Signal Transduction Biochemistry February 23 2005 Chapter 12

Signal Transduction Biochemistry – February 23, 2005 Chapter 12 – parts 12. 3, 12. 4
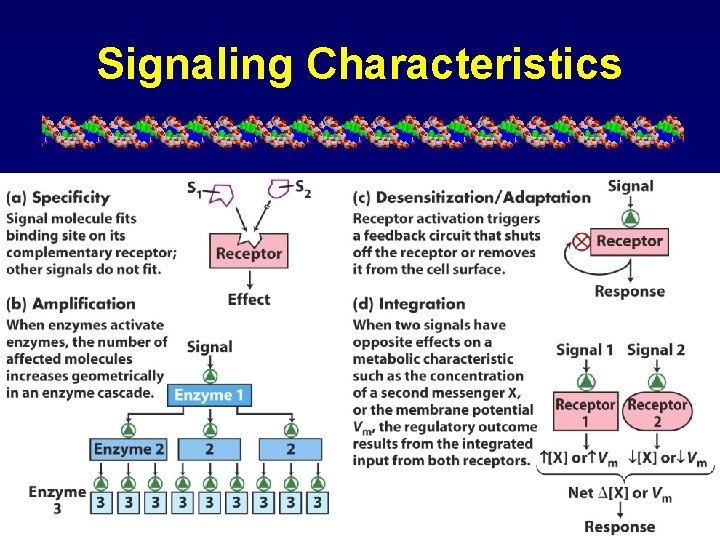
Signaling Characteristics
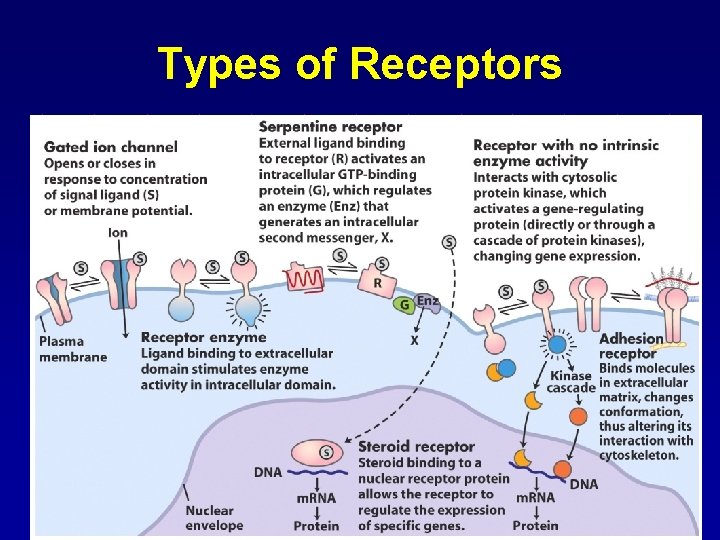
Types of Receptors
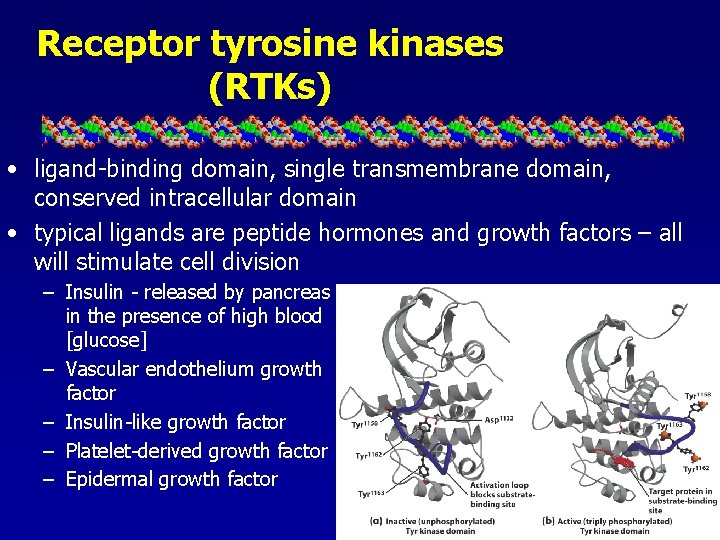
Receptor tyrosine kinases (RTKs) • ligand-binding domain, single transmembrane domain, conserved intracellular domain • typical ligands are peptide hormones and growth factors – all will stimulate cell division – Insulin - released by pancreas in the presence of high blood [glucose] – Vascular endothelium growth factor – Insulin-like growth factor – Platelet-derived growth factor – Epidermal growth factor
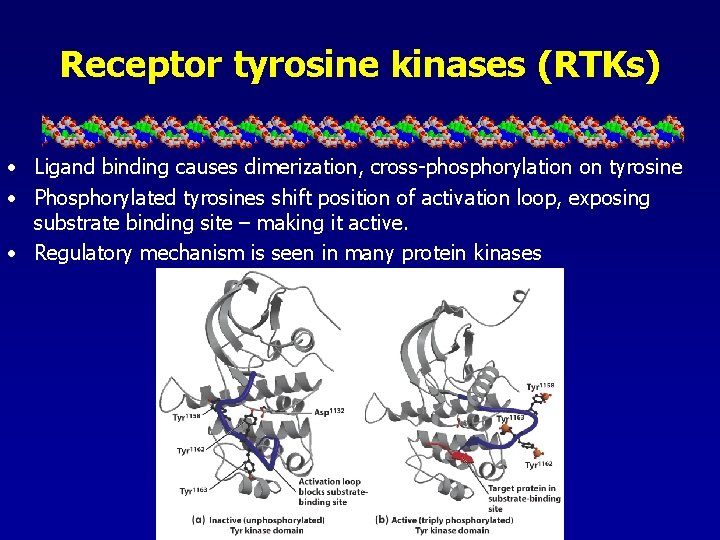
