Snap SOW Thermodynamics FOCI Use BornHaber cycles to
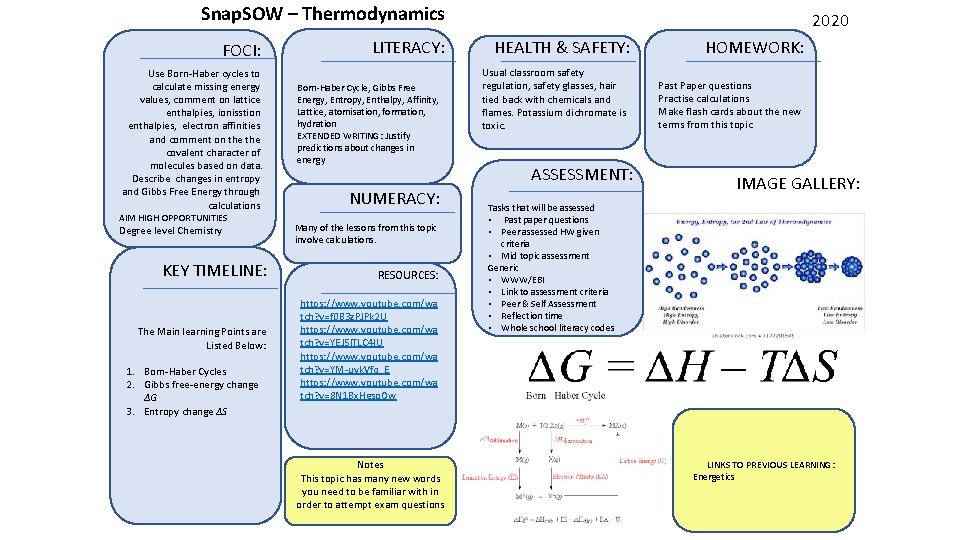
Snap. SOW – Thermodynamics FOCI: Use Born-Haber cycles to calculate missing energy values, comment on lattice enthalpies, ionisstion enthalpies, electron affinities and comment on the covalent character of molecules based on data. Describe changes in entropy and Gibbs Free Energy through calculations AIM HIGH OPPORTUNITIES Degree level Chemistry KEY TIMELINE: The Main learning Points are Listed Below: 1. Born-Haber Cycles 2. Gibbs free-energy change ΔG 3. Entropy change ΔS LITERACY: Born-Haber Cycle, Gibbs Free Energy, Entropy, Enthalpy, Affinity, Lattice, atomisation, formation, hydration EXTENDED WRITING: Justify predictions about changes in energy NUMERACY: Many of the lessons from this topic involve calculations. RESOURCES: https: //www. youtube. com/wa tch? v=f 0 B 3 z. PJPk 2 U https: //www. youtube. com/wa tch? v=YEJ 5 l. TLC 4 IU https: //www. youtube. com/wa tch? v=YM-uyk. Vfq_E https: //www. youtube. com/wa tch? v=8 N 1 Bx. Hgso. Ow Notes This topic has many new words you need to be familiar with in order to attempt exam questions 2020 HEALTH & SAFETY: Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. Potassium dichromate is toxic. ASSESSMENT: HOMEWORK: Past Paper questions Practise calculations Make flash cards about the new terms from this topic IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: Energetics
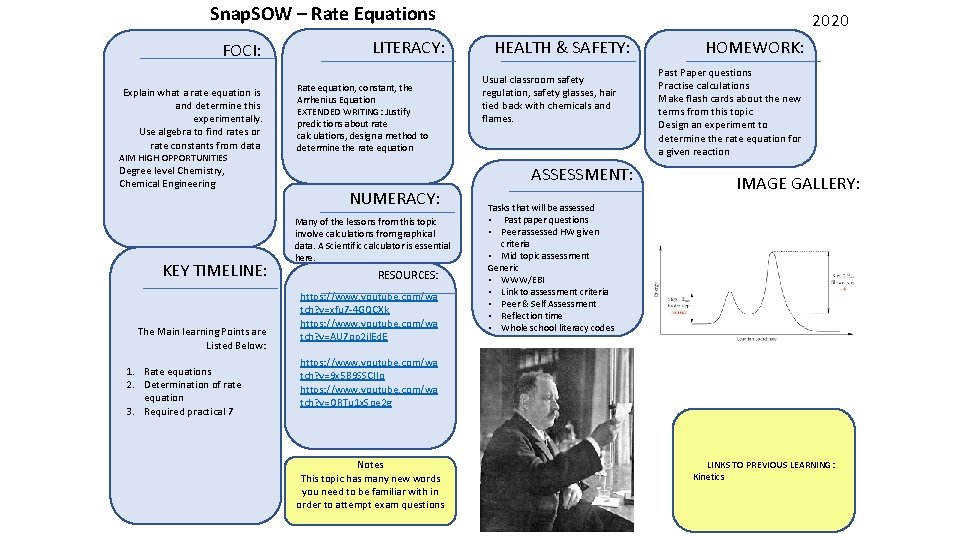
