Skeletal Formulae Written by A Bourne MChem 2014

Skeletal Formulae Written by A Bourne (MChem), 2014

Aims of this Lesson • Learn the different ways of drawing organic compounds. • Produce skeletal formulae for simple and more complex compounds • Increase your spatial skills and awareness using software designed by the CCDC

What are spatial skills? • The ability to mentally view and change 2 -dimensional and 3 -dimensional figures. • Important in chemistry, biology, medicine, mathematics, physics and everyday life. These are both the same molecule, viewed in 2 -D and 3 -D. What is the name of this simple molecule?
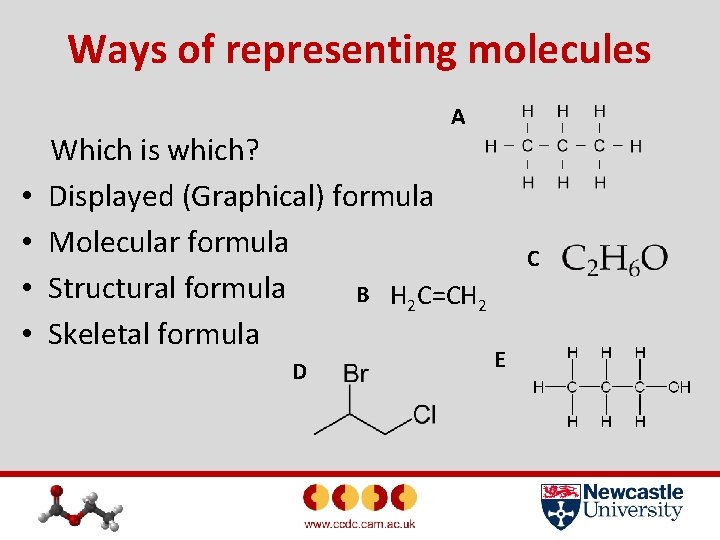
Ways of representing molecules A • • Which is which? Displayed (Graphical) formula Molecular formula Structural formula B H 2 C=CH 2 Skeletal formula D C E
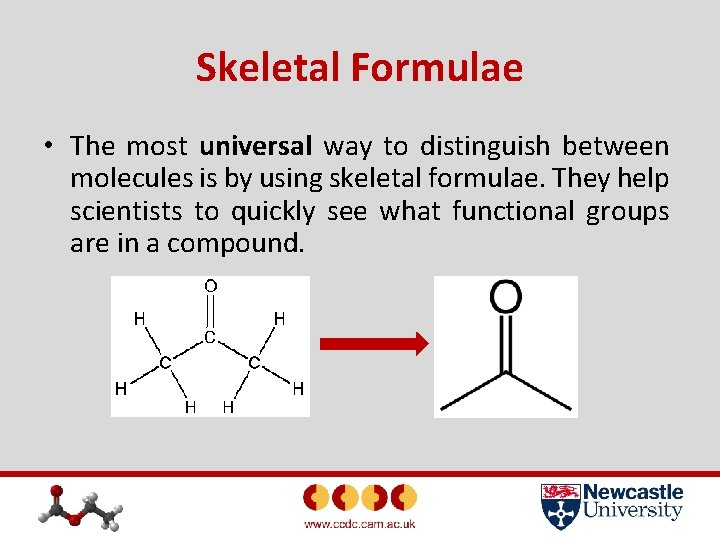
Skeletal Formulae • The most universal way to distinguish between molecules is by using skeletal formulae. They help scientists to quickly see what functional groups are in a compound.