Receptor tyrosine kinases (RTKs) • Ligand binding causes dimerization, cross-phosphorylation on tyrosine • Phosphorylated tyrosines shift position of activation loop, exposing substrate binding site – making it active. • Regulatory mechanism is seen in many protein kinases
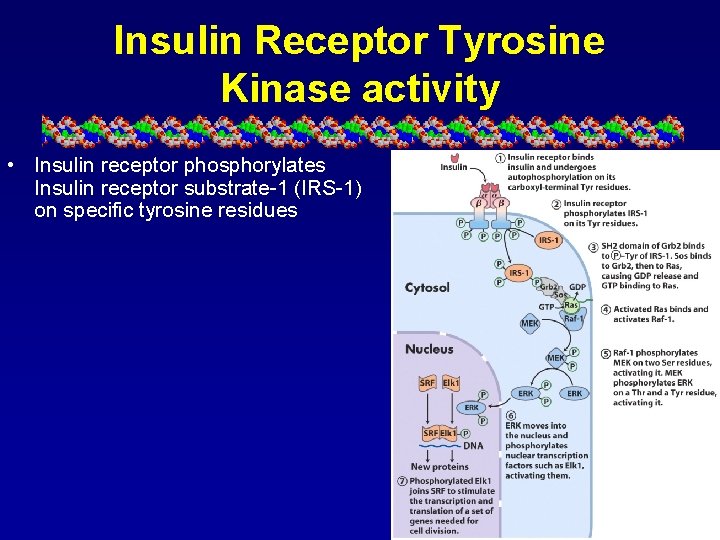
Insulin Receptor Tyrosine Kinase activity • Insulin receptor phosphorylates Insulin receptor substrate-1 (IRS-1) on specific tyrosine residues
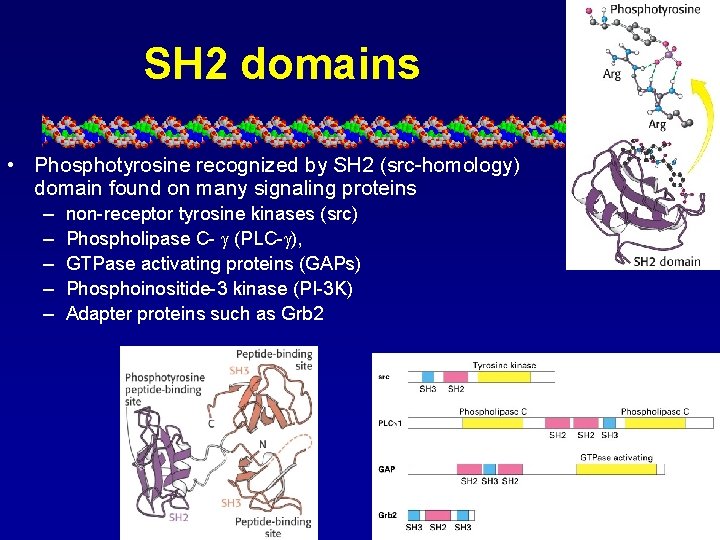
SH 2 domains • Phosphotyrosine recognized by SH 2 (src-homology) domain found on many signaling proteins – – – non-receptor tyrosine kinases (src) Phospholipase C- (PLC- ), GTPase activating proteins (GAPs) Phosphoinositide-3 kinase (PI-3 K) Adapter proteins such as Grb 2
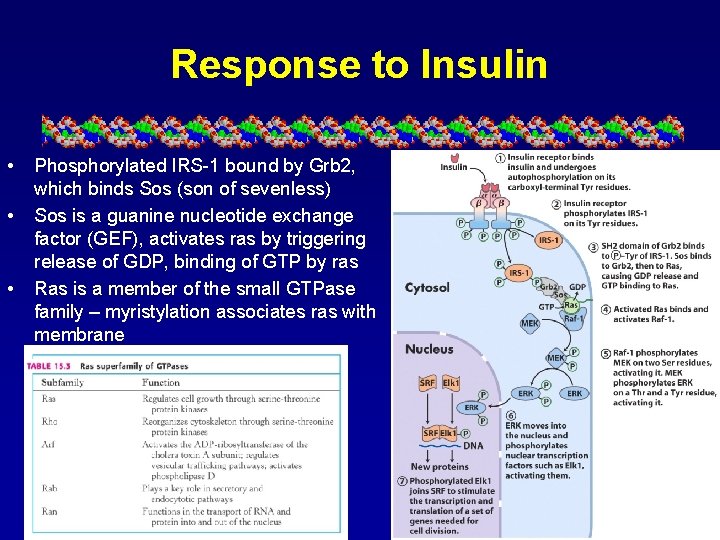
