Single Chamber Solid Oxide Fuel Cells SCSOFC Brandon

Single Chamber Solid Oxide Fuel Cells (SC-SOFC) Brandon E. Bürgler Nonmetallic Inorganic Materials ETH Zürich Swiss Federal Institute of Technology Zürich M a t e r i a l s Thursday, March 19 th, 2004 Nonmetallic Materials

Outline • • Single Chamber SOFCs Experiments Modelling Issues Outlook Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
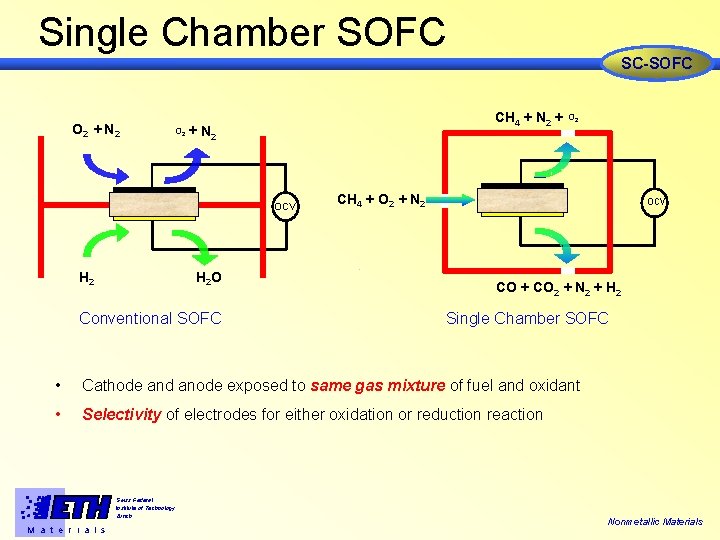
Single Chamber SOFC SC-SOFC O 2 + N 2 O 2 CH 4 + N 2 + + N 2 OCV H 2 O H 2 Conventional SOFC O 2 CH 4 + O 2 + N 2 ocv CO + CO 2 + N 2 + H 2 Single Chamber SOFC • Cathode and anode exposed to same gas mixture of fuel and oxidant • Selectivity of electrodes for either oxidation or reduction reaction Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

SC-SOFC ↔ conventional SOFC SC-SOFC Advantages • • Simplified cell sealing Elimination of complex flow field structures Fast start-up possible Costs Challenges • Non-equilibrium gas mixture (explosive from 5 to 15% CH 4 in air) • Fuel utilisation? • Parasitic chemical reactions Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
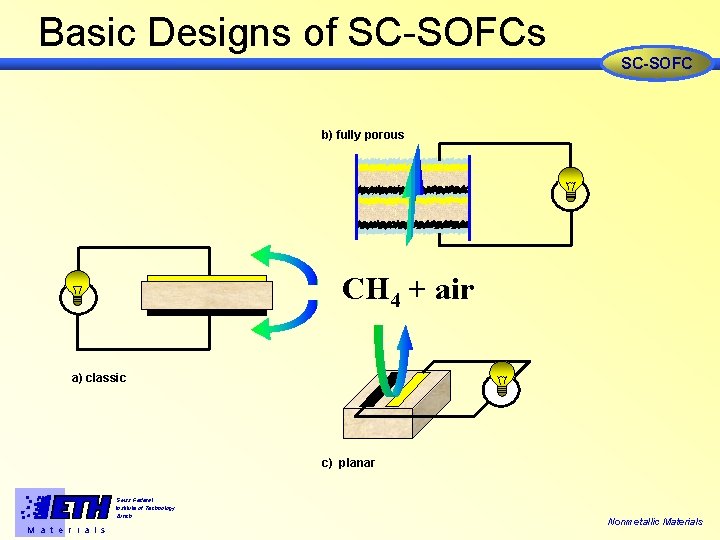
Basic Designs of SC-SOFCs SC-SOFC b) fully porous CH 4 + air a) classic c) planar Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

