Slovak University of Technology in Bratislava Institute of

Slovak University of Technology in Bratislava Institute of Information Engineering, Automation, and Mathematics Laboratory distillation column (Semester project) Student: Supervisor: Bc. Martin Mojto doc. Ing. Radoslav Paulen, Ph. D.
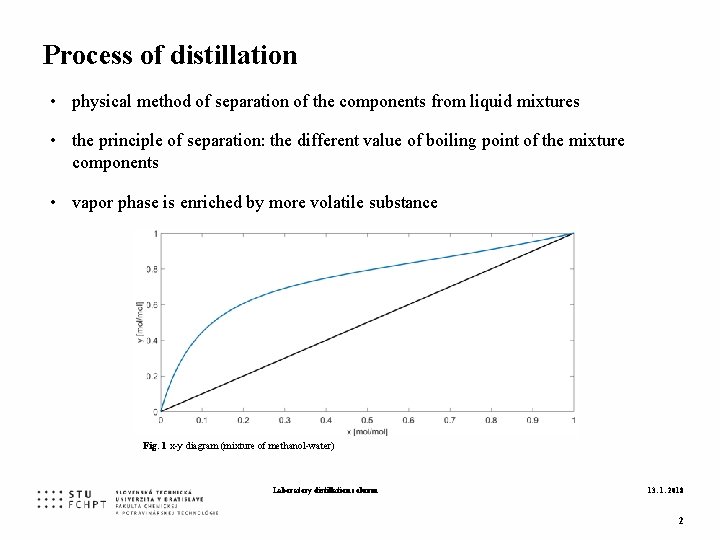
Process of distillation • physical method of separation of the components from liquid mixtures • the principle of separation: the different value of boiling point of the mixture components • vapor phase is enriched by more volatile substance Fig. 1 x-y diagram (mixture of methanol-water) Laboratory distillation column 13. 1. 2018 2
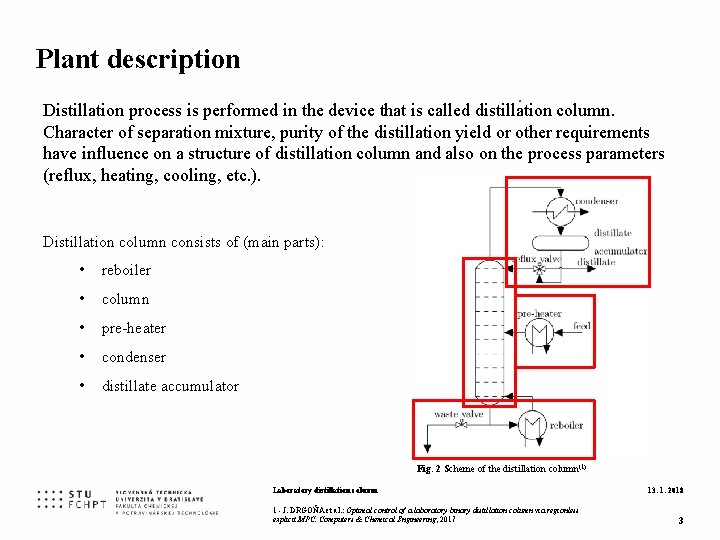
Plant description. Distillation process is performed in the device that is called distillation column. Character of separation mixture, purity of the distillation yield or other requirements have influence on a structure of distillation column and also on the process parameters (reflux, heating, cooling, etc. ). Distillation column consists of (main parts): • reboiler • column • pre-heater • condenser • distillate accumulator Fig. 2 Scheme of the distillation column(1) Laboratory distillation column 1 - J. DRGOŇA et al. : Optimal control of a laboratory binary distillation column via regionless explicit MPC. Computers & Chemical Engineering, 2017 13. 1. 2018 3

Plant description Laboratory distillation column (UOP 3 CC) q Reboiler § consist of a flameproof immersion type heating element § maximum power of 2. 5 k. W q Pre-heater § maximum power of 2 k. W q Column § § 8 sieve plate trays with 50 mm diameter feed flow introduced on the 5 th tray 2 m tall insulated q Condenser § distillate accumulator § reflux valve Fig. 3 Scheme of the distillation column(1) UOP 3 CC Laboratory distillation column 1 - J. DRGOŇA et al. : Optimal control of a laboratory binary distillation column via regionless explicit MPC. Computers & Chemical Engineering, 2017 13. 1. 2018 4