Response to Insulin • • • Phosphorylated IRS-1 bound by Grb 2, which binds Sos (son of sevenless) Sos is a guanine nucleotide exchange factor (GEF), activates ras by triggering release of GDP, binding of GTP by ras Ras is a member of the small GTPase family – myristylation associates ras with membrane
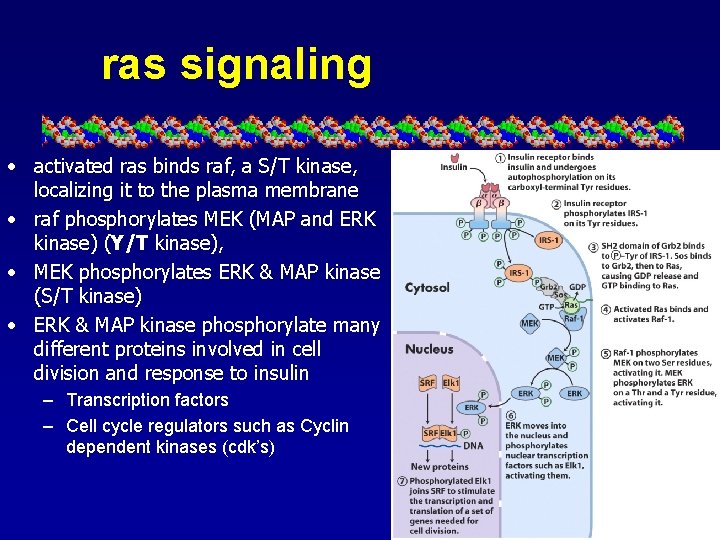
ras signaling • activated ras binds raf, a S/T kinase, localizing it to the plasma membrane • raf phosphorylates MEK (MAP and ERK kinase) (Y/T kinase), • MEK phosphorylates ERK & MAP kinase (S/T kinase) • ERK & MAP kinase phosphorylate many different proteins involved in cell division and response to insulin – Transcription factors – Cell cycle regulators such as Cyclin dependent kinases (cdk’s)
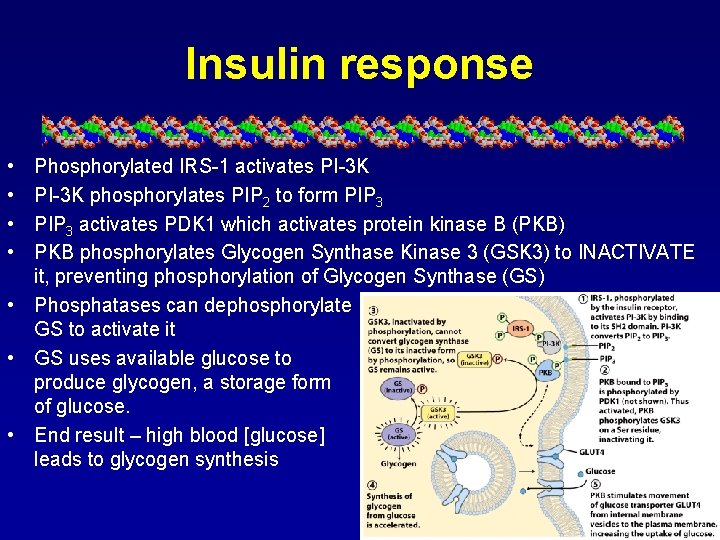
