Single Replacement Reactions Single Replacement Reactions A BX
Single Replacement Reactions
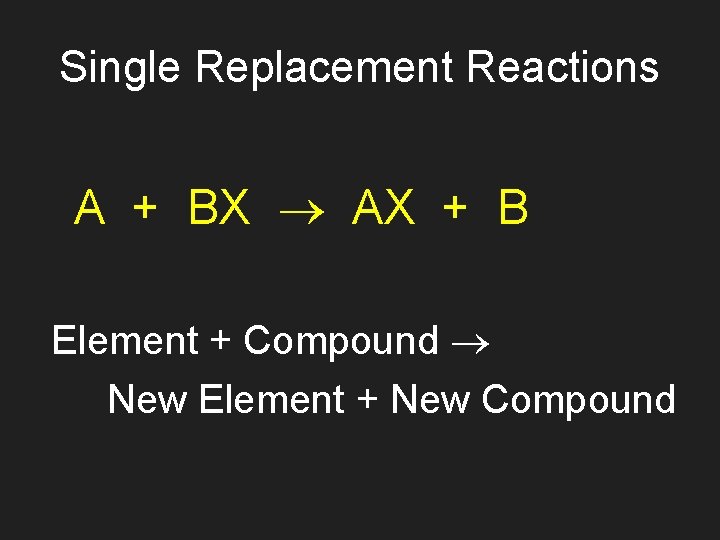
Single Replacement Reactions A + BX AX + B Element + Compound New Element + New Compound
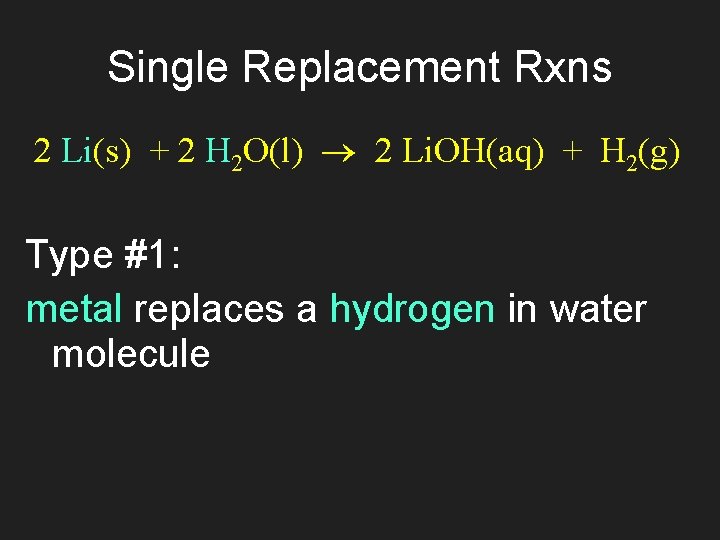
Single Replacement Rxns 2 Li(s) + 2 H 2 O(l) 2 Li. OH(aq) + H 2(g) Type #1: metal replaces a hydrogen in water molecule

Single Replacement Rxns Type #2: one metal replaces another metal in a compound Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2(aq)

Single Replacement Reactions Metals have different reactivities must predict if: given metal & given compound will react or not

Single Replacement Rxns • Table J is an activity series – more reactive a metal is, the easier it loses its valence electrons • most active metal at top • least active metal at bottom • use Table J to determine if reaction will occur

Single Replacement Rxns rule: metal CAN replace any metal listed BELOW itself on table J - CANNOT replace metal listed ABOVE itself on table J

Ag(s) + Cu(NO 3)2(aq) ? • What is the metal and the metal cation it wants to replace? Ag and Cu+2 • Locate Ag and Cu on Table J Ag below Cu so reaction will NOT go Ag is less reactive than Cu NR

Cu(s) + 2 Ag. NO 3(aq) ? • What is the metal and the metal cation? Cu and Ag+1 Locate Cu and Ag in Table J • Cu is above Ag – Cu more reactive than Ag so reaction WILL go 2 Ag(s) + Cu(NO 3)2(aq)

Single Replacement Reactions Type #3: non-metal replaces another nonmetal in compound • non-metal often is halogen • halogens have different reactivities (Table J) – most active halogen = F (top) – least active halogen = I (bottom)

Single Replacement Reactions rule: more active halogen replaces less active halogen in compound in aqueous solution F 2(g) + 2 Na. Br(aq) 2 Na. F(aq) + Br 2(l) This reaction occurs because F 2 is above Br 2

Single Replacement Reaction Br 2(g) + 2 Na. F(aq) ? Br 2 below F 2 in Table J - reaction does NOT occur NR

predict if reactions occur: • K(s) + Zn. Cl 2(aq) KCl(aq) + Zn(s) • Fe(s) + Na 3 PO 4(aq) • Cu(s) + Mg(NO 3)2(aq) • Al(s) + Sn. Cl 2(aq) • Ca(s) + KBr(aq) • Cl 2(g) + HF(aq) NR NR Al. Cl 3(aq) + Sn(s) NR NR • F 2(g) + Ca. I 2(aq) Ca. F 2(aq) + I 2(g)
- Slides: 13