Single Replacement Reactions are reactions that involve an



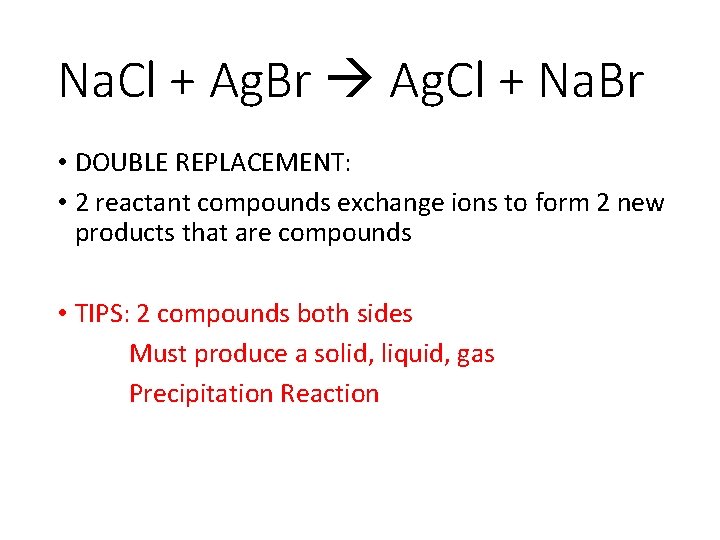






- Slides: 10

Single Replacement Reactions are reactions that involve an element replacing one part of a compound. The products include the displaced element and a new compound. The more reactive element (metal or nonmetal) will always end up as part of the compound; if the more reactive element is already in the compound, no reaction will occur.

2 KBr + Cl 2 2 KCl + Br 2 • SINGLE REPLACEMENT: • An element replacing one part of a compound forms a new element & compound • TIPS: Single element & a compound Element can be diatomic: Br. INCl. HOF

Double Replacement • Double Replacement reactions is where two reactant compounds exchange ions to form two new products that are compounds. The ions/elements from the reactants must appear in the produces. No new elements are added during the reaction.
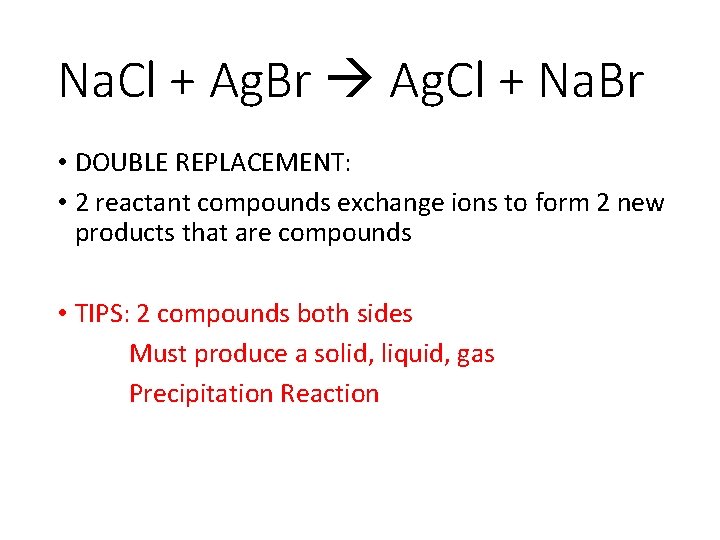
Na. Cl + Ag. Br Ag. Cl + Na. Br • DOUBLE REPLACEMENT: • 2 reactant compounds exchange ions to form 2 new products that are compounds • TIPS: 2 compounds both sides Must produce a solid, liquid, gas Precipitation Reaction

Combustion Reactions • Combustion Reactions occur when hydrocarbons or carbohydrates burn in oxygen. The products are ALWAYS water and carbon dioxide. • Oxygen = O 2

2 C 2 H 6 + 7 O 2 4 CO 2 + 6 H 2 O • COMBUSTION: • A carbohydrate or hydrocarbon is burned in air (O 2) to form carbon dioxide and water • TIPS: Only products are CO 2 & H 2 O Reactant must have O 2

Decomposition Reactions • Decomposition Reactions occur when a single reactant is broken down into two or more products, sometimes spontaneously and sometimes with the addition of heat.

2 KCl. O 3 2 KCl + 3 O 2 • DECOMPOSITION: • Single reactant is broken down into 2 or more products • TIPS: Always 1 Reactant Redox Rxn

Synthesis Reaction • Synthesis Reaction occurs when two substances combine to form a single new substance. A more complex product is always formed in synthesis rxns. This is the most common type of reaction.

H 2 + Cl 2 2 HCl • SYNTHESIS: • 2 substances combine to form 1 new substance • TIPS: Always 1 product produced Redox Rxn