Insulin response • • Phosphorylated IRS-1 activates PI-3 K phosphorylates PIP 2 to form PIP 3 activates PDK 1 which activates protein kinase B (PKB) PKB phosphorylates Glycogen Synthase Kinase 3 (GSK 3) to INACTIVATE it, preventing phosphorylation of Glycogen Synthase (GS) • Phosphatases can dephosphorylate GS to activate it • GS uses available glucose to produce glycogen, a storage form of glucose. • End result – high blood [glucose] leads to glycogen synthesis

Stop here!
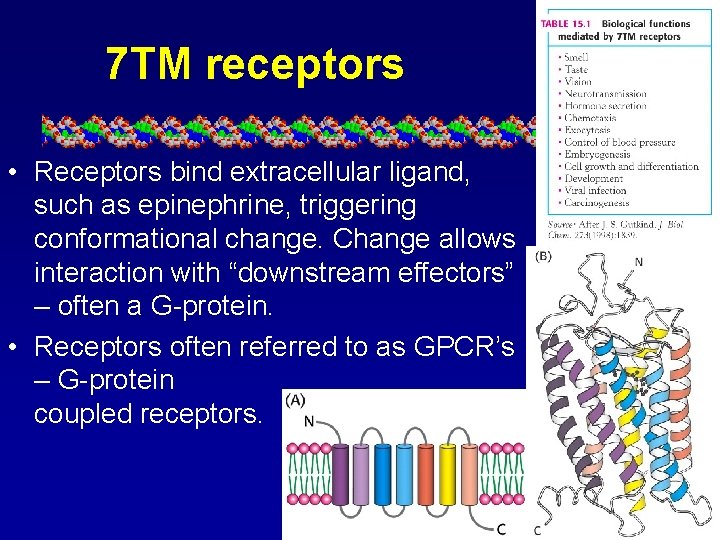
7 TM receptors • Receptors bind extracellular ligand, such as epinephrine, triggering conformational change. Change allows interaction with “downstream effectors” – often a G-protein. • Receptors often referred to as GPCR’s – G-protein coupled receptors.
