Skeletal Muscle Metabolism Presented by Asmaa Mohamed Noha

Skeletal Muscle Metabolism Presented by: Asmaa Mohamed Noha Salah
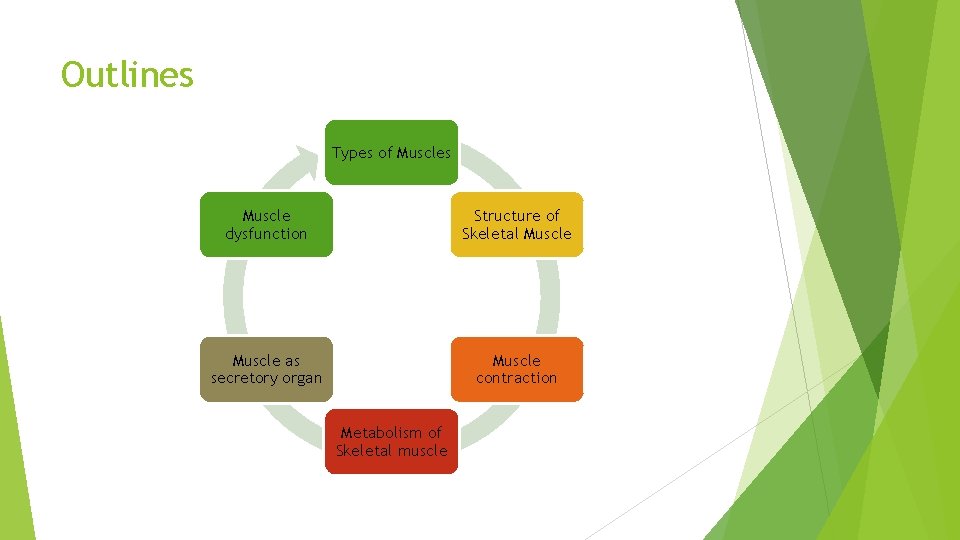
Outlines Types of Muscles Muscle dysfunction Structure of Skeletal Muscle as secretory organ Muscle contraction Metabolism of Skeletal muscle
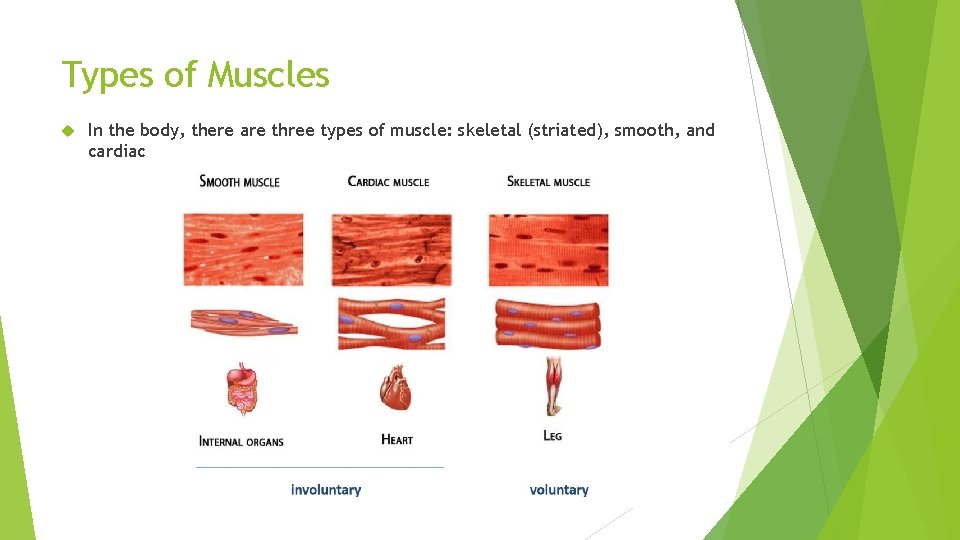
Types of Muscles In the body, there are three types of muscle: skeletal (striated), smooth, and cardiac
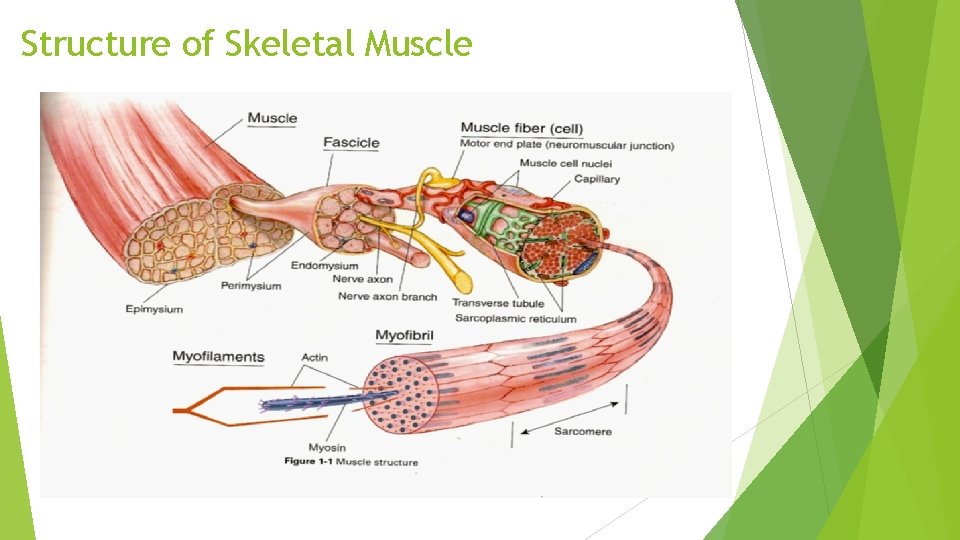
Structure of Skeletal Muscle
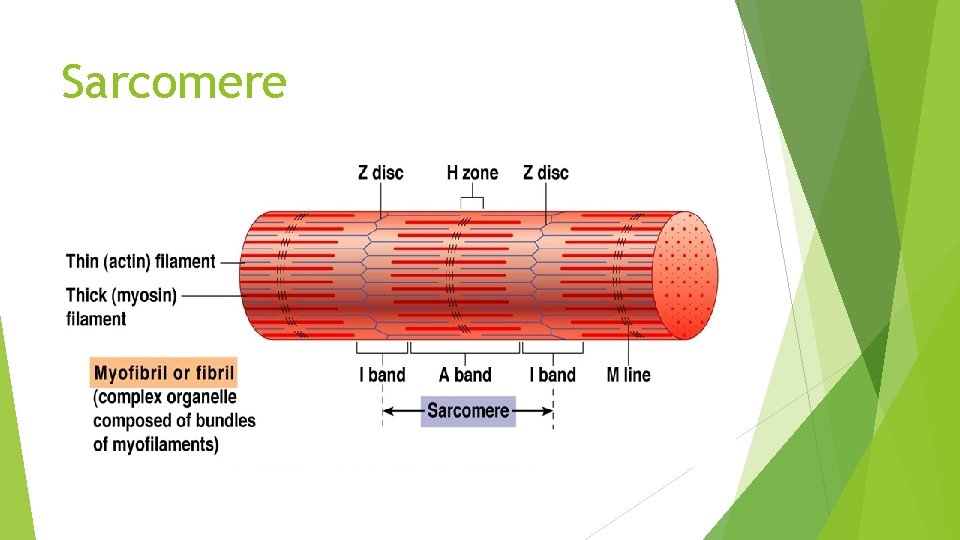
Sarcomere
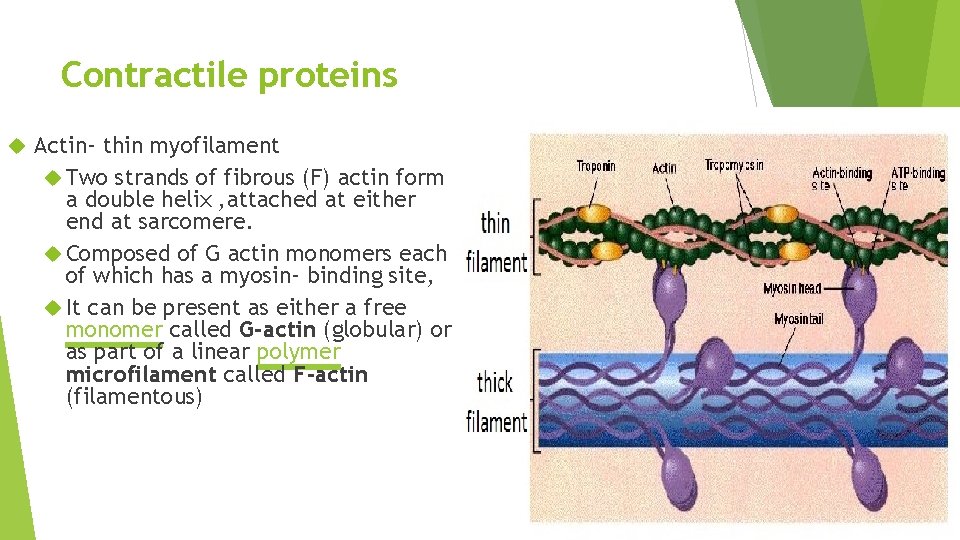
Contractile proteins Actin- thin myofilament Two strands of fibrous (F) actin form a double helix , attached at either end at sarcomere. Composed of G actin monomers each of which has a myosin- binding site, It can be present as either a free monomer called G-actin (globular) or as part of a linear polymer microfilament called F-actin (filamentous)
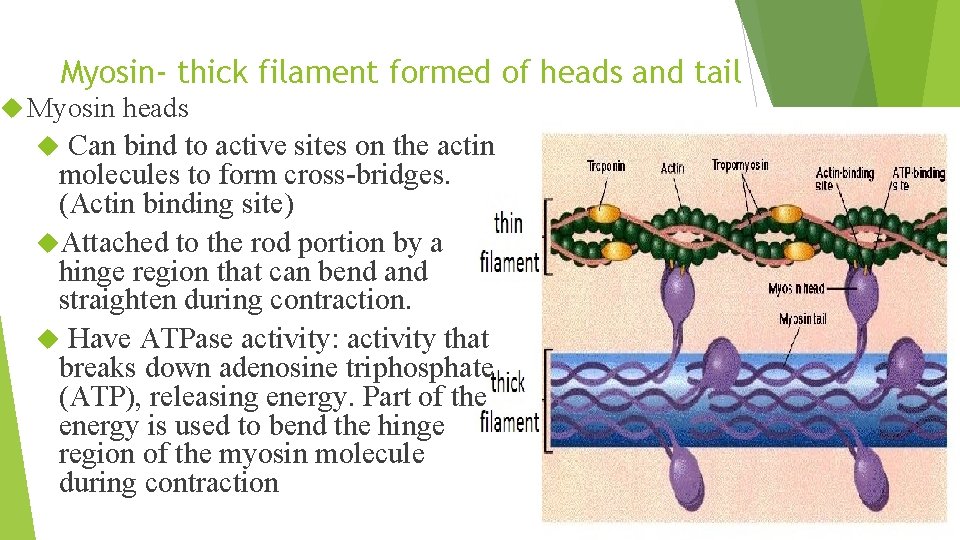
Myosin- thick filament formed of heads and tail Myosin heads Can bind to active sites on the actin molecules to form cross-bridges. (Actin binding site) Attached to the rod portion by a hinge