Snap. SOW – Rate Equations FOCI: Explain what a rate equation is and determine this experimentally. Use algebra to find rates or rate constants from data AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering KEY TIMELINE: The Main learning Points are Listed Below: 1. Rate equations 2. Determination of rate equation 3. Required practical 7 LITERACY: Rate equation, constant, the Arrhenius Equation EXTENDED WRITING: Justify predictions about rate calculations, design a method to determine the rate equation 2020 HEALTH & SAFETY: Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: NUMERACY: Many of the lessons from this topic involve calculations from graphical data. A Scientific calculator is essential here. RESOURCES: https: //www. youtube. com/wa tch? v=xfu 7 -4 GQCXk https: //www. youtube. com/wa tch? v=AU 7 op 2 jl. Ed. E HOMEWORK: Past Paper questions Practise calculations Make flash cards about the new terms from this topic Design an experiment to determine the rate equation for a given reaction IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes https: //www. youtube. com/wa tch? v=9 x 5 B 9 SSCIIo https: //www. youtube. com/wa tch? v=ORTu 1 x. Soe 2 g Notes This topic has many new words you need to be familiar with in order to attempt exam questions LINKS TO PREVIOUS LEARNING: Kinetics
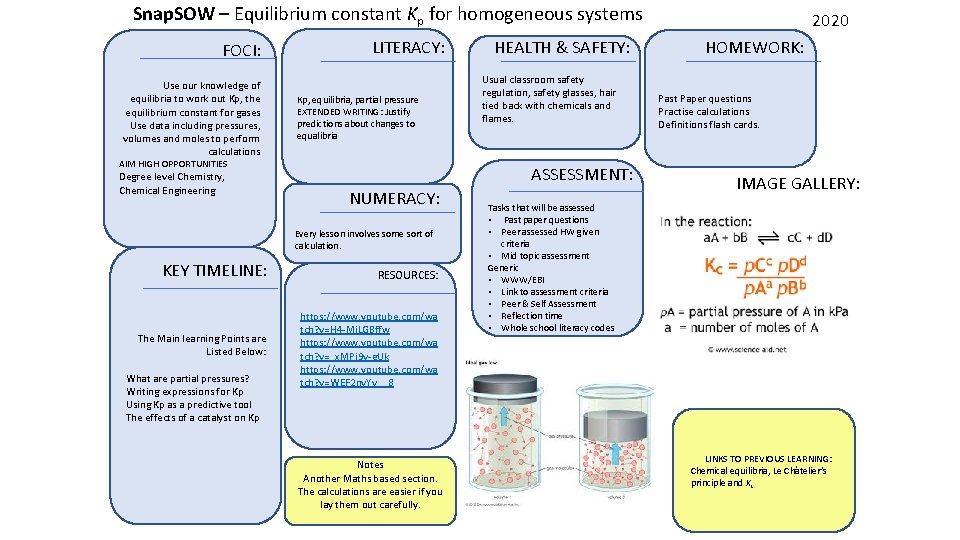
Snap. SOW – Equilibrium constant Kp for homogeneous systems FOCI: Use our knowledge of equilibria to work out Kp, the equilibrium constant for gases Use data including pressures, volumes and moles to perform calculations LITERACY: Kp, equilibria, partial pressure EXTENDED WRITING: Justify predictions about changes to equalibria AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: NUMERACY: Every lesson involves some sort of calculation. KEY TIMELINE: RESOURCES: The Main learning Points are Listed Below: https: //www. youtube. com/wa tch? v=H 4 -Mj. LGBffw https: //www. youtube. com/wa