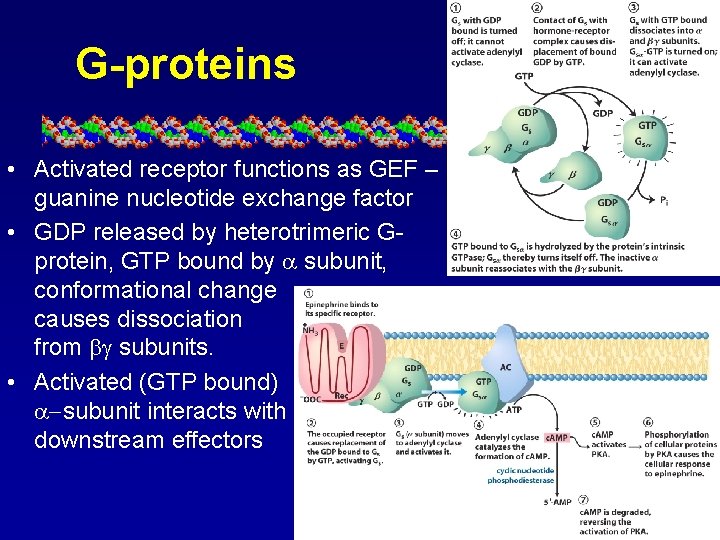
G-proteins • Activated receptor functions as GEF – guanine nucleotide exchange factor • GDP released by heterotrimeric Gprotein, GTP bound by subunit, conformational change causes dissociation from subunits. • Activated (GTP bound) -subunit interacts with downstream effectors
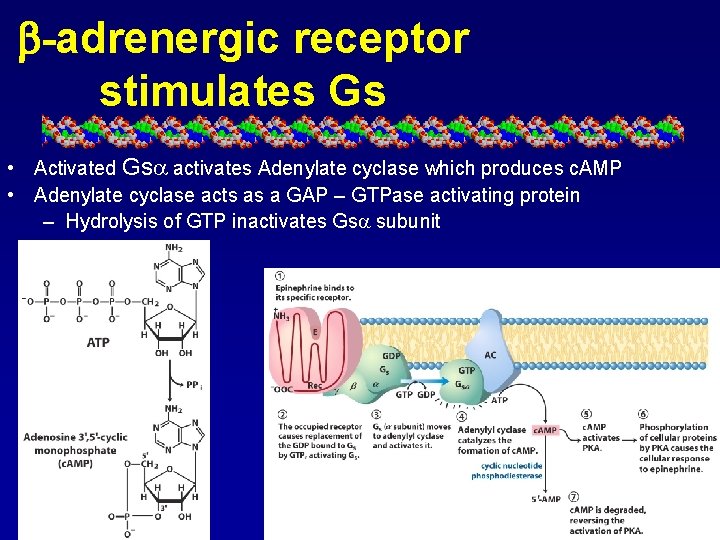
b-adrenergic receptor stimulates Gs • Activated Gs activates Adenylate cyclase which produces c. AMP • Adenylate cyclase acts as a GAP – GTPase activating protein – Hydrolysis of GTP inactivates Gs subunit
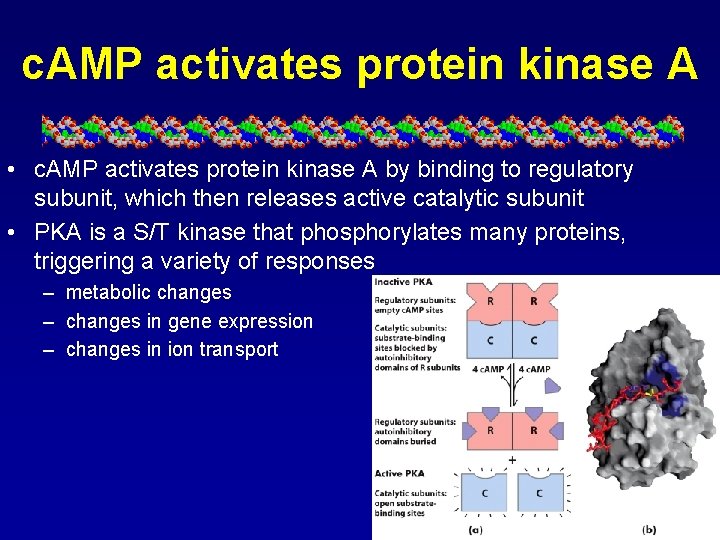
c. AMP activates protein kinase A • c. AMP activates protein kinase A by binding to regulatory subunit, which then releases active