region that can bend and straighten during contraction. Have ATPase activity: activity that breaks down adenosine triphosphate (ATP), releasing energy. Part of the energy is used to bend the hinge region of the myosin molecule during contraction
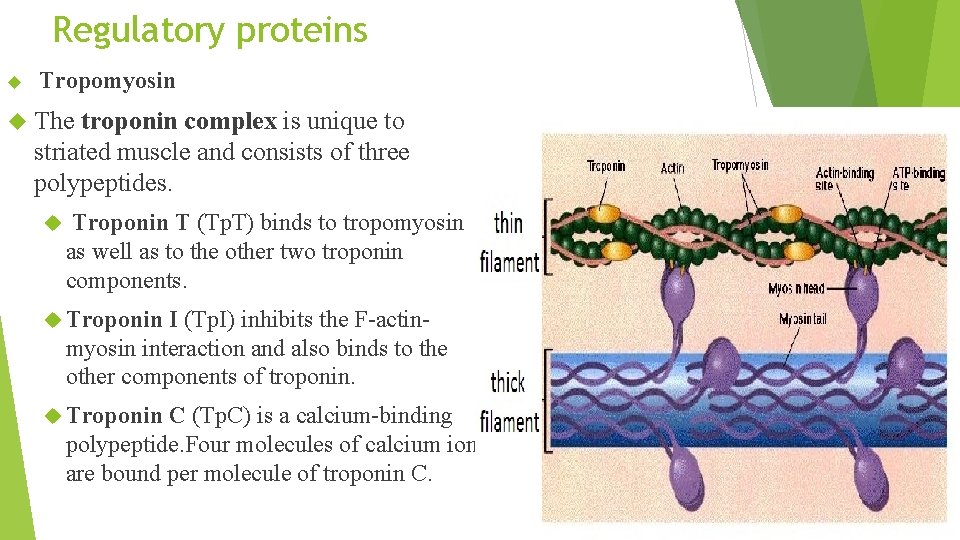
Regulatory proteins Tropomyosin The troponin complex is unique to striated muscle and consists of three polypeptides. Troponin T (Tp. T) binds to tropomyosin as well as to the other two troponin components. Troponin I (Tp. I) inhibits the F-actinmyosin interaction and also binds to the other components of troponin. Troponin C (Tp. C) is a calcium-binding polypeptide. Four molecules of calcium ion are bound per molecule of troponin C.
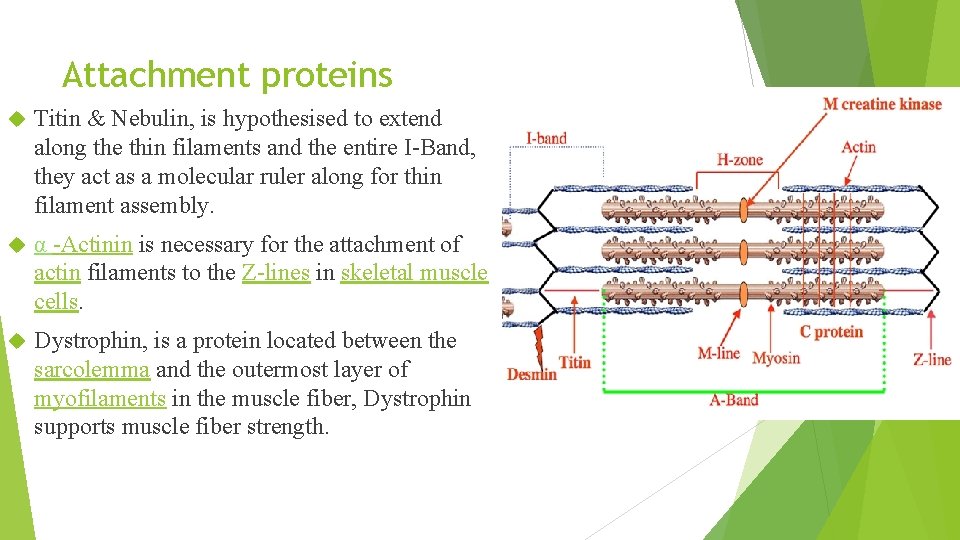
Attachment proteins Titin & Nebulin, is hypothesised to extend along the thin filaments and the entire I-Band, they act as a molecular ruler along for thin filament assembly. α -Actinin is necessary for the attachment of actin filaments to the Z-lines in skeletal muscle cells. Dystrophin, is a protein located between the sarcolemma and the outermost layer of myofilaments in the muscle fiber, Dystrophin supports muscle fiber strength.