tch? v=_x. MPj 9 v-g. Uk https: //www. youtube. com/wa tch? v=WEF 2 nv. Yv__8 What are partial pressures? Writing expressions for Kp Using Kp as a predictive tool The effects of a catalyst on Kp HEALTH & SAFETY: Notes Another Maths based section. The calculations are easier if you lay them out carefully. 2020 HOMEWORK: Past Paper questions Practise calculations Definitions flash cards. IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: Chemical equilibria, Le Châtelier’s principle and Kc
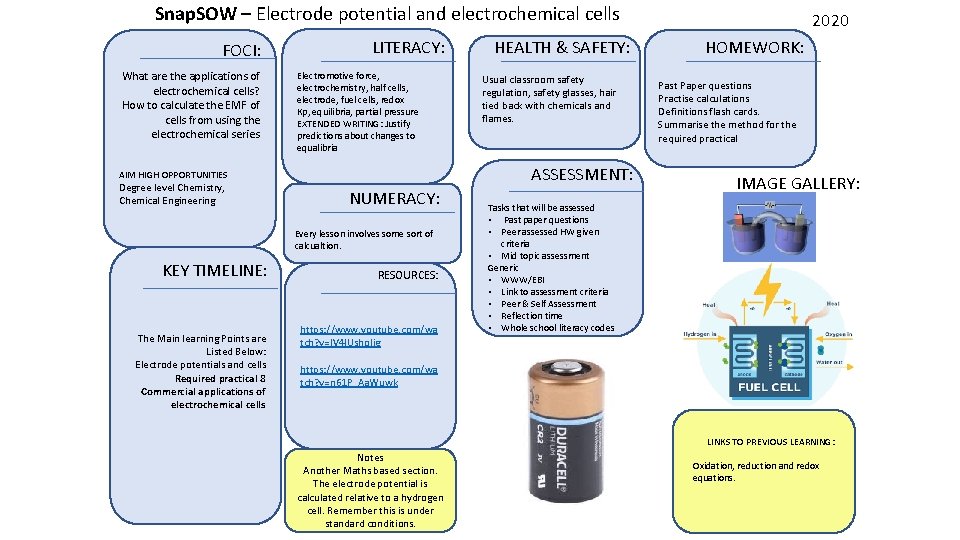
Snap. SOW – Electrode potential and electrochemical cells FOCI: What are the applications of electrochemical cells? How to calculate the EMF of cells from using the electrochemical series LITERACY: Electromotive force, electrochemistry, half cells, electrode, fuel cells, redox Kp, equilibria, partial pressure EXTENDED WRITING: Justify predictions about changes to equalibria NUMERACY: Every lesson involves some sort of calcualtion. KEY TIMELINE: The Main learning Points are Listed Below: Electrode potentials and cells Required practical 8 Commercial applications of electrochemical cells Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering HEALTH & SAFETY: RESOURCES: https: //www. youtube. com/wa tch? v=IV 4 IUsholjg 2020 HOMEWORK: Past Paper questions Practise calculations Definitions flash cards. Summarise the method for the required practical IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes https: //www. youtube. com/wa tch? v=n 61 P_Aa. Wuwk LINKS TO PREVIOUS LEARNING: Notes Another Maths based section. The electrode potential is calculated relative to a hydrogen cell. Remember this is under standard conditions. Oxidation, reduction and redox equations.