Assumptions • constant value of pressure in the entire column (atmospheric) • no heat loss (column is insulated) from the plant • pre-heater adjusts the temperature of the feed flow to the boiling point • a total condenser • ideal vapor phase and non-ideal liquid phase • ideal mixing of the liquid and vapor phases • 100% efficiency of each tray in the column • no hold-up of vapor phase on the trays, condenser or reboiler Laboratory distillation column 13. 1. 2018 5
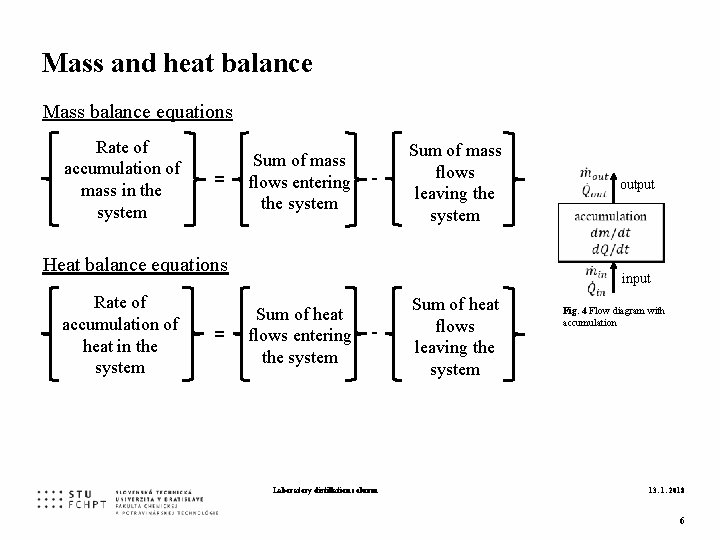
Mass and heat balance Mass balance equations Rate of accumulation of mass in the system = Sum of mass flows entering the system - Sum of mass flows leaving the system Heat balance equations Rate of accumulation of heat in the system = Sum of heat flows entering the system - Laboratory distillation column Sum of heat flows leaving the system output input Fig. 4 Flow diagram with accumulation 13. 1. 2018 6
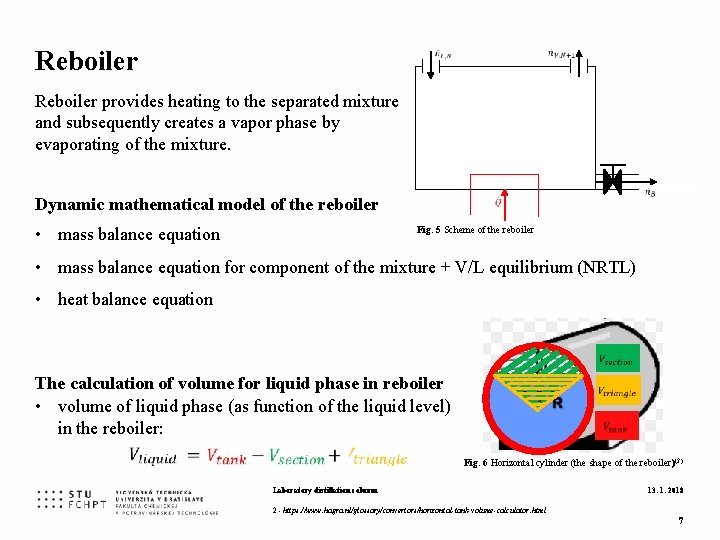
Reboiler provides heating to the separated mixture and subsequently creates a vapor phase by evaporating of the mixture. Dynamic mathematical model of the reboiler • mass balance equation Fig. 5 Scheme of the reboiler • mass balance equation for component of the mixture + V/L equilibrium (NRTL) • heat balance equation The calculation of volume for liquid phase in reboiler • volume of liquid phase (as function of the liquid level) in the reboiler: Fig. 6 Horizontal cylinder (the shape of the reboiler)(2) Laboratory distillation column 13. 1. 2018 2 - https: //www. hagra. nl/glossary/convertors/horizontal-tank-volume-calculator. html 7
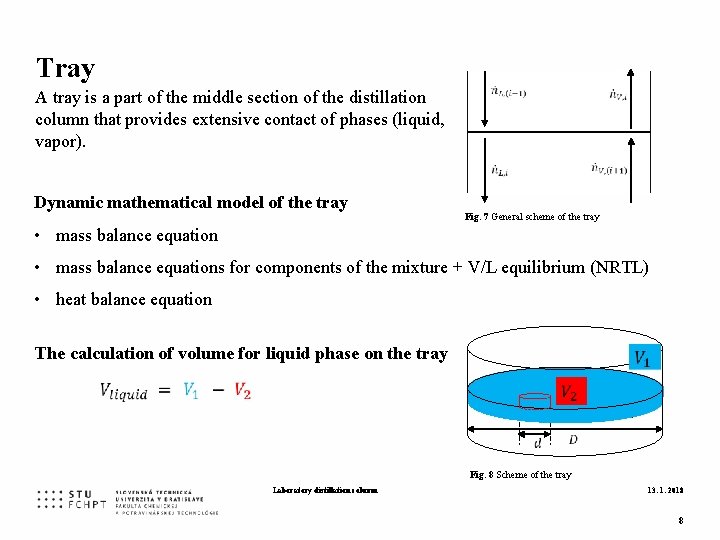
Tray A tray is a part of the middle section of the distillation column that provides extensive contact of phases (liquid, vapor). Dynamic mathematical model of the tray Fig. 7 General scheme of the tray • mass balance equations for components of the mixture + V/L equilibrium (NRTL) • heat balance equation The calculation of volume for liquid phase on the tray Fig. 8 Scheme of the tray Laboratory distillation column 13. 1. 2018 8
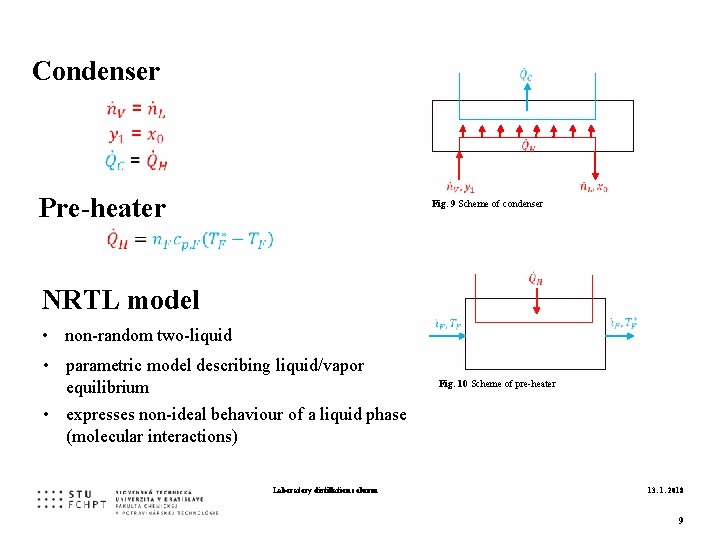
Condenser Pre-heater Fig. 9 Scheme of condenser NRTL model • non-random two-liquid • parametric model describing liquid/vapor equilibrium • expresses non-ideal behaviour of a liquid phase (molecular interactions) Laboratory distillation column Fig. 10 Scheme of pre-heater 13. 1. 2018 9

