Slide 1 of 21 Supplier Auditing Remote Auditing




















- Slides: 21

Slide 1 of 21 Supplier Auditing & Remote Auditing Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 2 of 21 Purpose of Supplier Audits • Qualify a New Supplier • On-going Evaluation of a Supplier • CAPA or Complaint Investigation Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 3 of 21 Can I justify fewer supplier audits? Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 4 of 21 Can you justify more supplier audits? Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

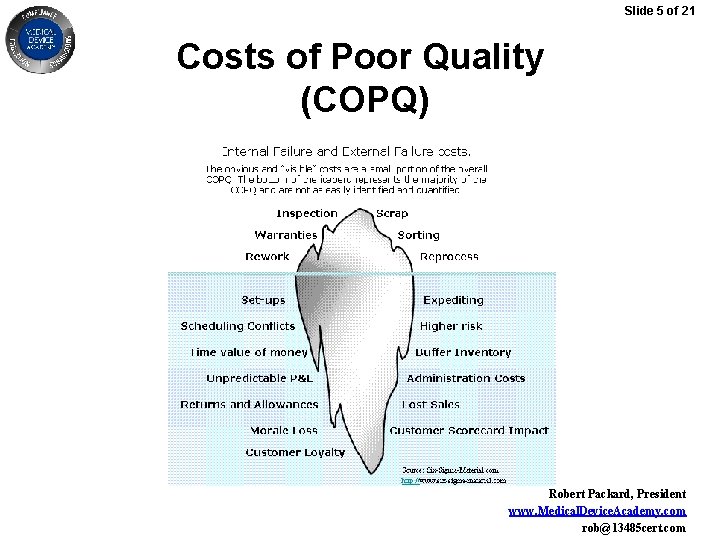
Slide 5 of 21 Costs of Poor Quality (COPQ) Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 6 of 21 Auditing 100% of your critical suppliers may seem impossible due to limited resources, but have you ever seen a cost/benefit analysis? Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 7 of 21 4 Most Common Auditing Methods 1. Element Approach 2. Procedural Approach 3. Product Approach 4. Process Approach Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 8 of 21 Prioritizing Supplier Audits • FDA Approach • Risk-based Approach • Logistical Approach • Strategic Approach Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 9 of 21 Risk-Based Approach http: //bit. ly/Audit-Recommendations • Critical Subcontractors – Finished Device Manufacturers – Contract Sterilizers – Contract Labeling & Packaging • Crucial Components Suppliers Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 10 of 21 Strategic Selection of Auditors Select Auditors with Technical Expertise Training Opportunities for New Auditors Gathering Technical Information Discussion of Design Changes / Process Changes • Use the Adjacent Link Auditing Approach • • http: //bit. ly/Adjacent. Link. Auditing Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 11 of 21 Use Supplier Audit Template • Focus on Production & Process Controls – Shipping – Final Inspection – Manufacturing – In-Process Inspection – Labeling – Warehouse – Incoming Inspection – Purchasing Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 12 of 21 Supplier Audit Agenda Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 13 of 21 Auditing SCAR Closure http: //www. medicaldeviceacademy. com/supplier-auditing-tool-kit Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 14 of 21 Remote Auditing http: //medicaldeviceacademy. com/4 -ways-make-best-use-remote-audits/ “Any time you can audit in a conference room, you could probably do the audit remotely. ” Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 15 of 21 When NOT to Audit Remotely • Onsite visits facilitate the building of suppliercustomer relationships. • Touring facilities and watching demonstration of processes improves understanding of a supplier’s processes better than reading documents and records can. • Cleanliness and capabilities of suppliers are best evaluated onsite, where camera angles can be used to crop out important details • Sometimes suppliers misrepresent their capabilities by showing photographs on their website of other companies. Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 16 of 21 What Areas Can You Audit Remotely? Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 17 of 21 Examples of Remote Audits • • • CAPA & SCARs Management Review Internal Auditing Supplier Controls Complaint Handling Adverse Event Reporting Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 18 of 21 Other Remote Audits… • Re-Audits (i. e. , SCAR follow-up) • When a critical team member can’t travel or is only needed for a short duration • To address issues of confidentiality when competitor product is also made at supplier. • Pre-cursor to a “for cause” audit / investigation • Participation in opening / closing meetings Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 19 of 21 Allocation of Resources Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 20 of 21 Q&A rob@13485 cert. com Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com

Slide 21 of 21 Need a quotation for a supplier audit? rob 13485 rob@13485 cert. com Rob Packard +1. 802. 281. 4381 Robert Packard, President www. Medical. Device. Academy. com rob@13485 cert. com