Slovak University of Technology Bratislava Faculty of Chemical

Slovak University of Technology, Bratislava Faculty of Chemical and Food Technology Dep. of Organic Technology Joint Slovak-Hungarian Laboratory for Development of Catalyzed Chemical Processes of Biomass Utilization* Catalytic transformation of polyols to carbonates *Founded by financial support of project No. HUSK/1101/1. 2. 1/0318 “Chemical Processes of Biomass Utilization in the Slovak. Hungarian Frontier Region” László Seemann, Alexander Kaszonyi, 17 th October, 2012, Bratislava

High boiling point Low toxicity Biodegradability Uses Properties Glycerol carbonate Cosmetics, medicines, polycarbonates and polyurethanes Replacement for hazardous and volatile organic solvents Other uses. . .

Preparation of glycerol carbonate TRANSESTERIFICATION OF EC WITH GLY 80°C, 90 min Catalyst: TBA-MCM 41 (X = 94%, S = 84%) H. J. Cho at al. , J. of Indust. and Engineering Chem. 16, 2006, 679 -683 40°C, 10 -13 MPa, 90 min in SC-CO 2 Cat. : Amberlist 26 (Y = 4, 7%), Purosiv (Y = 32%) C. Vievelle at al. , Catal. Letters 56, 1998, 245 -247

Preparation of glycerol carbonate TRANSESTERIFICATION OF DMC WITH GLY 75 -90°C, 50~90 min DMC/GLY = 3, 5 (mol/mol) KAT/GLY = 0, 06 -0, 08 (mol/mol) Azeotropic agent: benzene (1, 5×Me. OH) Cat. : Ca. O, Ca. CO 3 (ht, b; Y=~91%, X>91%) KOH, K 2 CO 3 (hm, b; Y, X = 100%) J. R. Oucha-Gómez at al. , App. Catal. A: Gen. 366, 2009, 315 -324, J. Li at al. , Chem. Engineering and Proc. 49, 2010, 530 -535, F. S. Hubertson Simanjuntak at al. , App. Cat. A: Gen. 401, 2011, 220 -225, R. Bai at al. , J. of Indust. And Engineering Chem. 17, 2011, 777 -781, M. Malyaadri at al. , App. Catal. A: Gen. 401, 2011, 153 -157 78°C, 50 min DMC/GLY 1 (mol/mol) KAT/GLY 4 (w/w) Cat. : KF/HAP, KF. 2 H 2 O (ht, b; Y, X >99%) 75°C, 90 min DMC/GLY = 1: 5 (mol/mol) KAT/GLY = 0, 1 (w/w) Cat. : MAZ-311 (Y, X ~95%)

Preparation of glycerol carbonate OXIDATIVE CARBONYLATION OF GLY 140°C, PCO = 2 MPa, PO 2 = 1 MPa, 2 h, Pd(phen)/KI (X = 92%, S = 99%) GLY/Pd = 400 (mol/mol) J. Hu at al. , App. Catal. A: Gen. 386, 2010, 188 -193 20°C, 0, 1 MPa K 2 CO 3 (5 eq. ), GLY (5 eq. ), Se (1 eq. ), (Y = 84%), DMF solvent T. Mizuno at al. , Heteroatom Chem. , 2010

Preparation of glycerol carbonate DIRECT CARBOXYLATION OF GLY 180°C, 5 MPa, 6 -15 h Sn based cat. (max X = 7%) M. Aresta a kol. , J. of Mol. Catal. A: Chem. 257, 2006, 149 -153 without cat. , 40°C, 10 MPa, 3 h, in SC-CO 2, Y = 0 of GC C. Vievelle at al. , Catal. Letters 56, 1998, 245 -247