g. PROMS Fig. 11 Workspace of the g. PROMS Process. Builder (libraries, project tree, etc. )(4) Laboratory distillation column 13. 1. 2018 4 – https: //www. psenterprise. com/products/gproms/processbuilder 10

g. PROMS • an environment for expert modelers to build, validate and execute steady-state and dynamic process models • suitable for solving the differential-algebraic equations • has a transparent workspace and easy access to models a composite model (linked with another models) a stand-alone model (no connection to other models) • each model can be simulated independently or linked with another models Fig. 12 Project tree Laboratory distillation column 13. 1. 2018 11
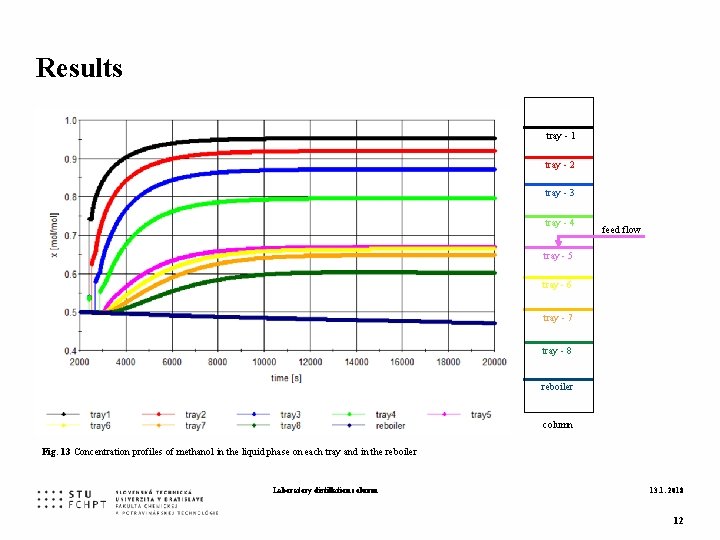
Results tray - 1 tray - 2 tray - 3 tray - 4 feed flow tray - 5 tray - 6 tray - 7 tray - 8 reboiler column Fig. 13 Concentration profiles of methanol in the liquid phase on each tray and in the reboiler Laboratory distillation column 13. 1. 2018 12