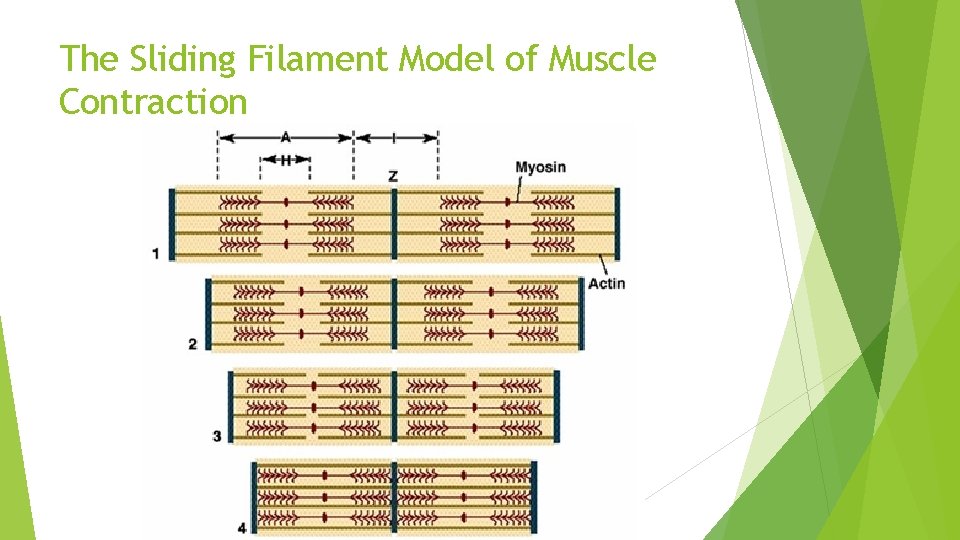
The Sliding Filament Model of Muscle Contraction
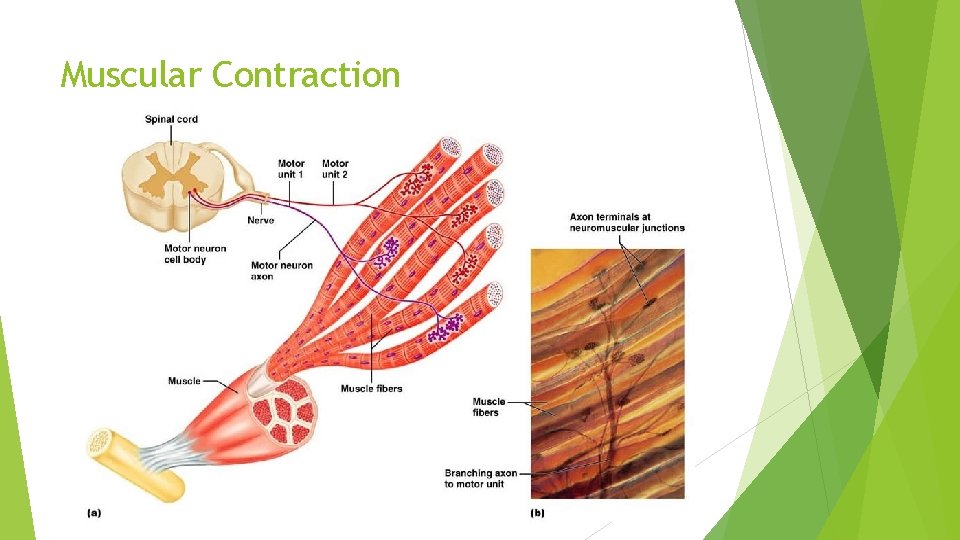
Muscular Contraction
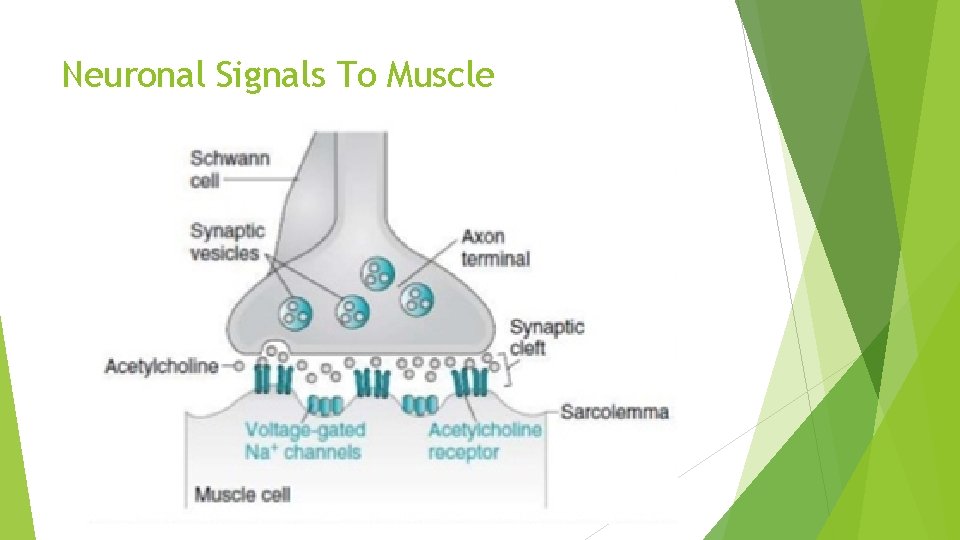
Neuronal Signals To Muscle
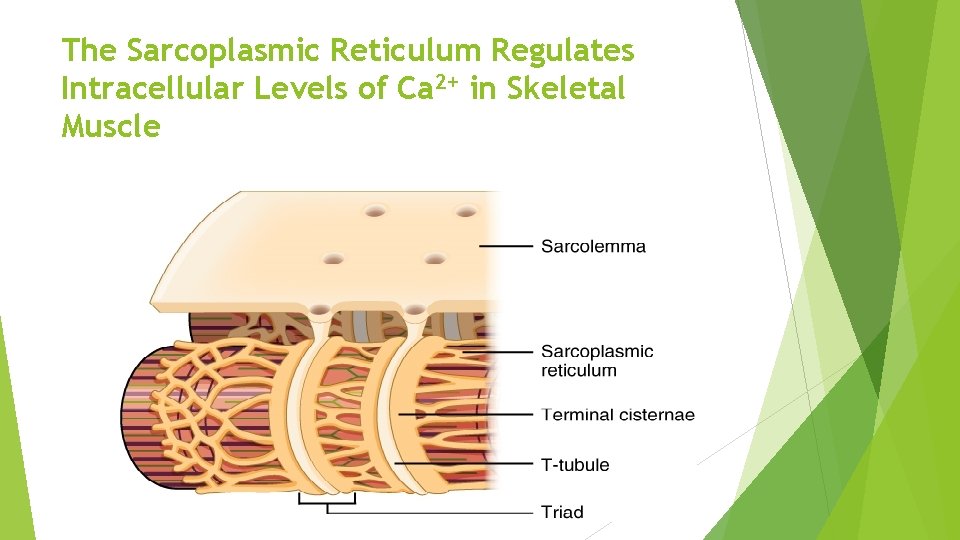
The Sarcoplasmic Reticulum Regulates Intracellular Levels of Ca 2+ in Skeletal Muscle
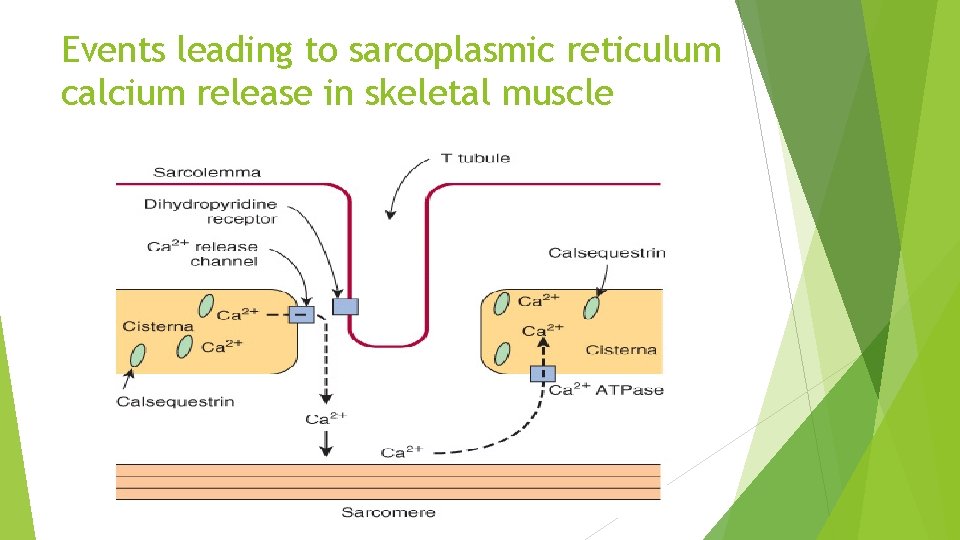
Events leading to sarcoplasmic reticulum calcium release in skeletal muscle
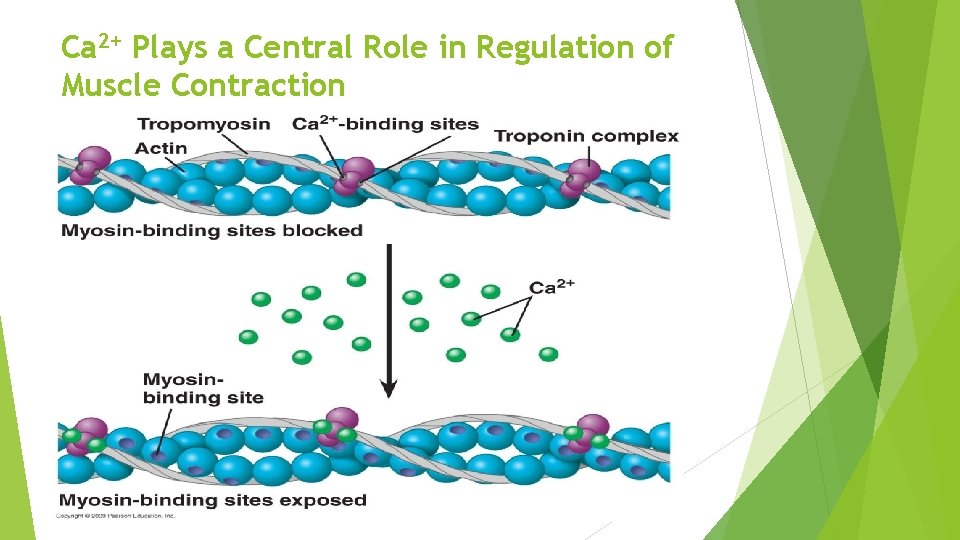
Ca 2+ Plays a Central Role in Regulation of Muscle Contraction
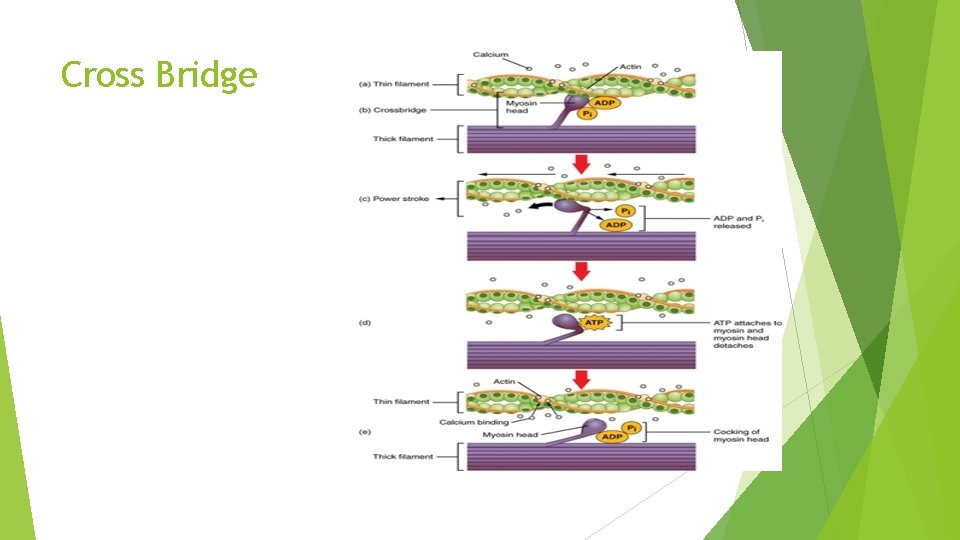
Cross Bridge
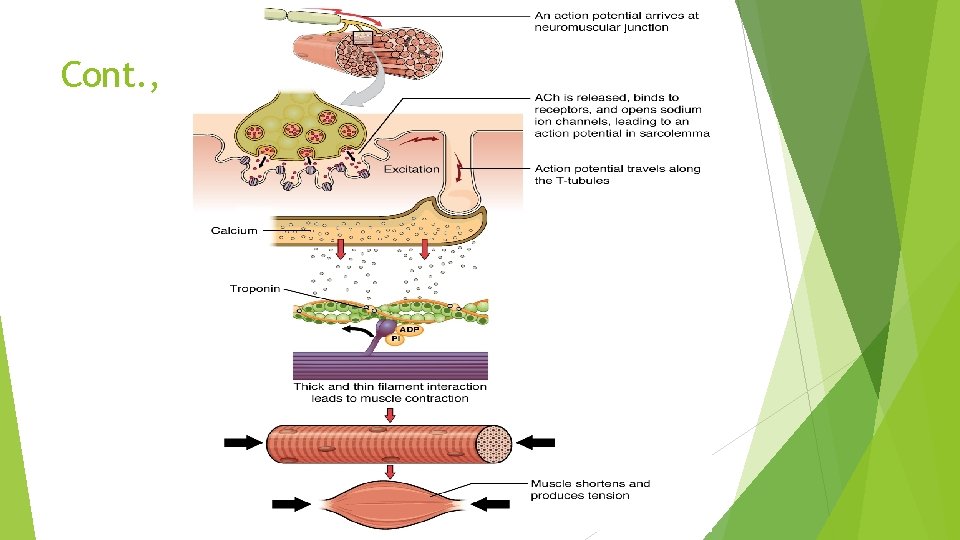
Cont. ,
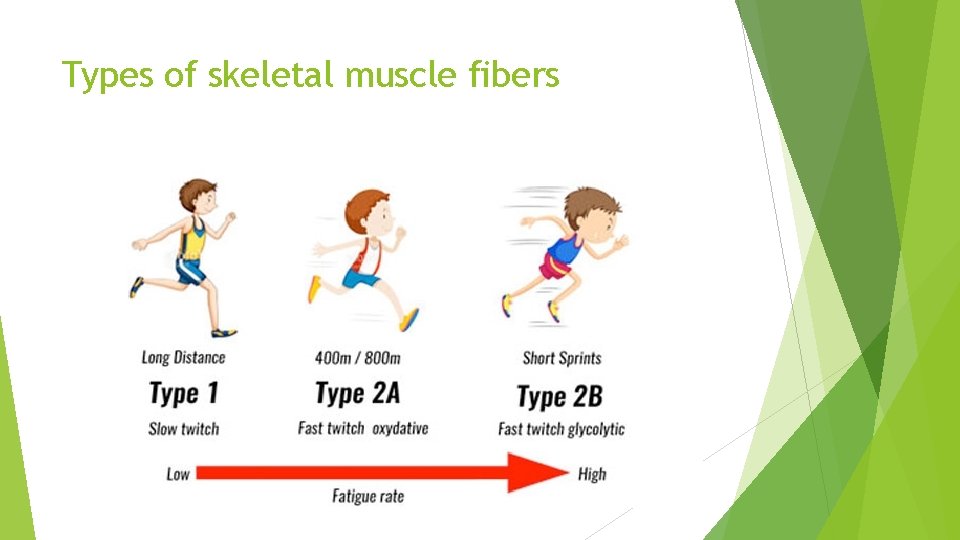
Types of skeletal muscle fibers
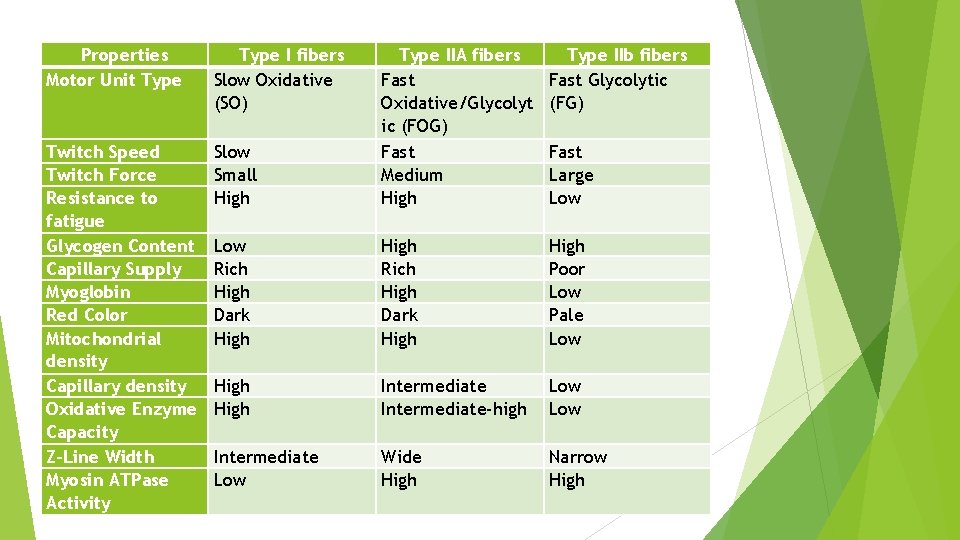
Properties Motor Unit Type I fibers Slow Oxidative (SO) Type IIb fibers Fast Glycolytic (FG) Slow Small High Type IIA fibers Fast Oxidative/Glycolyt ic (FOG) Fast Medium High Twitch Speed Twitch Force Resistance to fatigue Glycogen Content Capillary Supply Myoglobin Red Color Mitochondrial density Capillary density Oxidative Enzyme Capacity Z-Line Width Myosin ATPase Activity Low Rich High Dark High Poor Low Pale Low High Intermediate-high Low Intermediate Low Wide High Narrow High Fast Large Low

Muscle Metabolism Muscular activity accounts for much of the body's energy consumption. At rest, skeletal muscle consumes 54. 4 k. J/kg (13. 0 kcal/kg) per day. Muscles also keep a storage form of glucose in the form of glycogen. Muscle cells also contain globules of fat, which are used for energy during aerobic exercise.

Fuel Utilization at Rest During rest, most ATP required is obtained from aerobic metabolism. At almost anytime except the postprandial state free fatty acids are the preferred fuel for skeletal muscle. While glycolysis provides nearly half of the acetyl-Co. A used in the citric acid cycle in postprandial state. Branched-chain amino acid oxidation estimated to supply a maximum of 20% of the ATP supply of resting muscle.