Open Questions & Aims SC-SOFC • Which parameters influence the OCV and the maximum power output? • Fundamental model of SC-SOFC including nonideal electrodes and CH 4 as the fuel • High performance SC-SOFC Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
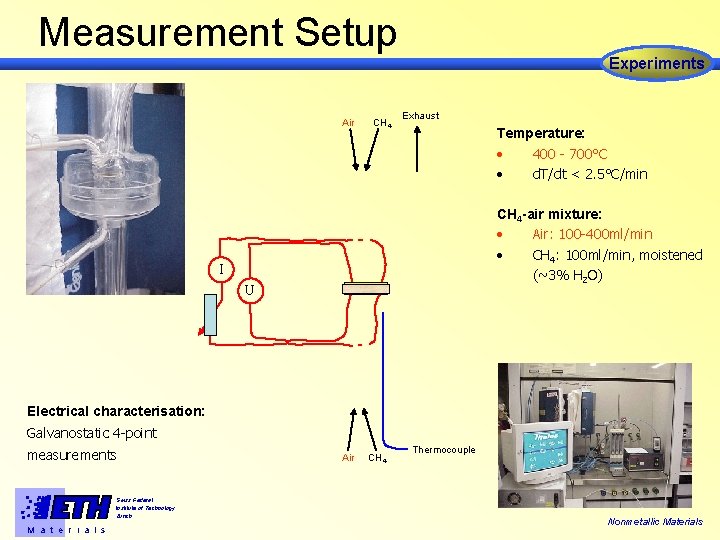
Measurement Setup Experiments Air CH 4 Exhaust Temperature: • 400 - 700°C • d. T/dt < 2. 5°C/min CH 4 -air mixture: • Air: 100 -400 ml/min • CH 4: 100 ml/min, moistened (~3% H 2 O) I U Electrical characterisation: Galvanostatic 4 -point measurements Swiss Federal Institute of Technology Zürich M a t e r i a l s Air CH 4 Thermocouple Nonmetallic Materials
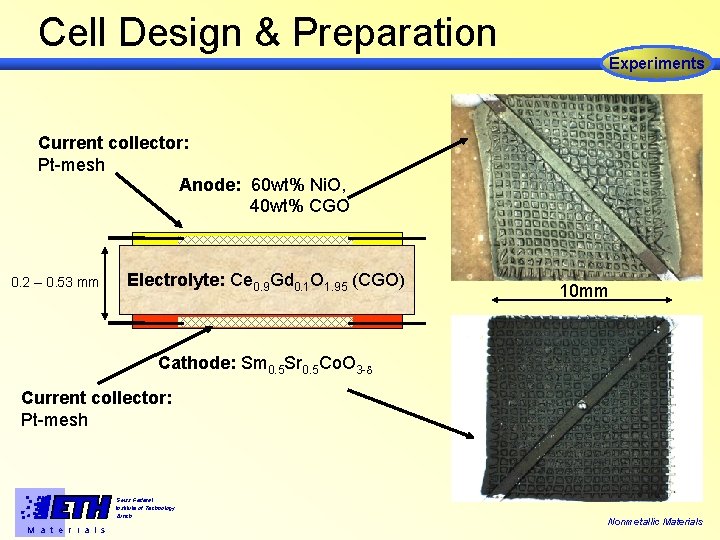
Cell Design & Preparation Experiments Current collector: Pt-mesh Anode: 60 wt% Ni. O, 40 wt% CGO 0. 2 – 0. 53 mm Electrolyte: Ce 0. 9 Gd 0. 1 O 1. 95 (CGO) 10 mm Cathode: Sm 0. 5 Sr 0. 5 Co. O 3 - Current collector: Pt-mesh Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
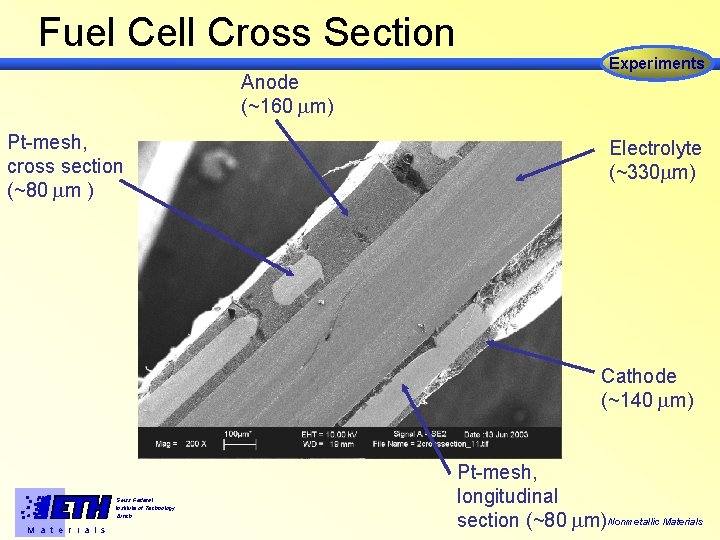
Fuel Cell Cross Section Anode (~160 m) Pt-mesh, cross section (~80 m ) Experiments Electrolyte (~330 m) Cathode (~140 m) Swiss Federal Institute of Technology Zürich M a t e r i a l s Pt-mesh, longitudinal section (~80 m) Nonmetallic Materials
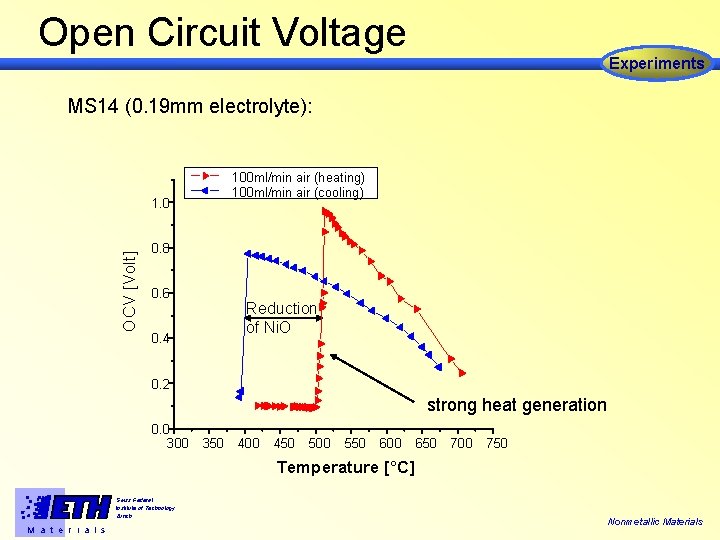