Snap. SOW – Acids and bases FOCI: Calculate, p. H, Kw and explain the differecnces between concentration and strength. Use graphical date to find the equivalence point during an acid- base reaction AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering LITERACY: Equivalence point, Kw, p. H, titration, buffer EXTENDED WRITING: Justify the point at which a neutralisation will occur and describe the graph of different acid-base reactions p. H changes. KEY TIMELINE: HEALTH & SAFETY: Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: NUMERACY: Every lesson involves some sort of calculation. The Main learning Points are Listed Below: 1. Brønsted–Lowry acid-base equilibria in aqueous solutions 2. Definition and determination of p. H 3. The ionic product of water Kw 4. Weak acids and bases; Ka for weak acids 5. p. H curves, titrations and indicators 6. Buffer action 7. Required practical 9 2020 RESOURCES: https: //www. youtube. com/wa tch? v=Ziokq. P 0 a. Z 1 E https: //www. youtube. com/wa tch? v=_g. YBbzkqrm. E https: //www. youtube. com/wa tch? v=LS 67 v. S 10 O 5 Y https: //www. youtube. com/wa tch? v=8 Fdt 5 Wn. Yn 1 k HOMEWORK: Past Paper questions Practise calculations Definitions flash cards. Summarise the method for the required practical IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: Notes Some of the calculations found here are similar to the Kc calculations we did last year. Chemical equilibria, Le Châtelier’s principle and Kc
Snap. SOW – Properties of Period 3 elements and their oxides LITERACY: FOCI: State and explain the trends and properties of period 3 elements. AIM HIGH OPPORTUNITIES Bonding, oxides, physical properties, spdf orbitals, shielding, electronegativity, ionisation energy EXTENDED WRITING: Predict the properties of elements in period 3 based on their atomic structure. Degree level Chemistry, Chemical Engineering Graphical data The Main learning Points are Listed Below: 1. Reactions of Na and Mg with water. 2. Reactions of Na, Mg, Al, Si, P and S with oxygen. 3. Melting points of period 3 oxides. 4. Reactions of period 3 oxides with water. Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: NUMERACY: KEY TIMELINE: HEALTH & SAFETY: RESOURCES: https: //www. youtube. com/wa tch? v=msq. Qq 3 TNekg https: //www. youtube. com/wa tch? v=67 Aq. Mm. Xg 0 NA 2020 HOMEWORK: Past Paper questions Practise calculations Flash cards with explanations of properties IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: Notes Go back over your y 12 work on periodicity as this follows directly on from this. . Bonding, Periodicity
Snap. SOW – Transition Metals FOCI: What are the transition metal elements properties? What are ligands and how do they interact with transition metal elements? What are their oxidation states? AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering LITERACY: Transition metals, oxidation states, ligands, substitution, complex ion, haemoglobin EXTENDED WRITING: Describe and explain what happens when ions are oxidised and reduced under certain conditions The Main learning Points are Listed Below: 1. General properties of the transition metals 1. Substitution reactions 2. Shapes of complex ions 3. Formation of coloured ions 4. Variable oxidation states 5. Catalysts HEALTH & SAFETY: Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: NUMERACY: Use data to draw conclusions KEY TIMELINE: 2020 RESOURCES: https: //www. youtube. com/wa tch? v=Lmu 1 Bv. ABs. NE https: //www. youtube. com/wa tch? v=u 2 g. OKRYZFGw https: //www. youtube. com/wa tch? v=ydaka 3 c 4 d_w Notes If you enlarge the image on the right it has a summary of some of the colour changes you may see. HOMEWORK: Past Paper questions Flash cards with shapes and colours of complexes Practise questions on substitutions IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: Atomic structure, Oxidation, reduction and redox reactions
Snap. SOW – Aldehydes, Ketones and Optical Isomerism, Carboxylic Acids, Acylation and Esters FOCI: How are optical isomers produced and what are their properties? What are the reactions of aldehydes and ketones, what is produced and why? How are esters made, used and hydrolysed? What is acylation? AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering KEY TIMELINE: The Main learning Points are Listed Below: 1. Optical Isomerism 2. The cause and nature of optical isomerism 3. Oxidation and reduction of ketones and aldehydes 4. The structure of carboxylic acids and esters 5. The uses and manufacture of esters 6. Hydrolysis of esters 7. Acylation 8. Required practical 10 LITERACY: Acylation, racemate, optical isomer, enantiomer, aldehyde, ketone, EXTENDED WRITING: Describe and explain how to get from starting materials to target products. HEALTH & SAFETY: Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: NUMERACY: Interleaving questions RESOURCES: https: //www. youtube. com/wa tch? v=RBtg. Az 70_JY https: //www. youtube. com/wa tch? v=Sf. Nih. K 9 Qbp. U https: //www. youtube. com/wa tch? v=3 Ye. XGp. Ddg. Zw https: //www. youtube. com/wa tch? v=mf. Xj. PT 9 J 5 Sc Notes: There a number of new terms in this topic. Make sure you are confident with them. HOMEWORK: 2020 Past Paper questions Practise questions on the reactions Produce a mind map of the pathways covered IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: Isomerism Aldehydes and ketones, Nomenclature Reaction mechanisms Oxidation of alcohols