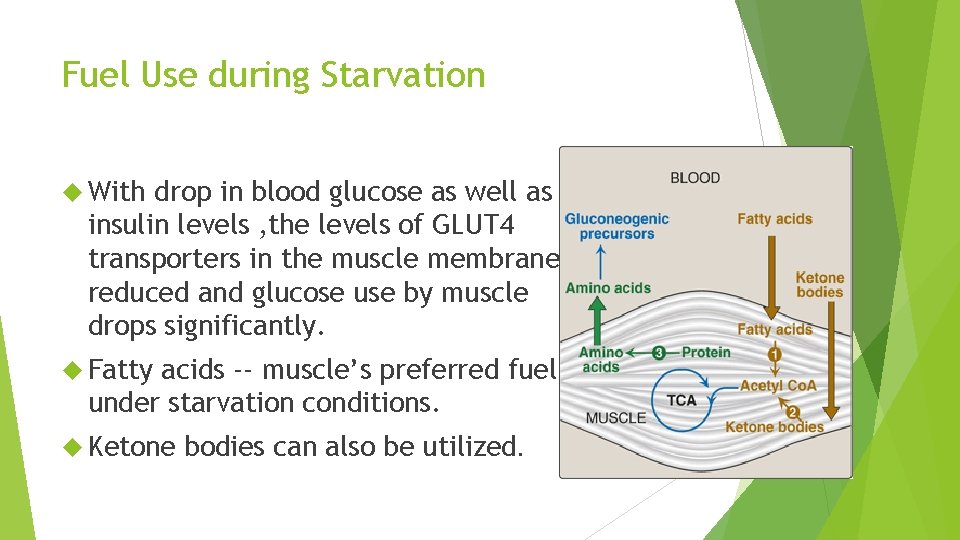
Fuel Use during Starvation With drop in blood glucose as well as insulin levels , the levels of GLUT 4 transporters in the muscle membrane reduced and glucose use by muscle drops significantly. Fatty acids -- muscle’s preferred fuel under starvation conditions. Ketone bodies can also be utilized.
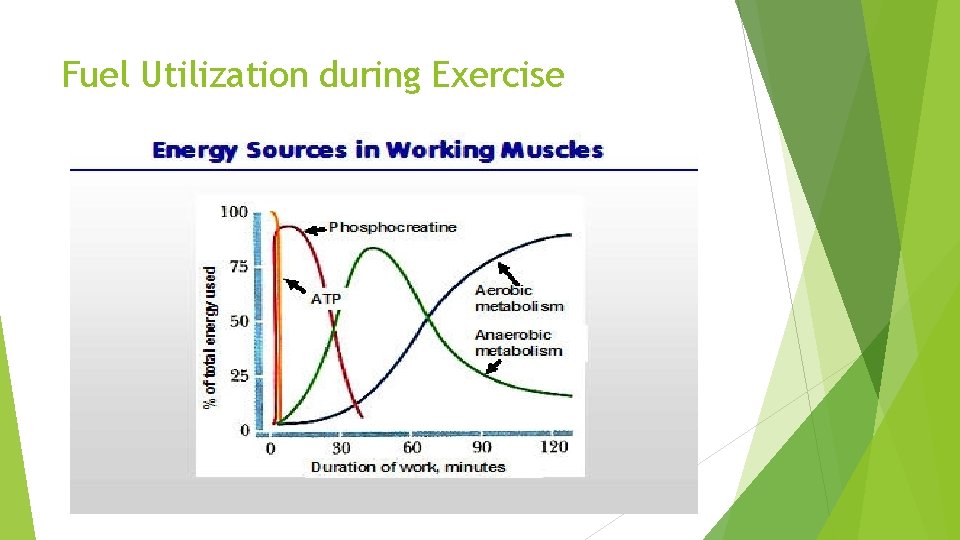
Fuel Utilization during Exercise

ATP and Creatine Phosphate ATP provides immediate energy for muscle contractions, to power the movement of the myosin heads. ATP in muscle could only work for 1 -2 seconds if not regenerated. ATP, is not a good choice as a molecule to store. Problem solved by storing high-energy phosphate bonds in the form of creatine. CP supplies are exhausted in about 20 seconds. Creatine phosphate which is generated from ATP and can regenerate ATP when needed with creatine kinase ATP also produced by Myokinase which converate 2 ADP to ATP & AMP

Anaerobic exercise Examples of anaerobic exercise include sprinting and weight lifting. Uses predominantly Type II or fast-twitch muscle fibers. Relies mainly on ATP or glucose for fuel. Consumes relatively little oxygen, and leads to: Biosynthesis of contractile & structural proteins → muscle hypertrophy. ↑ contraction force generation Produces large amounts of lactic acid and can not be sustained for long period.

Aerobic Exercise The most classic example being the marathon. Uses a higher percentage of Type I (or slowtwitch) muscle fibers. Consume a mixture of fat, protein and carbohydrates for energy, consume large amounts of oxygen and produce little lactic