Preparation of glycerol carbonate GLYCEROLYSIS OF UREA 140°C, 20 Pa, 3 h GLY/U = 1 (mol/mol) KAT/U = 100% (w/w) Cat. : g-Zr. P (X = 76%) 140°C, 3 k. Pa, 1 h GLY/U = 2 (mol/mol) KAT/GLY = 0, 22% (w/w) Cat. : La 2 O 3 (Y = 89%, X = 99%) P = atm. , N 2, 4 h (X = 60%, Y = 3%) 145°C, 4 k. Pa, 5 h GLY/U = 1 (mol/mol), KAT/GLY = 5% (w/w) Cat. : HTc-Zn (Y = 72, X = 82%) M. Aresta at al. , J. of Catal. 268, 2009, 106 -114, L. Wang at al. , Catal. Communications 12, 2011, 1458 -1462, M. J. Climent at al. , J. of Catal. 269, 2010, 140 -149, J. L. Dubois at al. , EP 2174937 A 1, 2010, J. L. Dubois at al. , WO 2010/040786 A 2, 2010

Overview of our studies Different types of homogeneous catalysts (Zn. Cl 2, Zn. O, Zn(NO 3)2, Mg. Cl 2, Mg. O, Alumina, also reaction without catalyst) Optimization of catalyst composition and reaction conditions(G/C, T, G/U) Heterogenization of the best homogeneous catalysts (Zn. O/Alumina, Zn. Cl 2/Silica, etc. )
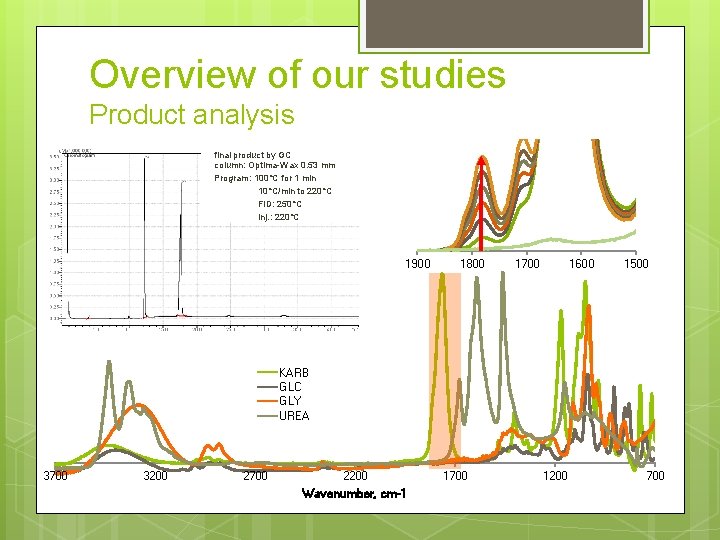
Overview of our studies Product analysis final product by GC column: Optima-Wax 0. 53 mm Program: 100°C for 1 min 10°C/min to 220°C FID: 250°C Inj. : 220°C 1900 1800 1700 1600 1500 KARB GLC GLY UREA 3700 3200 2700 2200 Wavenumber, cm-1 1700 1200 700
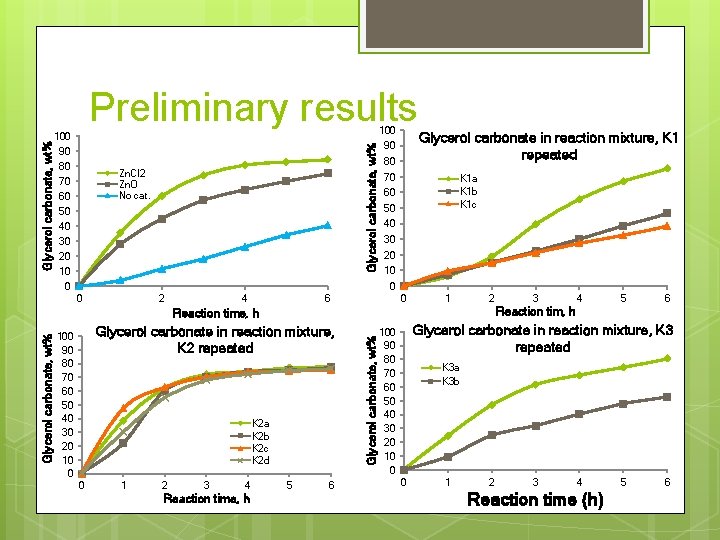