catalytic subunit • PKA is a S/T kinase that phosphorylates many proteins, triggering a variety of responses – metabolic changes – changes in gene expression – changes in ion transport
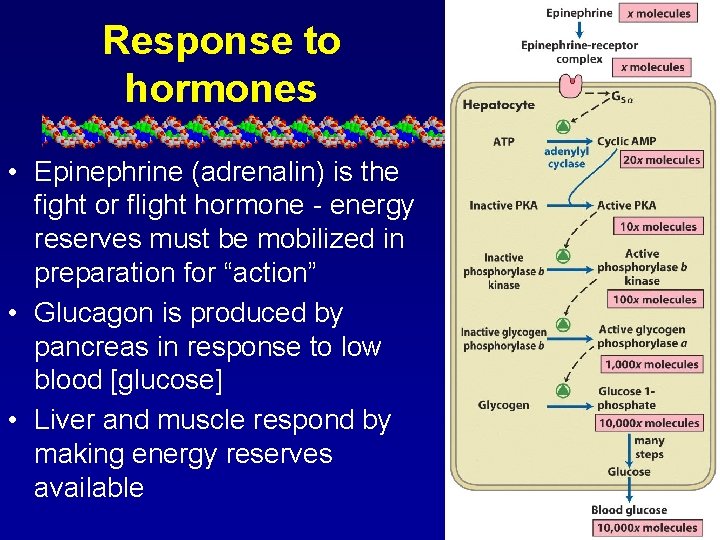
Response to hormones • Epinephrine (adrenalin) is the fight or flight hormone - energy reserves must be mobilized in preparation for “action” • Glucagon is produced by pancreas in response to low blood [glucose] • Liver and muscle respond by making energy reserves available
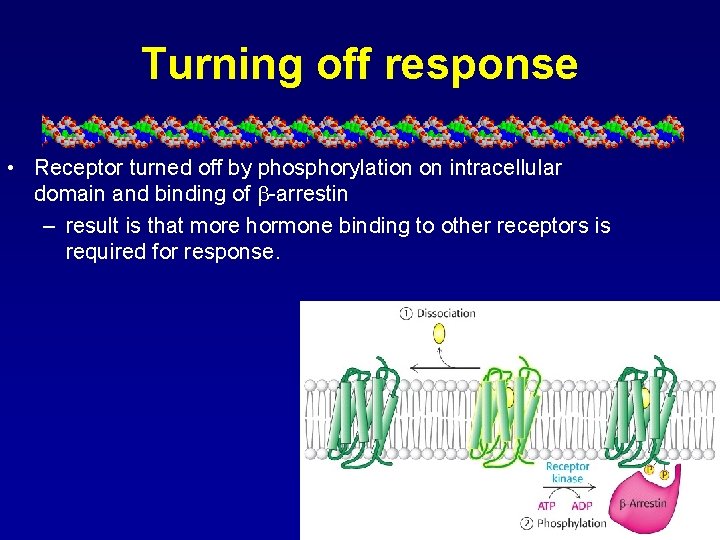
Turning off response • Receptor turned off by phosphorylation on intracellular domain and binding of -arrestin – result is that more hormone binding to other receptors is required for response.