Aerobic Exercise Aerobic exercises helps increase the consumption of oxygen by the body. It helps to ↑capillarization ↑ in energy metabolism Mitochondrial biogenesis Transformation Increases of fast-to-slow fiber type levels of good HDL cholesterol in blood and also reduces elevated triglycerides.
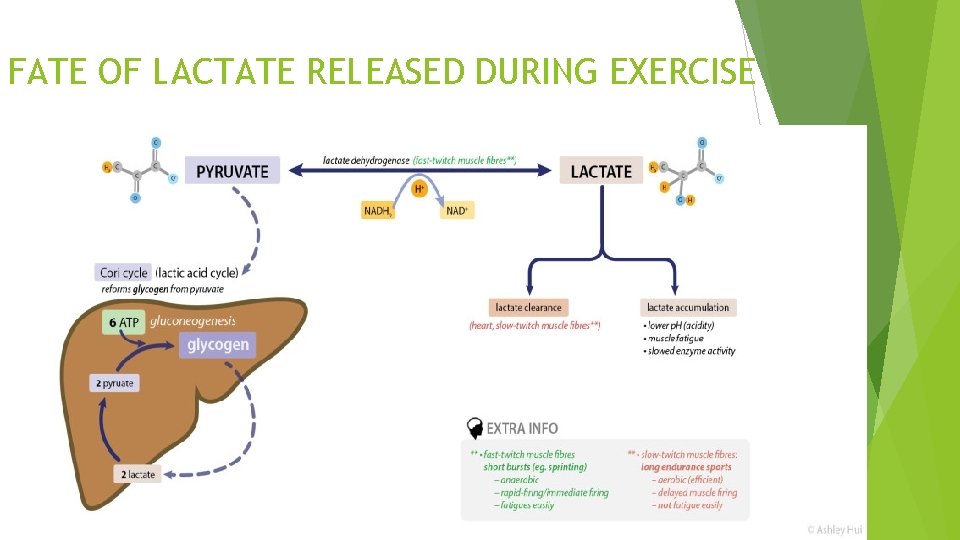
FATE OF LACTATE RELEASED DURING EXERCISE

Exersise lead to secretion of myokines
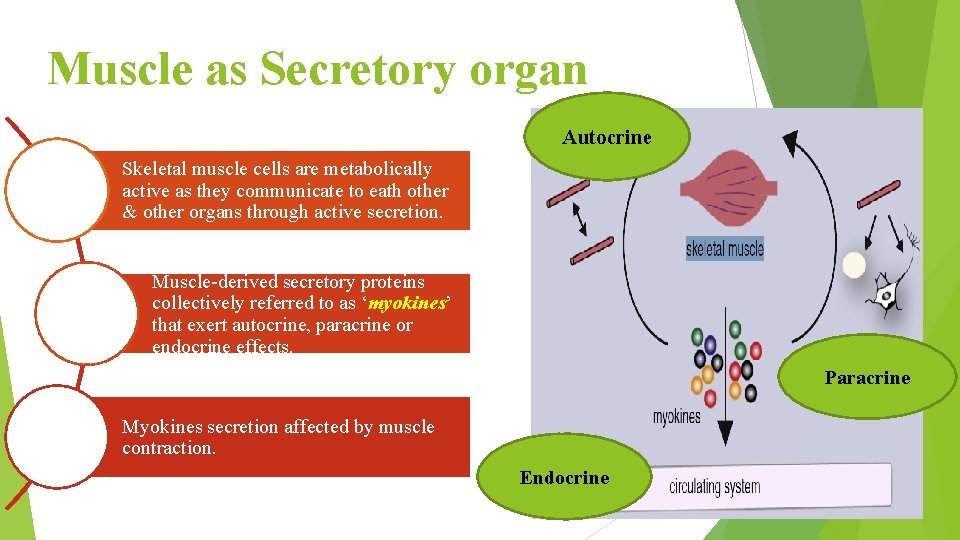
Muscle as Secretory organ Autocrine Skeletal muscle cells are metabolically active as they communicate to eath other & other organs through active secretion. Muscle-derived secretory proteins collectively referred to as ‘myokines’ that exert autocrine, paracrine or endocrine effects. Paracrine Myokines secretion affected by muscle contraction. Endocrine
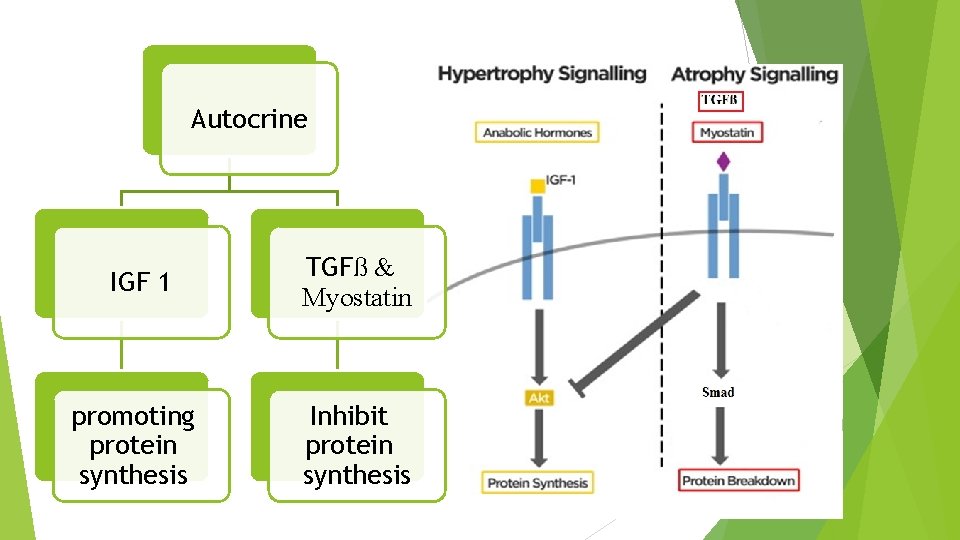
Autocrine IGF 1 promoting protein synthesis TGFß & Myostatin Inhibit protein synthesis
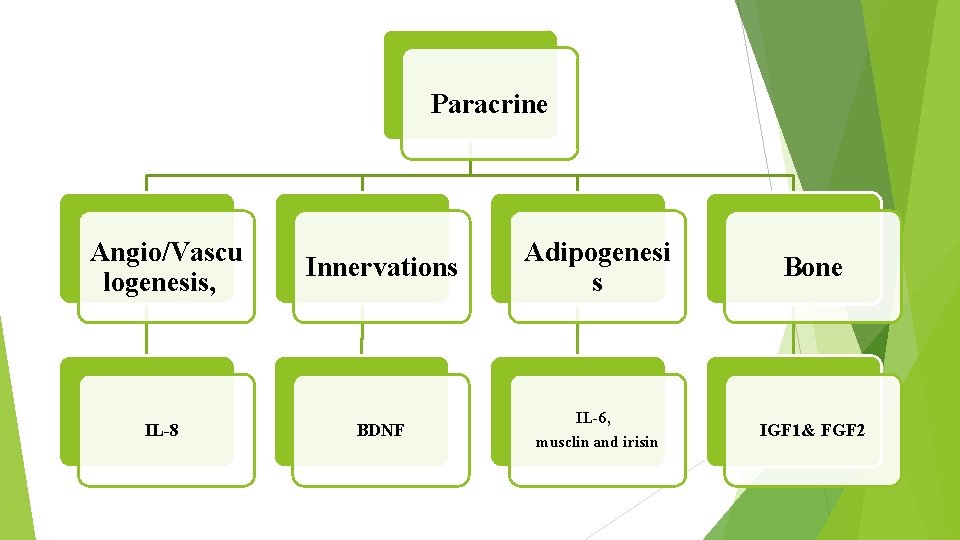
Paracrine Angio/Vascu logenesis, IL-8 Innervations Adipogenesi s Bone BDNF IL-6, musclin and irisin IGF 1& FGF 2
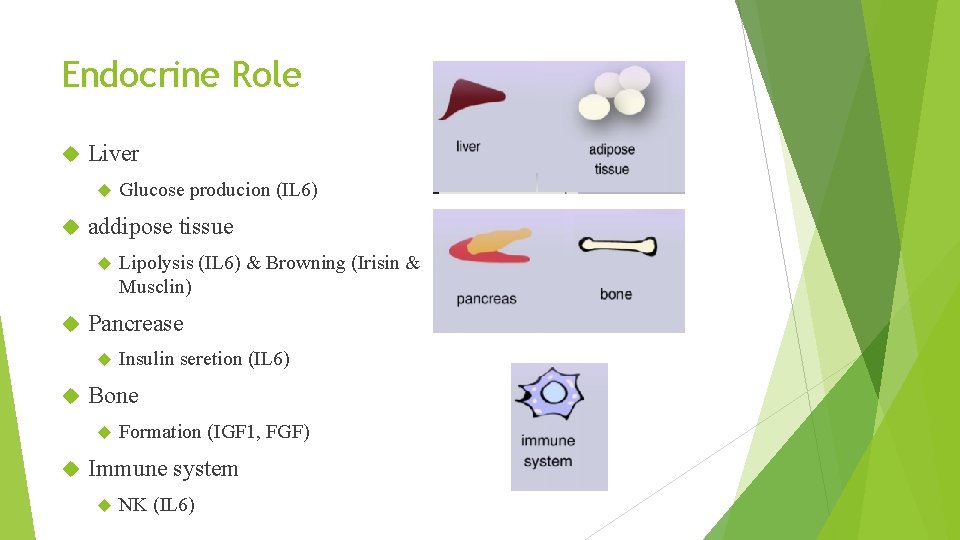
Endocrine Role Liver addipose tissue Insulin seretion (IL 6) Bone Lipolysis (IL 6) & Browning (Irisin & Musclin) Pancrease Glucose producion (IL 6) Formation (IGF 1, FGF) Immune system NK (IL 6)
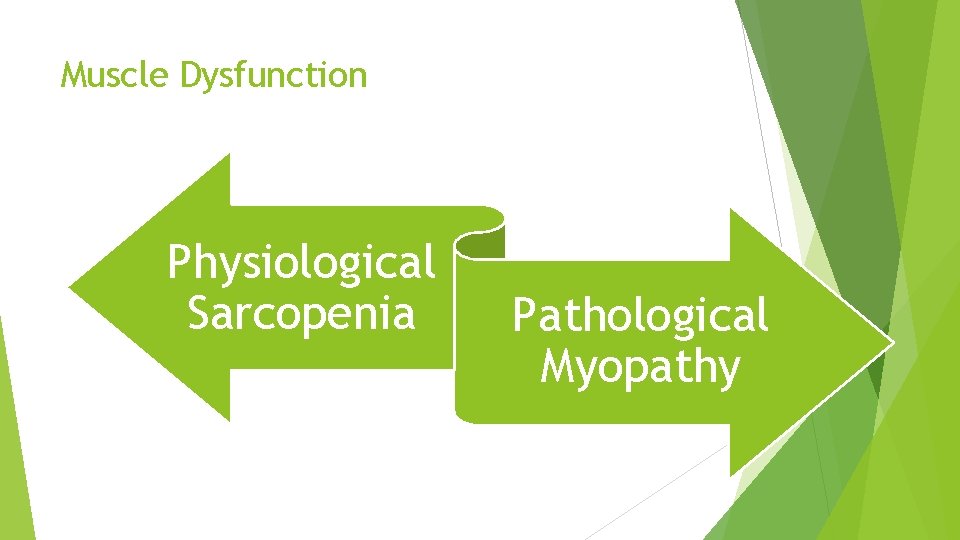
Muscle Dysfunction Physiological Sarcopenia Pathological Myopathy

