SKITTLES ISOTOPE LAB DONT EAT THE SKITTLES PRELAB

SKITTLES ISOTOPE LAB DON’T EAT THE SKITTLES

PRE-LAB QUESTIONS 1. What determines the atomic mass of an element? 2. Where is the vast majority of an atom’s mass found? 3. Compared to a proton, what is the relative mass of an electron? 4. What is an isotope?

SKITTLES ISOTOPE LAB PRE-LAB: COPY DOWN THIS PAGE IN PREPARATION Purpose: In this activity, we will calculate the average atomic mass of a sample of Skittle atoms by determining the number of each type of isotope. This is a model used to represent actual elements, which have multiple isotopes. The element is Skittle and the isotopes are represented by the different colors. Procedure 1. Write the color & atomic mass (see below) for each isotope in your sample in the data table 2. Count the number of each isotope. Multiply this number by the atomic mass of that isotope to find the total mass contributed by that isotope. 3. Find the total mass of your sample by adding up the total masses of all the isotopes. 4. Divide the total mass of the isotope by the total sample mass to get % of total mass. 5. Add all total number of isotopes together to get the total number of atoms. 6. Divide the total mass of the sample by the total number of atoms to get the average atomic mass of the sample. Red: 40 amu 45 amu Orange: 41 amu Yellow: 42 amu Green: 44 amu Purple:
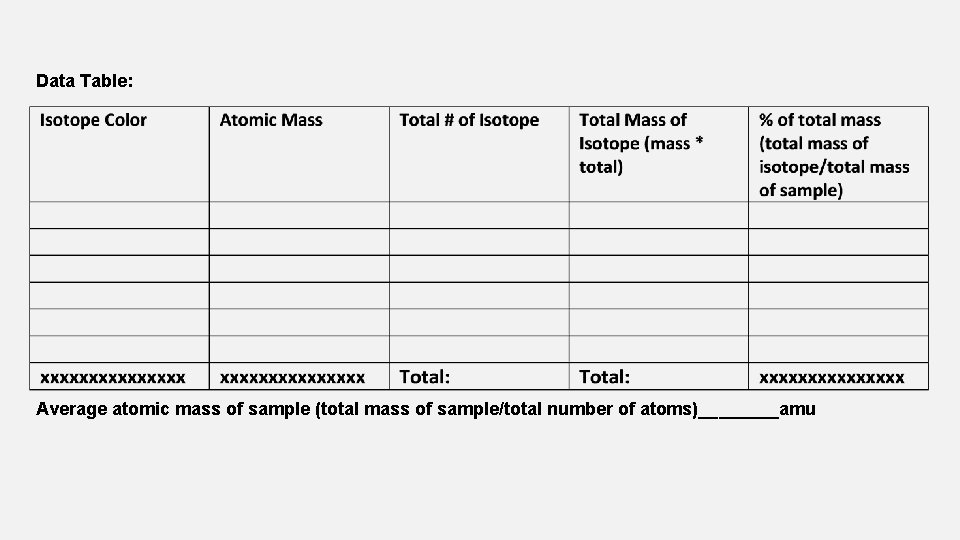
Data Table: Average atomic mass of sample (total mass of sample/total number of atoms)____amu

POST-LAB QUESTIONS 1. Which isotope was the most abundant in your skittles sample? 2. What do all of your skittle atoms have in common? 3. What could be different among the atoms of an element? 4. Can different atoms of an element (isotopes) have different atomic numbers? Explain.
- Slides: 5