company, New York, N. Y. , 1985. • Davidsohn, A. S. and B. Milwidsky, Synthetic Detergents, Longman Scientific and Technical, Burnt Mill, Harlow-England, 1987. § Mohammad F. A. , Bassam M. A. and James G. S. , “Handbook of Industrial Chemistry”, Mc. Graw-Hill, 2005. • D. J. Mc. Clements, “Food Emulsions: Principles, Practices, and Techniques”, CRC Press, 2 nd ed. , 2005. • K. Robert Lange, “Surfactants: A Practical Handbook”, Hanser, 1999. • J. Goodwin, “Colloids and interfaces with surfactants and polymers”, Wiley 2 nd ed. , 2009. • B. P. Binks, ” Particles as surfactants – similarities and differences”, Current Opinion Colloid Interface Sci. 7 (2002) 21 (review). • N. D. Denkov et al. , “Role of surfactant type and bubble surface mobility in • foam rheology”, Soft Matter 5 (2009) 3389 (review).

http: //www. bu. edu. eg/staff/mohamedaborya 7 -courses/12004/files