Sarcopenia Slow and progressive loss of muscle mass that is associated with aging in the absence of any underlying disease or condition which directly leads Ranges from 15% to increased at 65 years to 50% hospitalizations and at 80 years. disability
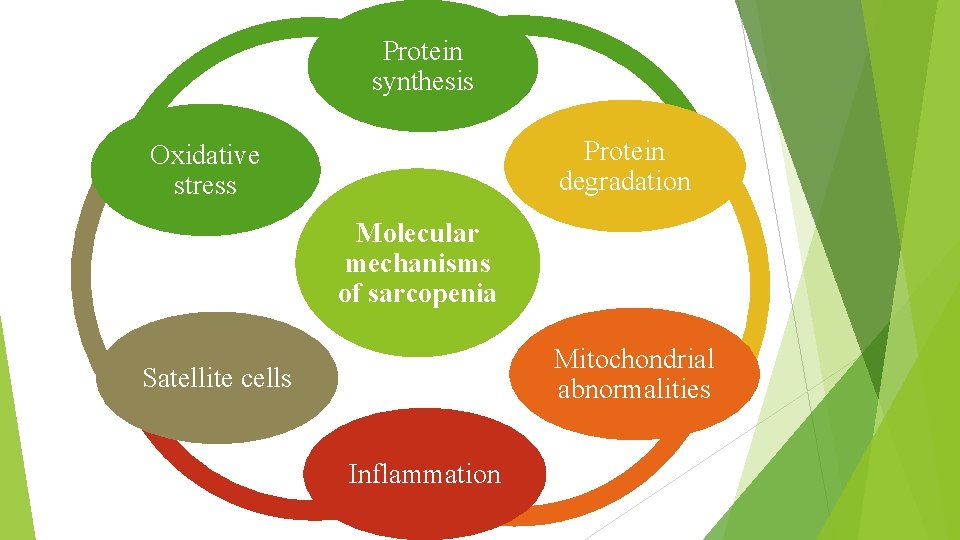
Protein synthesis Protein degradation Oxidative stress Molecular mechanisms of sarcopenia Mitochondrial abnormalities Satellite cells Inflammation
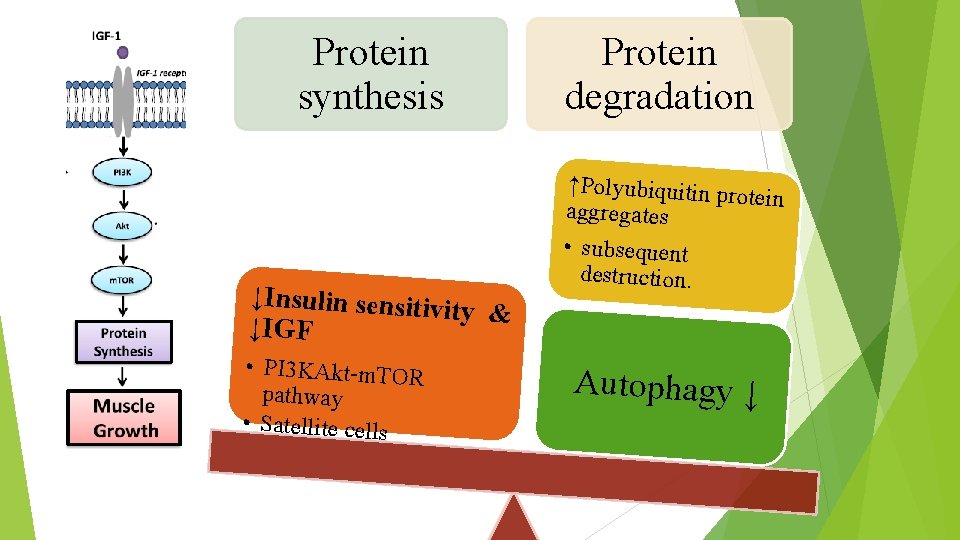
Protein synthesis ↓Insulin sensitivi ty & ↓IGF • PI 3 KAkt-m. TOR pathway • Satellite cells Protein degradation ↑Polyubiquitin pro tein aggregates • subsequent destruction. Autophagy ↓
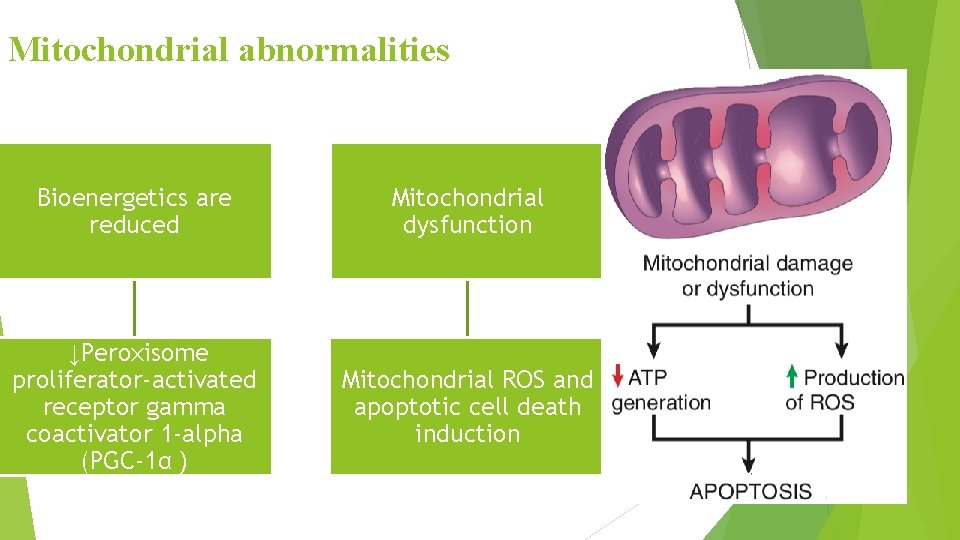
Mitochondrial abnormalities Bioenergetics are reduced Mitochondrial dysfunction ↓Peroxisome proliferator-activated receptor gamma coactivator 1 -alpha (PGC-1α ) Mitochondrial ROS and apoptotic cell death induction

Inflammation TNF-α ↓ muscle mass and strength • UPS • Impairing mitochondrial function.
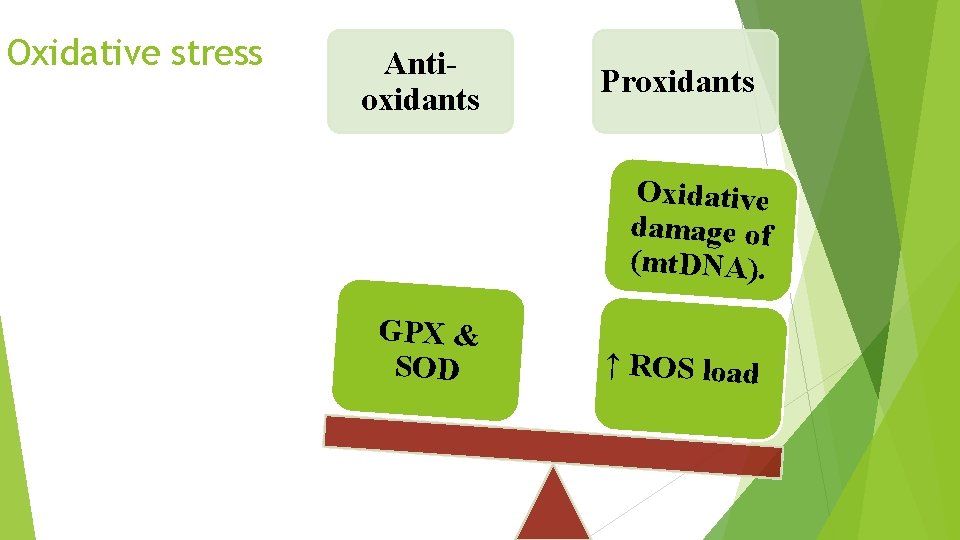
Oxidative stress Antioxidants Proxidants Oxidative damage of (mt. DNA). GPX & SOD ↑ ROS load
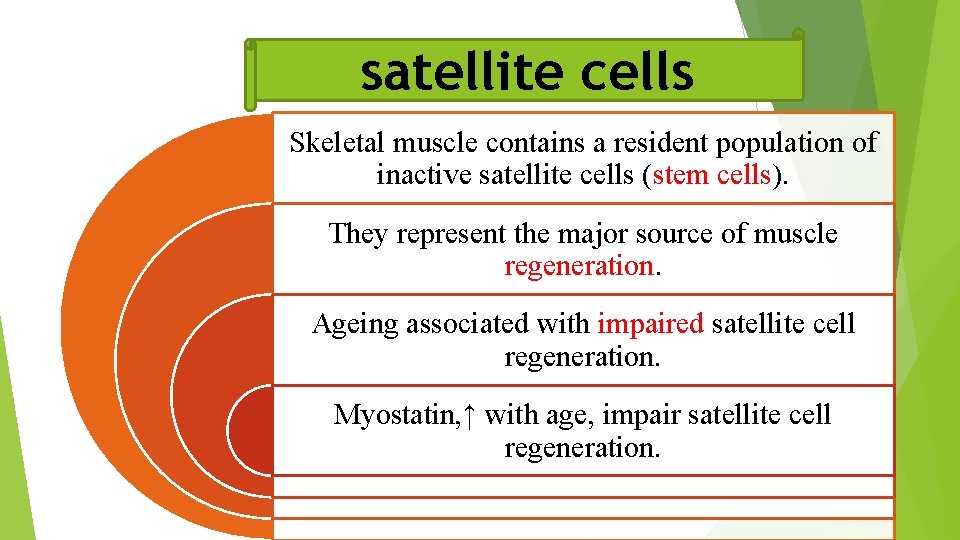
satellite cells Skeletal muscle contains a resident population of inactive satellite cells (stem cells). They represent the major source of muscle regeneration. Ageing associated with impaired satellite cell regeneration. Myostatin, ↑ with age, impair satellite cell regeneration.
Myopathies Muscular dystrophies. Skeletal muscle diseases or myopathies are disorders with structural changes or functional impairment of muscles. Metabolic myopathies. Inflammatory myopathies. Toxic myopathies. Diseases of the neuromuscular junction.