Snap. SOW – Aromatic Chemistry, Electrophilic Substitution and Amines FOCI: Describe the bonding in aromatic compounds Explain the mechanism and conditions for electrophilic substitutions and the preparation of amines AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering LITERACY: Electrophile, aromatic, aliphatic, amine, Friedel-Crafts, quaternary salts, acyl chloride, acid anhydride EXTENDED WRITING: transform reaction mechanisms into detailed descriptions and vice versa. The Main learning Points are Listed Below: 1. Bonding in aromatic chemicals 2. Electrophilic substitution 3. Preparation of Amines 4. Base properties of amines 5. Nucleophilic properties of amines Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: NUMERACY: Interleaving questions KEY TIMELINE: HEALTH & SAFETY: RESOURCES: https: //www. youtube. com/wa tch? v=k. XFEex-d. ABU https: //www. youtube. com/wa tch? v=fpq 0 e. ICju. SI https: //www. youtube. com/wa tch? v=JGr. S 8 nood 8 U Notes: Make sure to revise your mechanisms work before starting this topic. HOMEWORK: 2020 Past Paper questions Practise questions on the reactions Make flash cards to revise mechanisms IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: Isomerism Nomenclature Reaction mechanisms Nucleophilic substitution
Snap. SOW – Polymers FOCI: Describe how different types of polymers form. Explain the structure of different synthetic and natural polymers. Explain the problems with synthetic polymers and some solutions to these. AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering, Biochemistry KEY TIMELINE: The Main learning Points are Listed Below: 1. Condensation polymers 2. Biodegradability and disposal of polymers 3. Amino acids, proteins and DNA 4. Amino acids 5. Proteins 6. Enzymes 7. DNA 8. Action of anti-cancer drugs LITERACY: Polyesters, polyamides, condensation polymerisation, hydrolysis, zwitterion EXTENDED WRITING: Explain the structure of proteins HEALTH & SAFETY: Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: NUMERACY: Interleaving questions RESOURCES: https: //www. youtube. com/wa tch? v=r. Hxx. LYz. J 8 Sw https: //www. youtube. com/wa tch? v=H 82 bc. KFB 4 i. Y https: //www. youtube. com/wa tch? v=D 3 m. IRev. Wzc 8 Notes: Make sure to revise your mechanisms work before starting this topic. HOMEWORK: 2020 Past Paper questions Practise questions on the reactions Make flash cards to revise how amino acids bond and interact IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: Isomerism Nomenclature Addition polymers Forces between molecules Carboxylic acids Amines Chromatography
Snap. SOW – Organic Synthesis and Analysis FOCI: Explain how to produce target molecules form given starting compounds. Understand different analytical techniques to deduce the identity of unknown compounds. AIM HIGH OPPORTUNITIES Degree level Chemistry, Chemical Engineering, Biochemistry, Biological Sciences KEY TIMELINE: The Main learning Points are Listed Below: 1. Organic synthesis 2. Nuclear magnetic resonance spectroscopy 3. Chromatography 4. Required practical 12 LITERACY: Thin layer chromatography, NMR, EXTENDED WRITING: Devise routes to produce target chemicals and explain these in text or diagrams. Explain how to analyse unknown chemicals, explaining the conditions and steps to do so. NUMERACY: Interpreting NMR spectra and chromatograms RESOURCES: https: //www. youtube. com/wa tch? v=rh 0 Tn_o. PS 30 https: //www. youtube. com/wa tch? v=4 G_RU 51 t_UM https: //www. youtube. com/wa tch? v=SBir 5 w. US 3 Bo https: //www. youtube. com/wa tch? v=qdm. KGsk. Cyh 8 Notes: This topic draw in aspects from all of the organic topics in A level. HEALTH & SAFETY: Usual classroom safety regulation, safety glasses, hair tied back with chemicals and flames. ASSESSMENT: HOMEWORK: 2020 Past Paper questions Practise questions on the reactions Writing up required practical 12 IMAGE GALLERY: Tasks that will be assessed • Past paper questions • Peer assessed HW given criteria • Mid topic assessment Generic • WWW/EBI • Link to assessment criteria • Peer & Self Assessment • Reflection time • Whole school literacy codes LINKS TO PREVIOUS LEARNING: All organic chemistry topics Organic analysis Nomenclature Amino acids, proteins and DNA
- Slides: 11