Open Circuit Voltage Experiments MS 14 (0. 19 mm electrolyte): 100 ml/min air (heating) 100 ml/min air (cooling) OCV [Volt] 1. 0 0. 8 0. 6 Reduction of Ni. O 0. 4 0. 2 strong heat generation 0. 0 300 350 400 450 500 550 600 650 700 750 Temperature [°C] Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
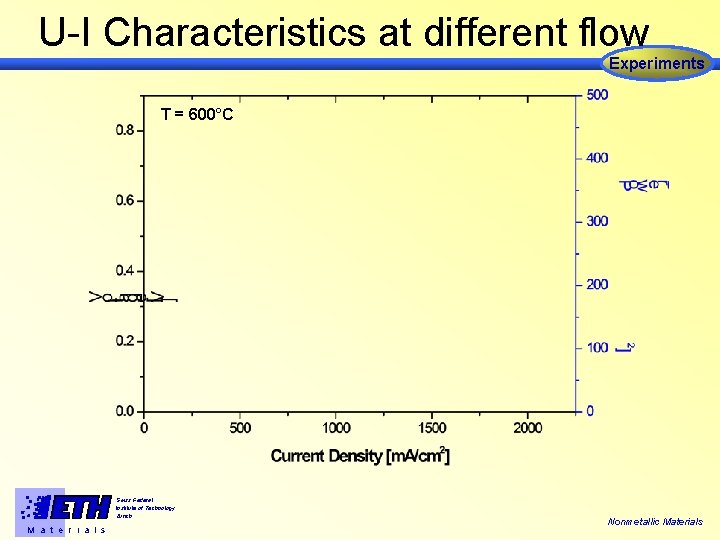
U-I Characteristics at different flow Experiments T = 600°C Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
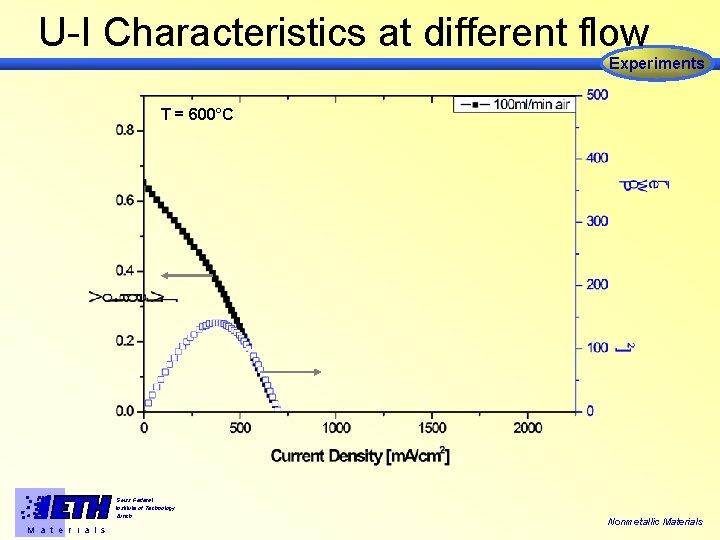
U-I Characteristics at different flow Experiments T = 600°C Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
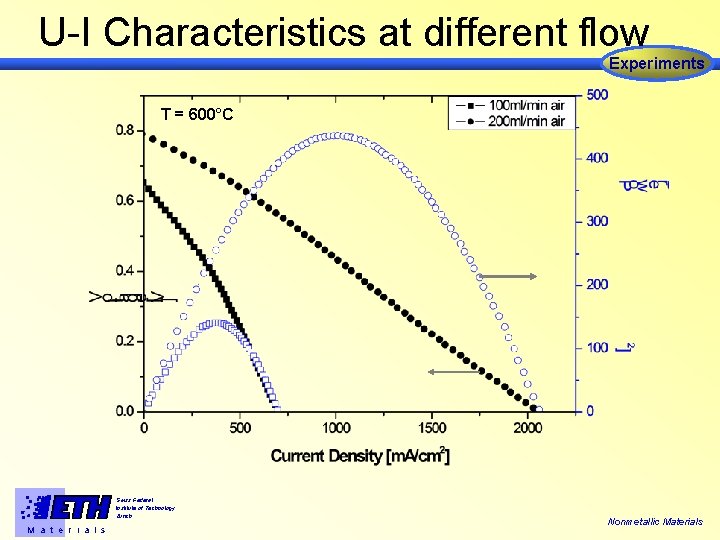
U-I Characteristics at different flow Experiments T = 600°C Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
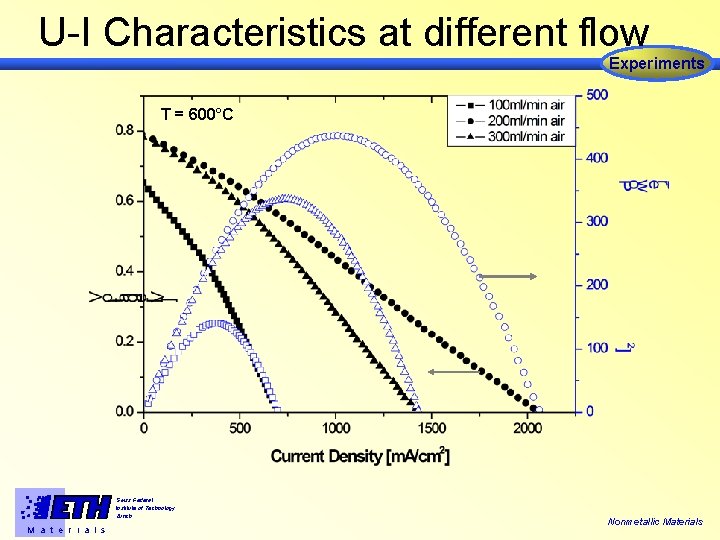
U-I Characteristics at different flow Experiments T = 600°C Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
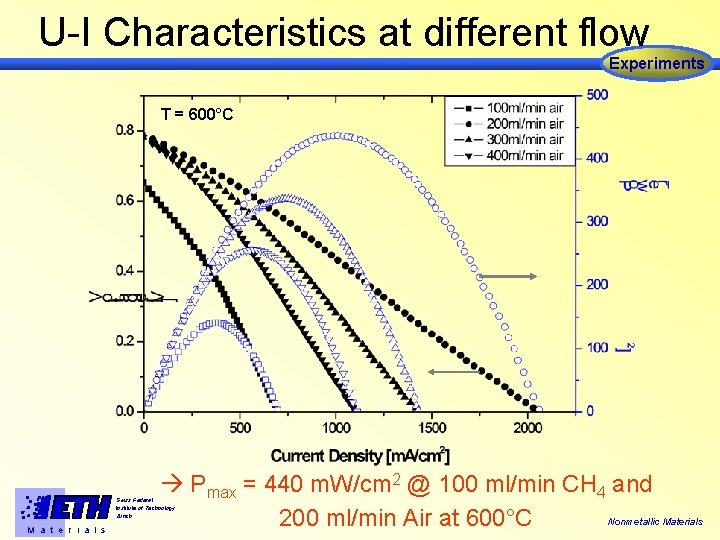
U-I Characteristics at different flow Experiments T = 600°C Pmax = 440 m. W/cm 2 @ 100 ml/min CH 4 and Nonmetallic Materials 200 ml/min Air at 600°C Swiss Federal Institute of Technology Zürich M a t e r i a l s
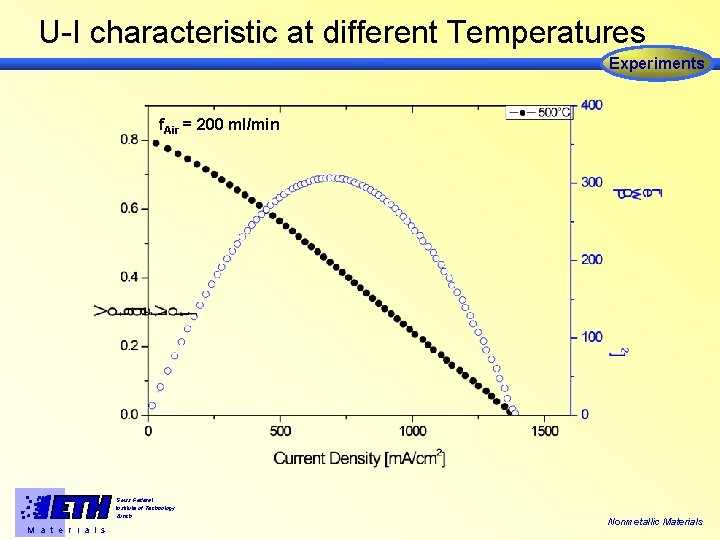
U-I characteristic at different Temperatures Experiments f. Air = 200 ml/min Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