Muscular dystrophies Group of muscle diseases that results in progressive muscle weakness and wasting Duchenne muscular dystrophy Becker muscular dystrophy • absence of dystrophin • dystrophin protein is protein due to nonsense only partially functional or frame shift due to missense mutation.
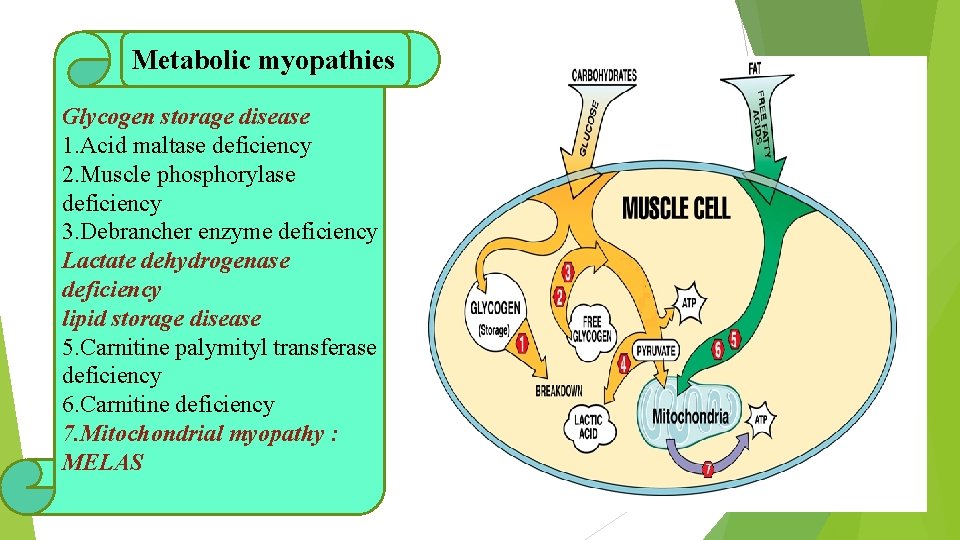
Metabolic myopathies Glycogen storage disease 1. Acid maltase deficiency 2. Muscle phosphorylase deficiency 3. Debrancher enzyme deficiency Lactate dehydrogenase deficiency lipid storage disease 5. Carnitine palymityl transferase deficiency 6. Carnitine deficiency 7. Mitochondrial myopathy : MELAS
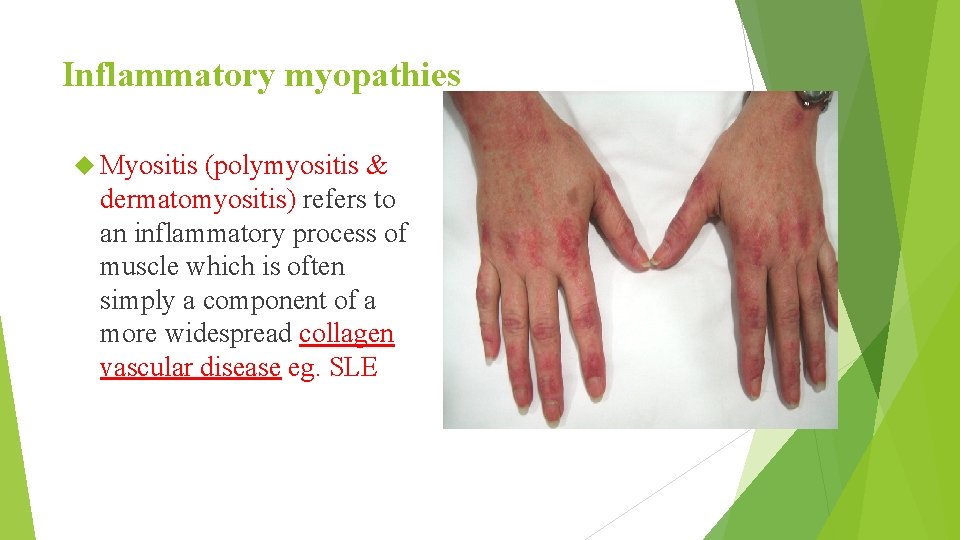
Inflammatory myopathies Myositis (polymyositis & dermatomyositis) refers to an inflammatory process of muscle which is often simply a component of a more widespread collagen vascular disease eg. SLE
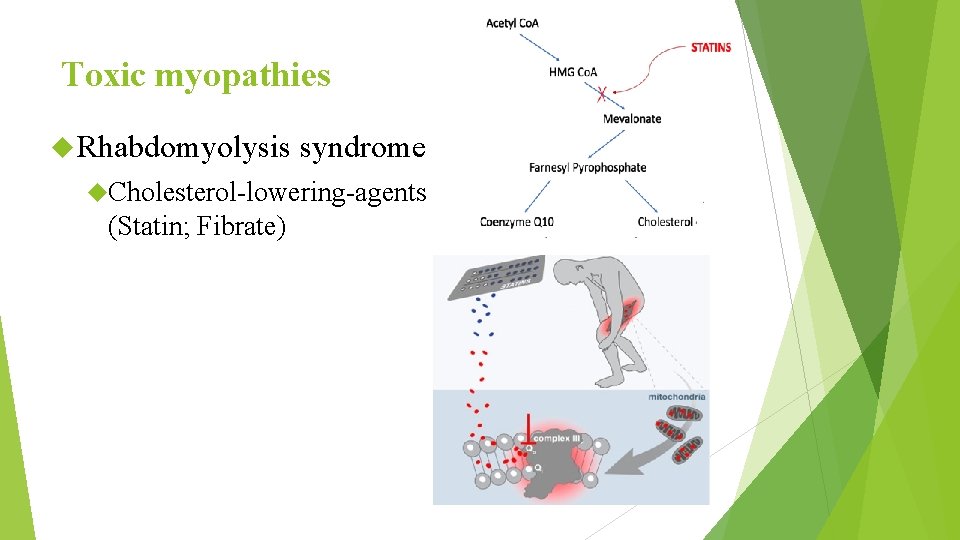
Toxic myopathies Rhabdomyolysis syndrome Cholesterol-lowering-agents (Statin; Fibrate)
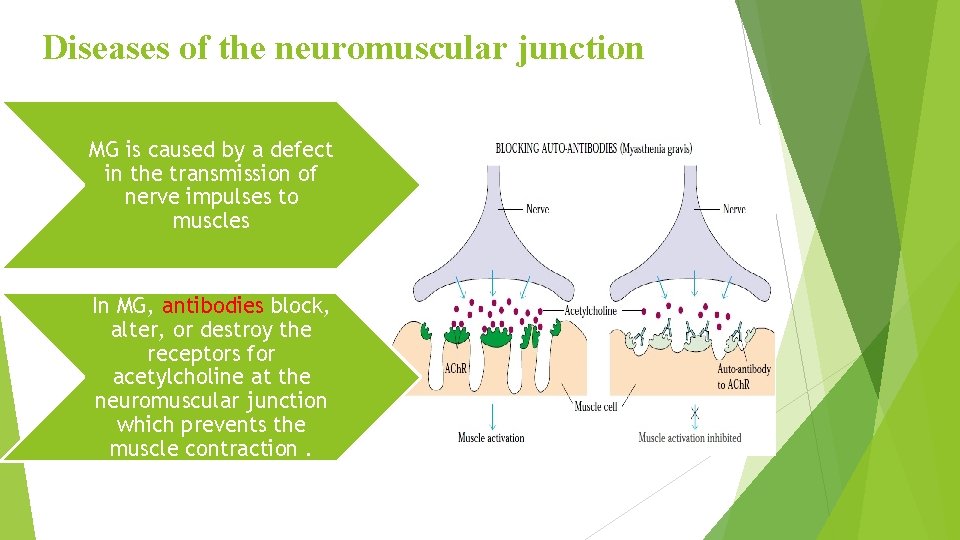
Diseases of the neuromuscular junction MG is caused by a defect in the transmission of nerve impulses to muscles In MG, antibodies block, alter, or destroy the receptors for acetylcholine at the neuromuscular junction which prevents the muscle contraction.

References Giudice, J. , & Taylor, J. M. (2017). Muscle as a paracrine and endocrine organ. Current opinion in pharmacology, 34, 49 -55. Demonbreun, A. R. , & Mc. Nally, E. M. (2017). Muscle cell communication in development and repair. Current opinion in pharmacology, 34, 7 -14. Jiao, J. , & Demontis, F. (2017). Skeletal muscle autophagy and its role in sarcopenia and organismal aging. Current opinion in pharmacology, 34, 1 -6. Bowen, T. S. , Schuler, G. , & Adams, V. (2015). Skeletal muscle wasting in cachexia and sarcopenia: molecular pathophysiology and impact of exercise training. Journal of cachexia, sarcopenia and muscle, 6(3), 197 -207.

- Slides: 49