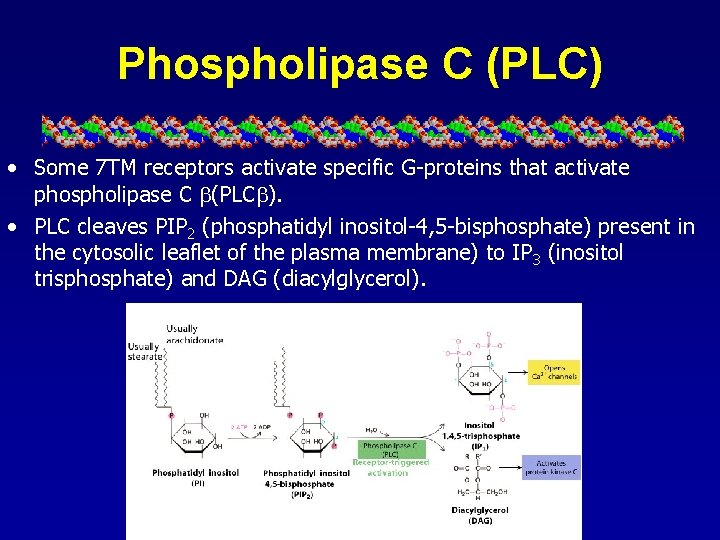
Phospholipase C (PLC) • Some 7 TM receptors activate specific G-proteins that activate phospholipase C (PLC ). • PLC cleaves PIP 2 (phosphatidyl inositol-4, 5 -bisphosphate) present in the cytosolic leaflet of the plasma membrane) to IP 3 (inositol trisphosphate) and DAG (diacylglycerol).
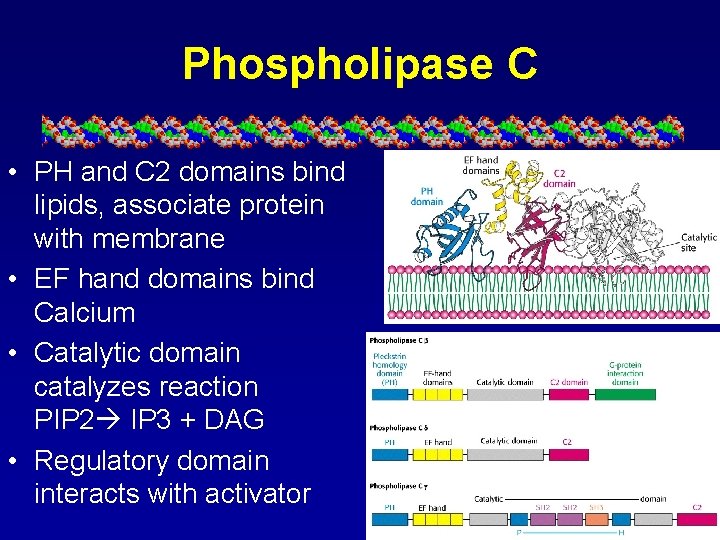
Phospholipase C • PH and C 2 domains bind lipids, associate protein with membrane • EF hand domains bind Calcium • Catalytic domain catalyzes reaction PIP 2 IP 3 + DAG • Regulatory domain interacts with activator
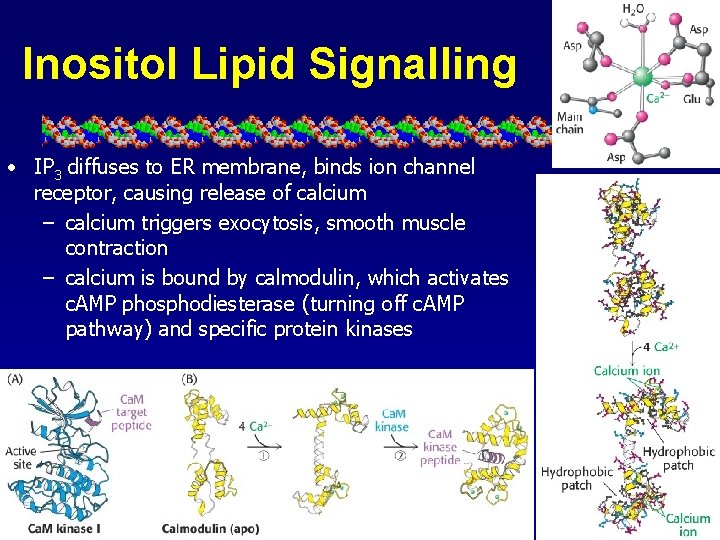
Inositol Lipid Signalling • IP 3 diffuses to ER membrane, binds ion channel receptor, causing release of calcium – calcium