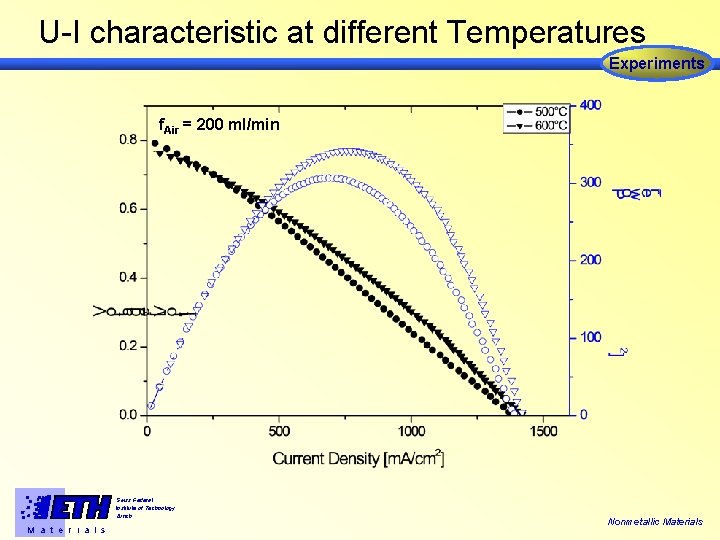
U-I characteristic at different Temperatures Experiments f. Air = 200 ml/min Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
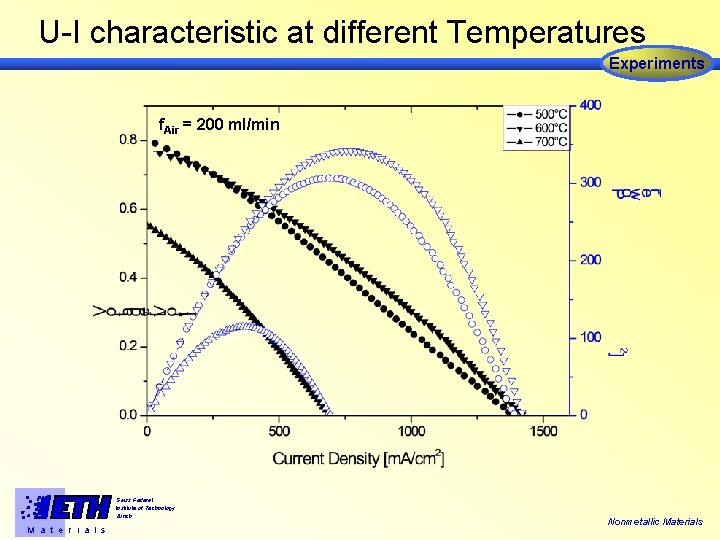
U-I characteristic at different Temperatures Experiments f. Air = 200 ml/min Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
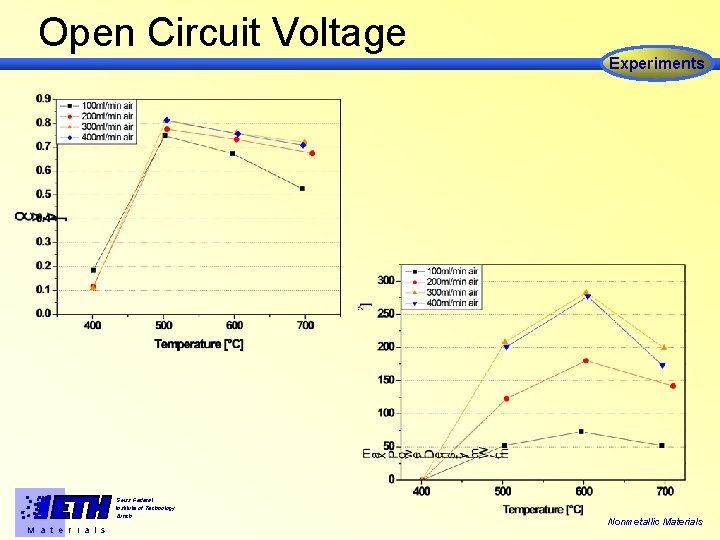
Open Circuit Voltage Experiments Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
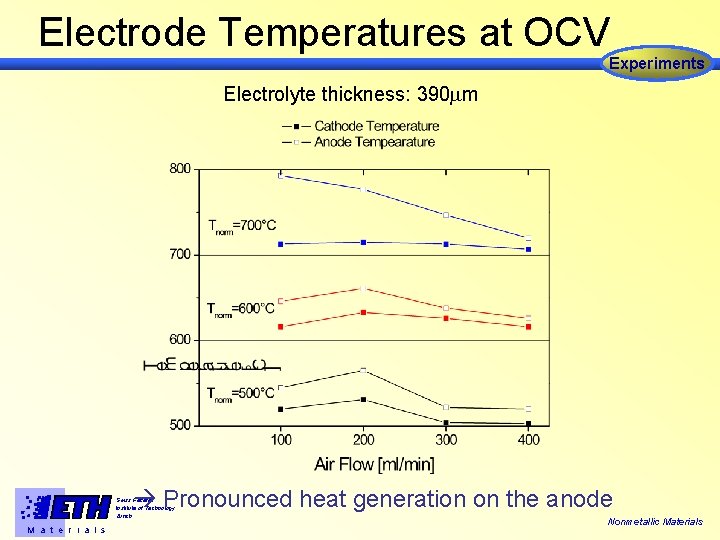
Electrode Temperatures at OCV Experiments Electrolyte thickness: 390 m Pronounced heat generation on the anode Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

Conclusions from Experiments • Cells operate at T > 500°C • Optimum conditions for maximum Power output at T= 600°C and fair = 300 ml/min • Pronounced heat generation at the anode Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

Modelling of SC-SOFCs Modelling 1. Single Chamber SOFC versus Double Chamber: Driving force for ionic current? 2. Calculations of Equilibrium gas mixtures at anode 3. Mixed ionic electronic conducting electrolyte Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

