Single Replacement Online Lab Double Replacement Solubility Objective

Single Replacement Online Lab Double Replacement Solubility

• Objective: – Today I will be able to: • Predict the products of single replacement and double replacement reactions • Apply the solubility rules to determining the state of a compound in a double replacement reaction • Use the activity series of metals and the periodic table to determine if a single replacement reaction will occur • Evaluation/Assessment: – Informal assessment: Listening to student interactions as they complete the lab and practice – Formal assessment: Analyzing student responses to the lab and practice problems • Common Core Connection – Use technology and digital media strategically and capably – Value Evidence – Build Strong Content Knowledge – Use appropriate tools strategically – Make sense of problem and persevere in solving them

Lesson Sequence • Evaluate: Warm – Up • Explain: Single Replacement Reaction Notes • Explore and Elaborate – Stations • Activity 1: Single Replacement Online Lab • Activity 2: Double Replacement solubility rules • Evaluate: Exit Ticket

Warm - Up • Translate, balance and identify the following reaction – Potassium phospate + hydrogen chloride potassium chloride + hydrogen phospate

Objective • Today I will be able to:

Homework • Finish Classwork Activities • Study for Translating, Balancing, Identifying Reactions Mini – Exam – B-day – Thursday, February 7 – A-day – Friday, February 8 • Wear Closed Toe Shoes Next Class!

Agenda • Warm – Up • Single Replacement predicting product notes • Stations – Activity 1: Single Replacement Online Lab – Activity 2: Double Replacement solubility rules • Exit Ticket

Single Replacement Reaction Notes Predicting Products

Review: What is a single replacement reaction? A free element reacts with a compound to form a new compound and to release one of the elements of the original compound General Equation: AB + C AC + B Examples • • • – – 2 HCl + Mg Mg. Cl 2 + H 2 2 KI + Cl 2 2 KCl + I 2

Two types of single replacement reactions Metallic Free • Free element is a metal • Most common • Example • Mg + 2 HCl Mg. Cl 2 + H 2 Non – Metallic Free • Free element is a non-metal • Example • KI + Cl 2 KCl + I 2

Free elements will not always switch places… so how can we determine when a single replacement reaction will occur?

If the single replacement reaction is metallic free: • Use the activity series of metals • Anything higher on the list as a free element will replace anything lower on the list from a compound

Examples: • 2 Na + Pb. Cl 2 2 Na. Cl + Pb • This reaction occurs because sodium is higher on the activity series than lead • Al + Na. Cl No Reaction • This reaction does not occur because aluminum is NOT higher on the activity series than sodium

If the single replacement reaction is non-metallic free: • Use the periodic table • Any free element higher in a family on the Periodic Table can replace any lower element from a compound. • Why does this work?
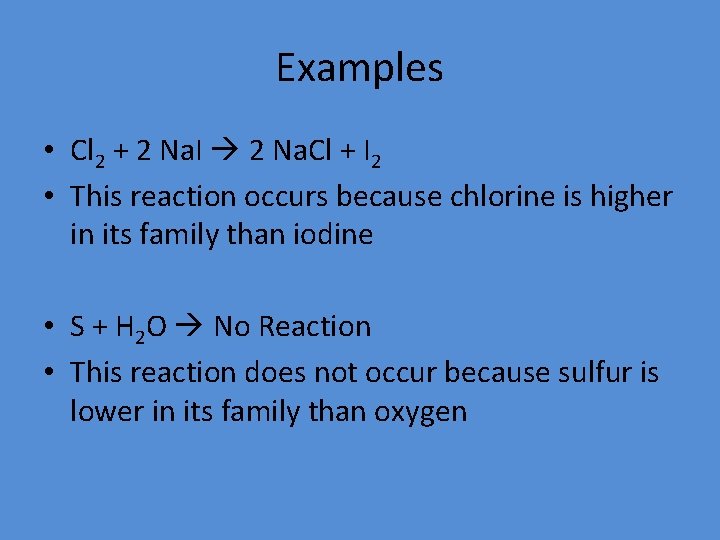
Examples • Cl 2 + 2 Na. I 2 Na. Cl + I 2 • This reaction occurs because chlorine is higher in its family than iodine • S + H 2 O No Reaction • This reaction does not occur because sulfur is lower in its family than oxygen

Activity Stations: Today you will complete 2 activities. The single replacement online lab and the solubility rules.

Station 1: Single Replacement Online Lab • Half of the class will complete first • Follow this shortened link: http: //bit. ly/pb. RDEi • The directions for the lab are at each computer. Take your data table and complete following the directions

Station 2: Double Replacement and Solubility Rules • Stay in the desk area and complete with Ms. Ose • When done, each station will flip –flop places

Double Replacement Reaction Notes Predicting Products and States

Remember Double Replacement Reactions Two ionic compounds react to form two new ionic compounds; “switching partners” General Equation: AB + CD CB + AD Examples: • • • – 3 Ca. Cl 2 + 2 Al. N 2 Al. Cl 3 + Ca 3 N 2

Double Replacement Reactions • Cations switch places with cations OR think of it as “metals switching with metals” • 3 Ca. Cl 2 + 2 Al. N 2 Al. Cl 3 + Ca 3 N 2

• In double replacement reactions: – Each compound formed must be checked to see if it forms a precipitate, liquid or gas • • Gas(g) Precipitate (s) Aqueous solution (aq) Liquid (l) – If a gas, water, or precipitate is not formed, the reaction will not take place – Use solubility rules to determine the state of the product formed

Examples • Formation of a precipitate - Ag. NO 3(aq) + Na. Cl(aq) Na. NO 3(aq) + Ag. Cl(s) • Formation of a gas - Fe. S(s) + 2 HCl(aq) 2 H 2 S(g) + Fe. Cl 2(aq) • Formation of water - HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(l)

Complete Double Replacement Reaction Predicting Product Practice

Exit Ticket • Single Replacement Online Lab: – What did you find most challenging about the online lab? Why? • Double Replacement Practice – Which problem was the most challenging? Why?
- Slides: 25