The Rules All hydrogen atoms attached to a carbon chain are removed. Carbon atoms are also removed, represented just by lines. An end/junction/node represents another carbon atom. All other elements are shown. Some functional groups are simplified (e. g. cyanide (-C≡N) becomes -CN Remember: A carbon atom has 4 bonds. The number of hydrogens attached = 4 – bonds to other elements.
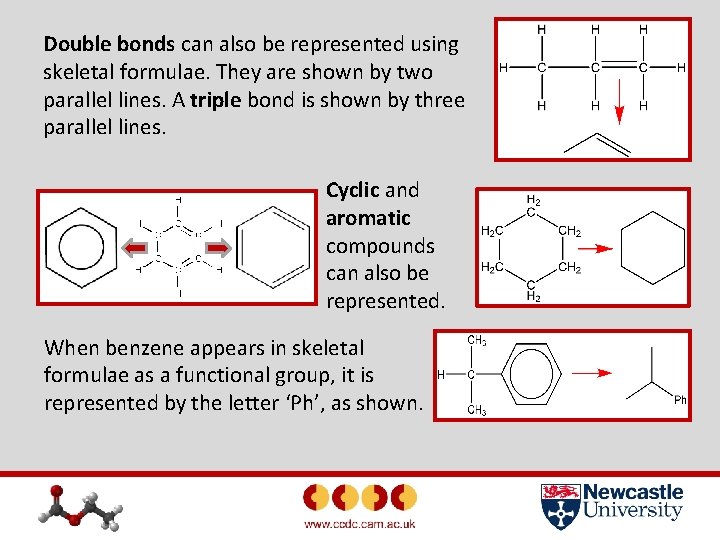
Double bonds can also be represented using skeletal formulae. They are shown by two parallel lines. A triple bond is shown by three parallel lines. Cyclic and aromatic compounds can also be represented. When benzene appears in skeletal formulae as a functional group, it is represented by the letter ‘Ph’, as shown.
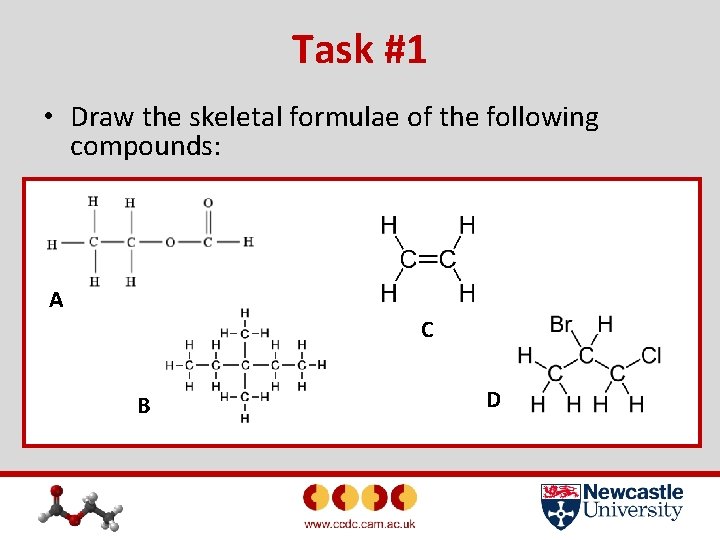
Task #1 • Draw the skeletal formulae of the following compounds: A C B D
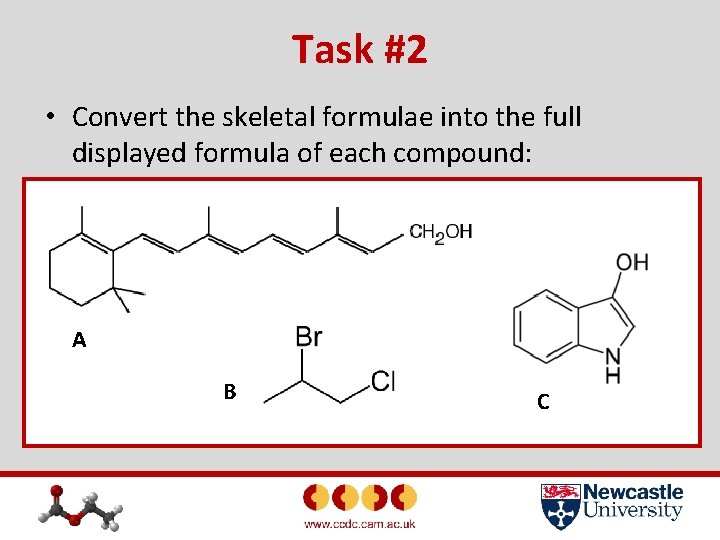
Task #2 • Convert the skeletal formulae into the full displayed formula of each compound: A B C
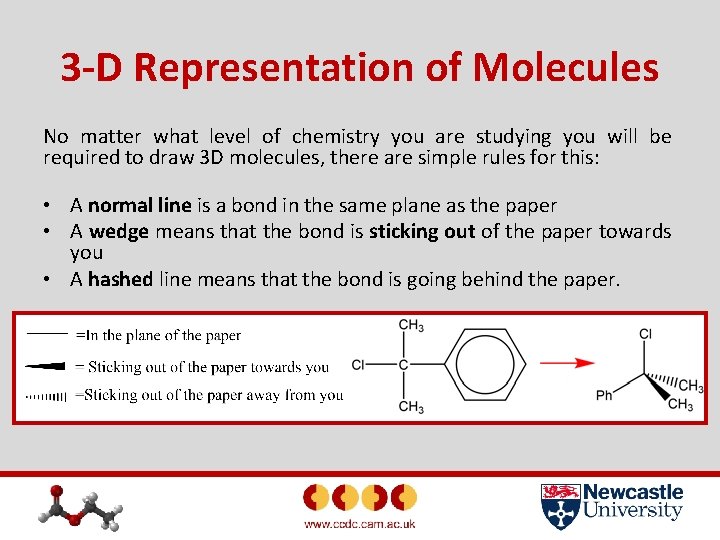
3 -D Representation of Molecules No matter what level of chemistry you are studying you will be required to draw 3 D molecules, there are simple rules for this: • A normal line is a bond in the same plane as the paper • A wedge means that the bond is sticking out of the paper towards you • A hashed line means that the bond is going behind the paper.
- Slides: 10