What is the driving force for the ionic current? Modelling Assumptions: -Hydrogen as fuel, air as oxidant -Reversible and perfectly selective electrodes for H 2 or O 2 -Electrolyte only O 2 --conductor Cathode Anode ½ O 2 (gas) + 2 e- → O 2 - (C) ½ H 2 (gas) → H+ (A) + 2 e. O 2 - (SE/A) + 2 H+ (A)→ H 2 O (gas) (C) (A) (SE/A) Swiss Federal Institute of Technology Zürich M a t e r i a l s Riess, I. , van der Put, P. J. (1995). "Solid oxide fuel cells operating on uniform mixtures of fuel and air. " Solid State Ionics 82: 1 -4. Nonmetallic Materials
Calculation of D O 2 - Modelling ½ O 2 (gas) + 2 e- ↔ O 2 - (C) ½ H 2 (gas) ↔ H+ (A) + e- O 2 - (SE/A) + 2 H+ (A)↔ H 2 O (gas) Combination of (7), (8) and (9) yields The Nernst Voltage is the same for SC- and conventional SOFCs Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

Comment Modelling • Electrodes are not ideally selective nor reversible. • Direct oxidation of the fuel (=parasitical) lowers OCV. • Improving selectivity of the electrodes will improve efficiency and reduce fuel waste. Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
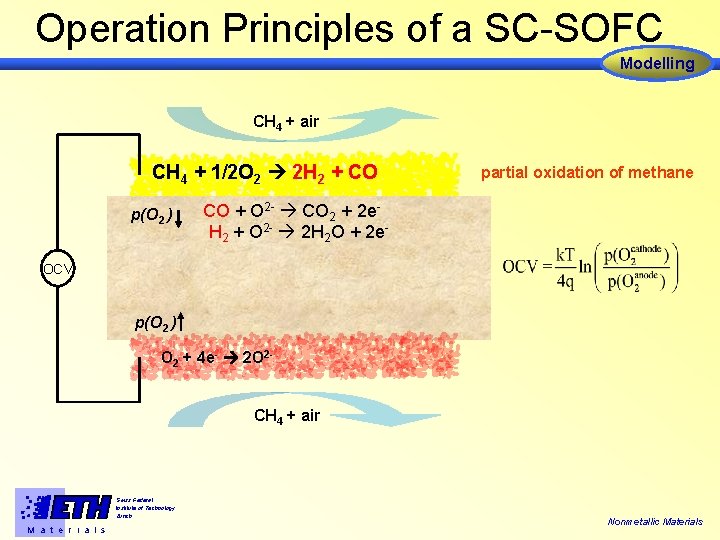
Operation Principles of a SC-SOFC Modelling CH 4 + air CH 4 + 1/2 O 2 2 H 2 + CO p(O 2 ) partial oxidation of methane CO + O 2 - CO 2 + 2 e. H 2 + O 2 - 2 H 2 O + 2 e- OCV p(O 2 ) O 2 + 4 e- 2 O 2 CH 4 + air Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

