Snow Flakes Naming Binary Covalent Compounds The first

Snow Flakes

Naming Binary Covalent Compounds • The first element in the formula is named first, followed by the second with the suffix -ide • Prefixes are added to the elements name for both the first and the second monoditritetrapentahexaheptaoctanonadeca- 1 2 3 4 5 6 7 8 9 10 Never use mono- for the first element

Polar and Non-polar Covalent Bonds • Reminder: The ability of an atom within a molecule to draw electrons toward itself is called electronegativity. • An electron pair shared between two atoms of the same element is called a non-polar covalent bond – N 2, H 2, F 2 all have non-polar covalent bonds – Each element has the same electronegativity so both atoms attract electrons equally.

Polar and Non-polar Covalent Bonds • What if two different atoms with different electronegativities? – HF – H = 2. 2, F = 4. 0 so F will attract electron better • An electron pair is more likely to reside closer to the nucleus of the atom with the greater electronegativity and forms a polar covalent bond. The greater the difference between the electronegativites, the more polar the bond. H F

Polar and Nonpolar Covalent Bonds • What if the difference between the electronegativities is really large? – Na and F • Na = 0. 93, F = 4. 0 Difference is 3. 07! – Called an ionic bond.
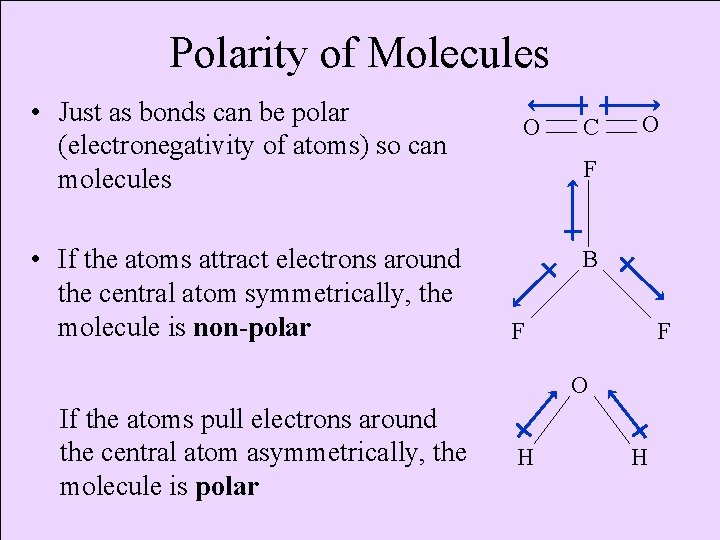
Polarity of Molecules • Just as bonds can be polar (electronegativity of atoms) so can molecules • If the atoms attract electrons around the central atom symmetrically, the molecule is non-polar O C O F B F F O If the atoms pull electrons around the central atom asymmetrically, the molecule is polar H H

Oil and Water • Oil is non-polar • Water is polar • That is why oil and water don’t mix