100 Glycerol carbonate, wt% Preliminary results 90 80 Zn. Cl 2 Zn. O No cat. 70 60 50 40 30 20 10 90 80 Glycerol carbonate in reaction mixture, K 1 repeated 70 K 1 a K 1 b K 1 c 60 50 40 30 20 10 0 2 4 0 6 1 2 Glycerol carbonate in reaction mixture, K 2 repeated K 2 a K 2 b K 2 c K 2 d 0 1 2 3 4 Reaction time, h 5 6 Glycerol carbonate, wt% Reaction time, h 100 90 80 70 60 50 40 30 20 10 0 3 4 5 6 Reaction tim, h Glycerol carbonate in reaction mixture, K 3 repeated 100 90 80 70 60 50 40 30 20 10 0 K 3 a K 3 b 0 1 2 3 4 Reaction time (h) 5 6
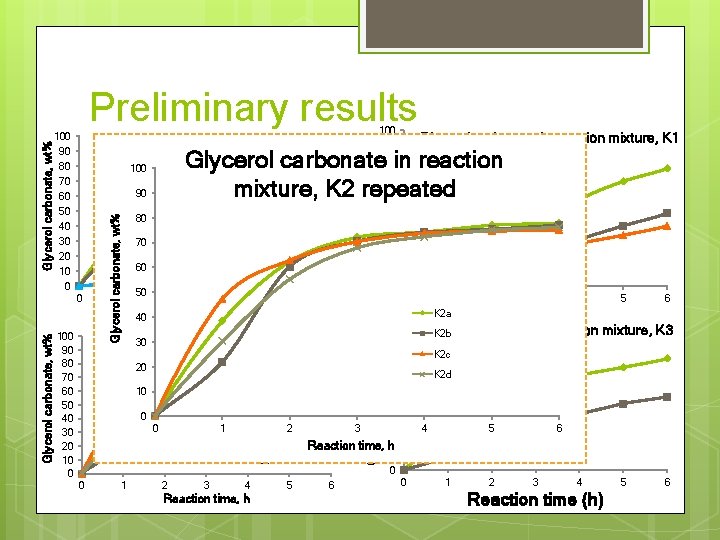
100 90 60 50 40 30 20 10 0 80 70 60 2 4 30 20 10 0 1 K 2 a Reaction time, h 40 10 0 0 1 40 6 20 0 50 Glycerol carbonate in reaction mixture, 30 K 2 repeated 100 90 80 70 60 50 40 30 20 10 0 Glycerol carbonate in reaction mixture, K 1 repeated Glycerol carbonate 80 in reaction 70 K 1 a K 1 b 60 mixture, K 2 repeated K 1 c 100 Zn. Cl 2 Zn. O 90 No cat. 70 90 K 2 a K 2 b K 2 c K 2 d 1 2 3 4 Reaction time, h 2 5 6 2 3 4 5 6 Reaction tim, h Glycerol carbonate in reaction mixture, K 3 K 2 b repeated K 2 c 100 90 80 70 60 50 40 3 30 Reaction time, 20 h 10 0 Glycerol carbonate, wt% 80 0 Glycerol carbonate, wt% 100 Glycerol carbonate, wt% Preliminary results K 3 a K 2 d K 3 b 4 0 5 1 2 6 3 4 Reaction time (h) 5 6

Aim of studies Preparation of stable heterogenized catalyst Catalyst characterization Rate of GC formation and decomposition at different reaction conditions Purification of GC
Thank you for your attention This work was supported by the Slovak Research and Development Agency under the contract No. APVV-0133 -11
- Slides: 13