Modelling of SC-SOFCs Modelling 1. Single Chamber SOFC versus Double Chamber. Driving force for ionic current? 2. Calculations of Equilibrium gas mixtures at anode 3. Mixed ionic electronic conducting electrolyte Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

Calculation of equilibrium gas mixtures Modelling CH 4 + Air Input: T, Composition X(C), X(O), X(H) Basic idea: Anode: Equilibrium reached very fast. p. O 2 ≈ 10 -26 atm Cathode: non-equilibrium gas mixtures remains unreacted p. O 2 ≈ 0. 05 – 0. 17 atm Thermocalc™ Mi Output: nim is Fre ation e E of ne Gib rgy bs Swiss Federal Institute of Technology Zürich M a t e r i a l s Concentrations of species: CH 4, O 2, H 2, CO 2 Nonmetallic Materials
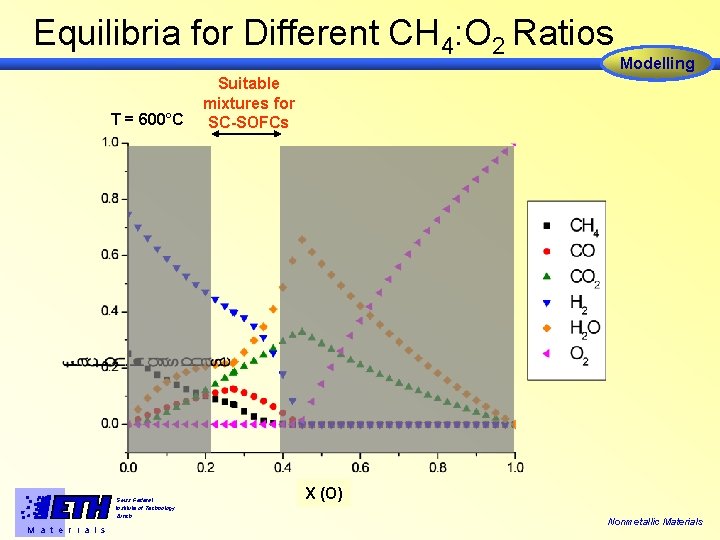
Equilibria for Different CH 4: O 2 Ratios T = 600°C Swiss Federal Institute of Technology Zürich M a t e r i a l s Modelling Suitable mixtures for SC-SOFCs X (O) Nonmetallic Materials
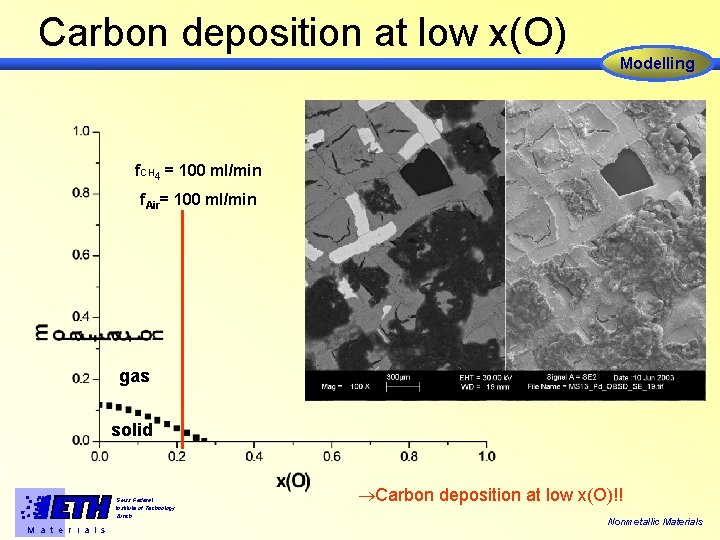
Carbon deposition at low x(O) Modelling f. CH 4 = 100 ml/min f. Air= 100 ml/min gas solid Swiss Federal Institute of Technology Zürich M a t e r i a l s Carbon deposition at low x(O)!! Nonmetallic Materials