VSEPR Theory • Valence Shell Electron Pair Repulsion Theory • Use VSEPR to predict the shape of molecules based on the Lewis structure • Shape of the molecule is affected by each of the valence shell electron pairs surrounding a central atom • The regions of electron space around the atom repel each other and strive to get as far away from each other a possible • Region of electron space are: – a non-bonding lone pair – bonding electrons
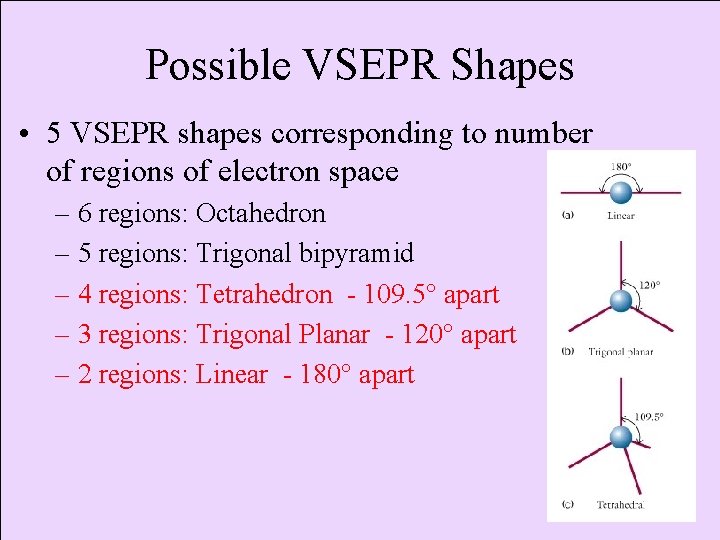
Possible VSEPR Shapes • 5 VSEPR shapes corresponding to number of regions of electron space – 6 regions: Octahedron – 5 regions: Trigonal bipyramid – 4 regions: Tetrahedron - 109. 5° apart – 3 regions: Trigonal Planar - 120° apart – 2 regions: Linear - 180° apart
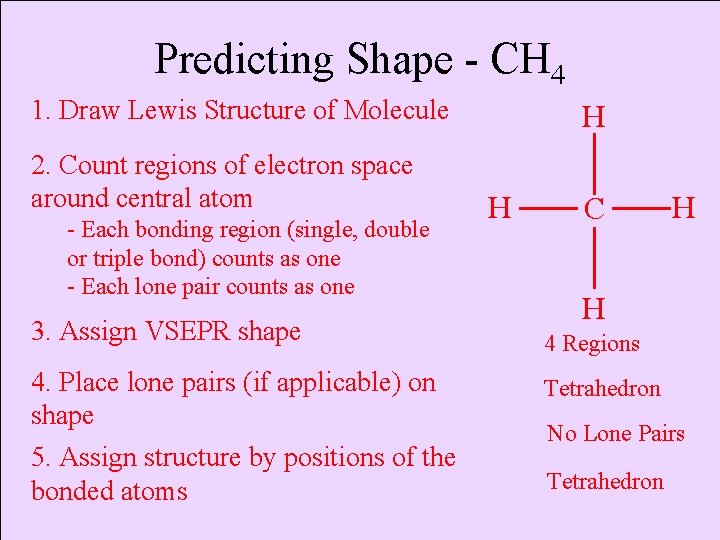
Predicting Shape - CH 4 1. Draw Lewis Structure of Molecule 2. Count regions of electron space around central atom - Each bonding region (single, double or triple bond) counts as one - Each lone pair counts as one H H C H H 3. Assign VSEPR shape 4 Regions 4. Place lone pairs (if applicable) on shape Tetrahedron 5. Assign structure by positions of the bonded atoms Tetrahedron No Lone Pairs
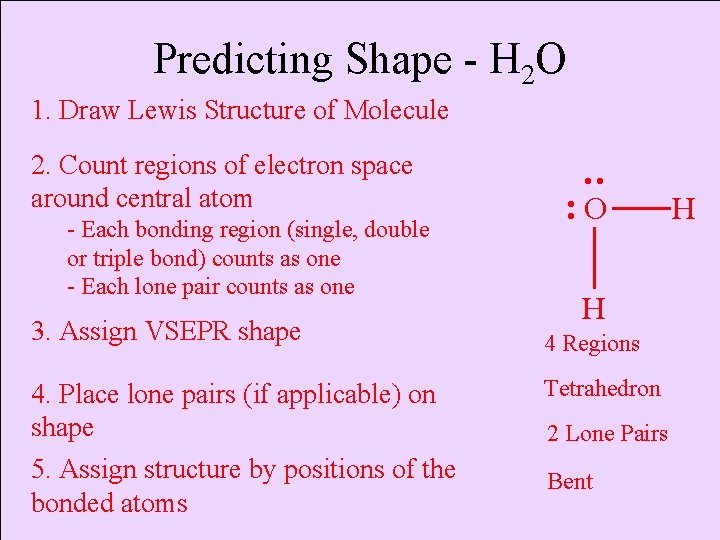
Predicting Shape - H 2 O 1. Draw Lewis Structure of Molecule - Each bonding region (single, double or triple bond) counts as one - Each lone pair counts as one 3. Assign VSEPR shape 4. Place lone pairs (if applicable) on shape 5. Assign structure by positions of the bonded atoms • • 2. Count regions of electron space around central atom O H 4 Regions Tetrahedron 2 Lone Pairs Bent H

Shapes of Molecules

Group Work • Predict the shape and polarity (polar or nonpolar) of the following molecules • NH 3 • BCl 3 • HF

Interactions between molecules • Three phases of matter – Solid • ordered structure • fixed volume and shape independent of container • Strong interaction between molecules – Liquid • loosely ordered • fixed volume, but not fixed shape - dependent on container • Moderate interaction between molecules – Gas • • No order Volume dependent on container Always occupies all of its container Almost no interaction between molecules
- Slides: 14