Conclusion Laboratory distillation column • mathematical model was analysed and created according to specific parameters • dynamic behaviour of the column resulted from mass and heat balance equations • simulation was performed in the g. PROMS environment • results were validated by theory of heat and mass transfer Future work • compare simulated data against experimental data Laboratory distillation column 13. 1. 2018 13

Backup page Laboratory distillation column 13. 1. 2018 14

Backup page Laboratory distillation column 13. 1. 2018 15

Backup page Laboratory distillation column 5 – PRAUSNITZ et al. : Molecular Thermodynamics of Fluid-Phase Equilibria, University of California, Berkeley, 1999, ISBN 0 -13 -977745 -8 13. 1. 2018 16

Backup page Laboratory distillation column 13. 1. 2018 3 – BAFRNCOVA S. , SEFCIKOVA M. , VAJDA M. ; Chemické inžinierstvo – Tabuľky a grafy 17
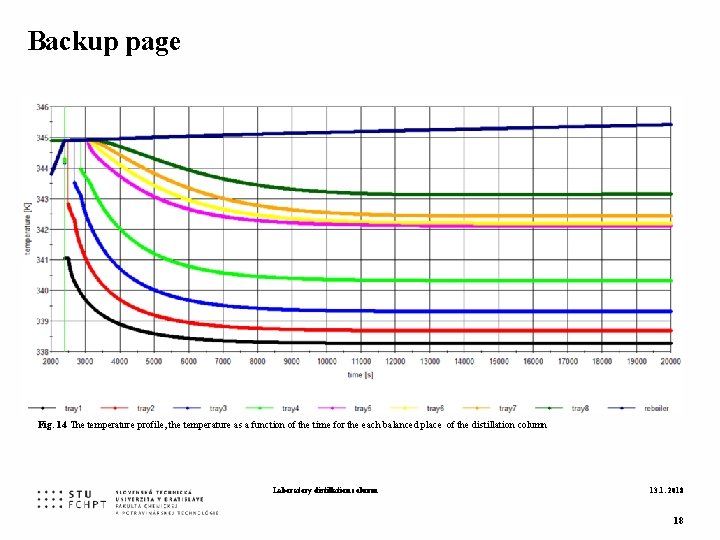
Backup page Fig. 14 The temperature profile, the temperature as a function of the time for the each balanced place of the distillation column Laboratory distillation column 13. 1. 2018 18
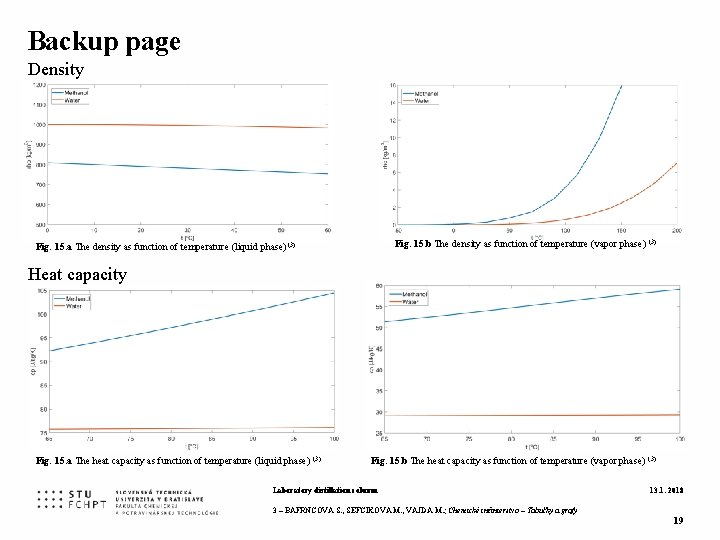
Backup page Density Fig. 15. b The density as function of temperature (vapor phase) (3) Fig. 15. a The density as function of temperature (liquid phase) (3) Heat capacity Fig. 15. a The heat capacity as function of temperature (liquid phase) (3) Fig. 15. b The heat capacity as function of temperature (vapor phase) (3) Laboratory distillation column 13. 1. 2018 3 – BAFRNCOVA S. , SEFCIKOVA M. , VAJDA M. ; Chemické inžinierstvo – Tabuľky a grafy 19
- Slides: 19