Modelling of SC-SOFCs Modelling 1. Single Chamber SOFC versus Double Chamber. Driving force for ionic current? 2. Calculations of Equilibrium gas mixtures at anode 3. Mixed ionic electronic conducting electrolyte Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
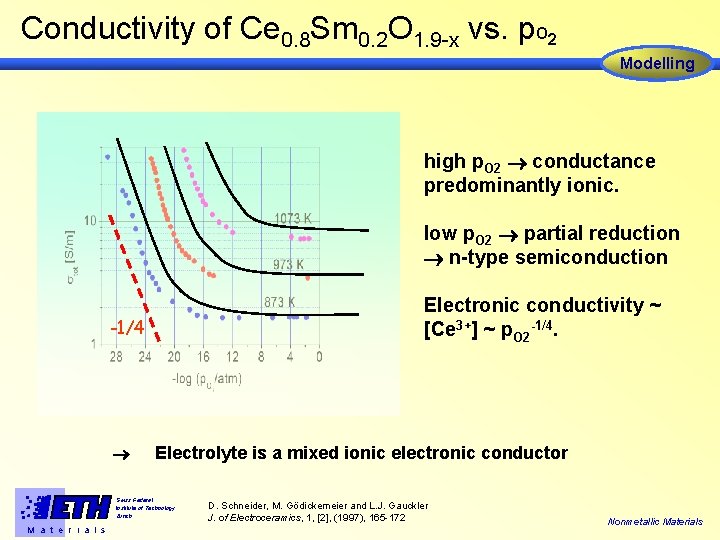
Conductivity of Ce 0. 8 Sm 0. 2 O 1. 9 -x vs. p. O 2 Modelling high p. O 2 conductance predominantly ionic. low p. O 2 partial reduction n-type semiconduction Electronic conductivity ~ [Ce 3+] ~ p. O 2 -1/4. -1/4 Electrolyte is a mixed ionic electronic conductor Swiss Federal Institute of Technology Zürich M a t e r i a l s D. Schneider, M. Gödickemeier and L. J. Gauckler J. of Electroceramics, 1, [2], (1997), 165 -172 Nonmetallic Materials
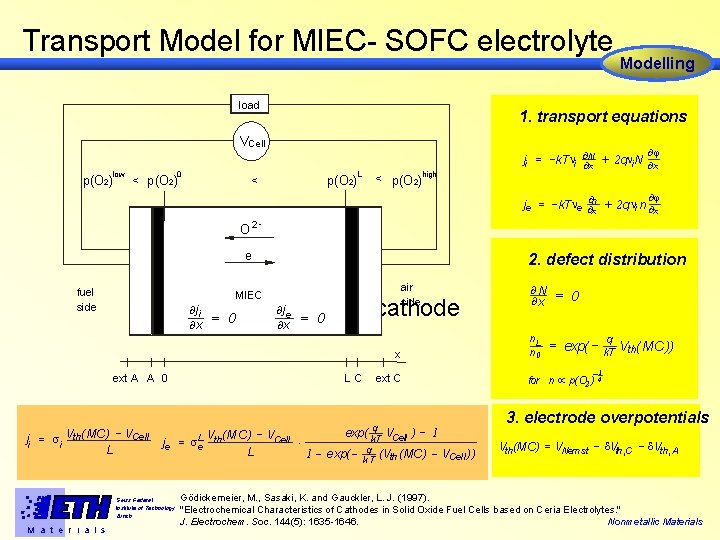
Transport Model for MIEC- SOFC electrolyte load 1. transport equations VCell low p(O 2) 0 < p(O 2) Modelling L < p(O 2)high ¶j + 2 qni. N ¶ ji = -k. Tni ¶¶N x x ¶j je = -k. Tne ¶¶nx + 2 q ni n ¶x O 2 - e fuel side anode ¶j i = ¶x 2. defect distribution air side MIEC 0 ¶j e ¶x cathode = 0 n. L n 0 x ext A A 0 ji = si Vth( MC ) VCell L V ( MC ) - VCell × je = s. Le th L 1 - Swiss Federal Institute of Technology Zürich M a t e r i a l s LC = 0 = exp( - q Vth( MC )) k. T -1 ext C q exp( k. T VCell ) q exp( - k. T (Vth (MC ) ¶N ¶x for n µ p(O 2) 1 - VCell )) 4 3. electrode overpotentials Vth(MC) = VNern st - Vth, C - Vth, A Gödickemeier, M. , Sasaki, K. and Gauckler, L. J. (1997). "Electrochemical Characteristics of Cathodes in Solid Oxide Fuel Cells based on Ceria Electrolytes. " J. Electrochem. Soc. 144(5): 1635 -1646. Nonmetallic Materials
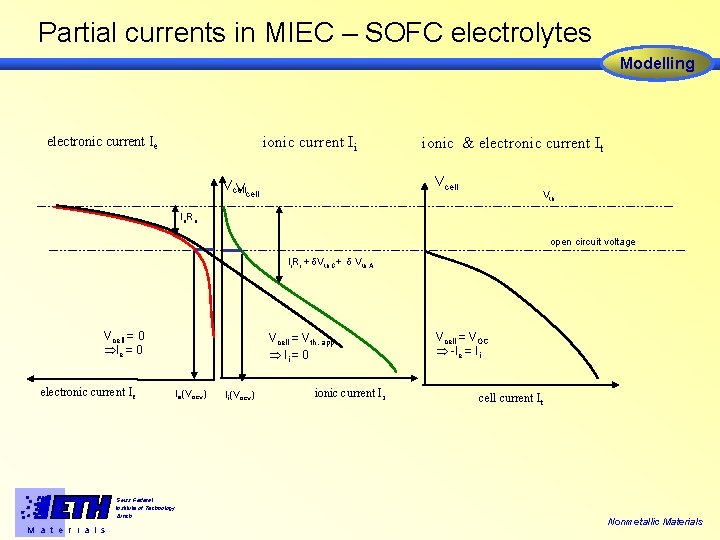
Partial currents in MIEC – SOFC electrolytes Modelling ionic current Ii electronic current Ie ionic & electronic current It Vcell Vth Ie. Re open circuit voltage Ii. Ri + Vth, C+ Vth, A Vcell = 0 Ie = 0 electronic current Ie Vcell = Vth, app Ii = 0 Ie(Vocv) Swiss Federal Institute of Technology Zürich M a t e r i a l s Ii(Vocv) ionic current Ii Vcell = VOC -Ie = Ii cell current I t Nonmetallic Materials

Conclusions Modelling 1. Single Chamber SOFC versus Double ü Chamber. Driving force for ionic current? 2. Calculations of Equilibrium gas mixtures at anode ü 3. Mixed ionic electronic conducting electrolyte ü 4. Thermal Reactor ---- Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

Acknowledgements • • • Prof. Dr. L. J. Gauckler A. Nicholas Grundy Michel Prestat SOFC group The entire Nonmets Group Diploma students • Marco Siegrist • Srdan Vasic Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials

Thank you for your kind attention Swiss Federal Institute of Technology Zürich M a t e r i a l s Nonmetallic Materials
- Slides: 37