triggers exocytosis, smooth muscle contraction – calcium is bound by calmodulin, which activates c. AMP phosphodiesterase (turning off c. AMP pathway) and specific protein kinases
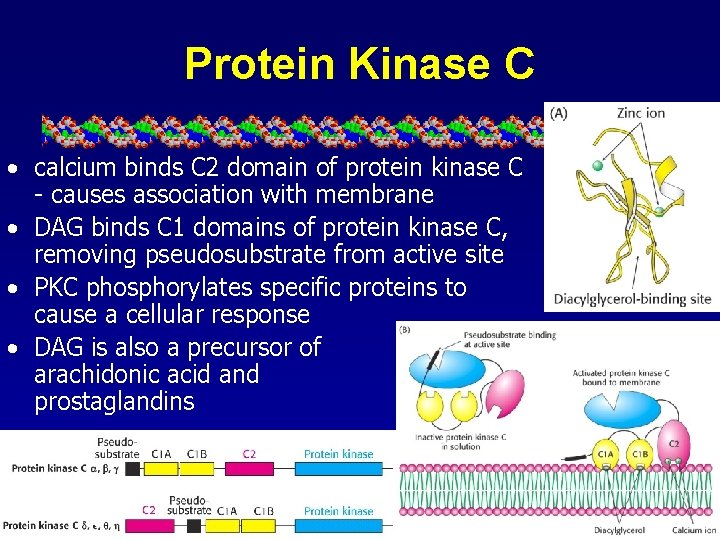
Protein Kinase C • calcium binds C 2 domain of protein kinase C - causes association with membrane • DAG binds C 1 domains of protein kinase C, removing pseudosubstrate from active site • PKC phosphorylates specific proteins to cause a cellular response • DAG is also a precursor of arachidonic acid and prostaglandins
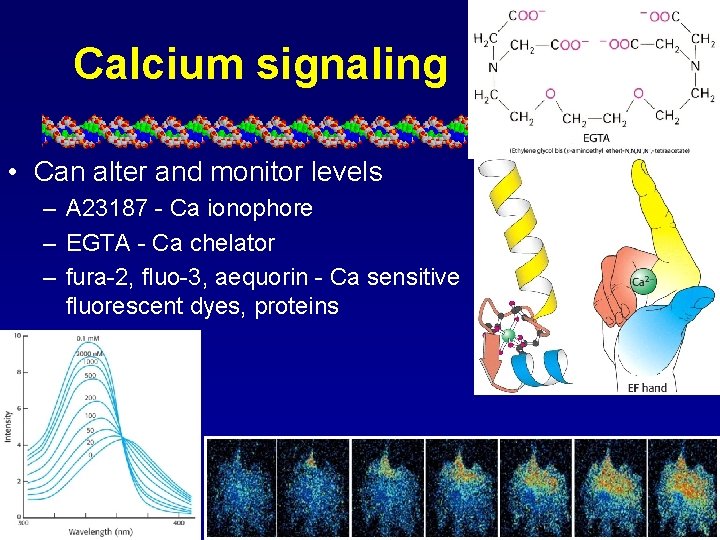
Calcium signaling • Can alter and monitor levels – A 23187 - Ca ionophore – EGTA - Ca chelator – fura-2, fluo-3, aequorin - Ca sensitive fluorescent dyes, proteins
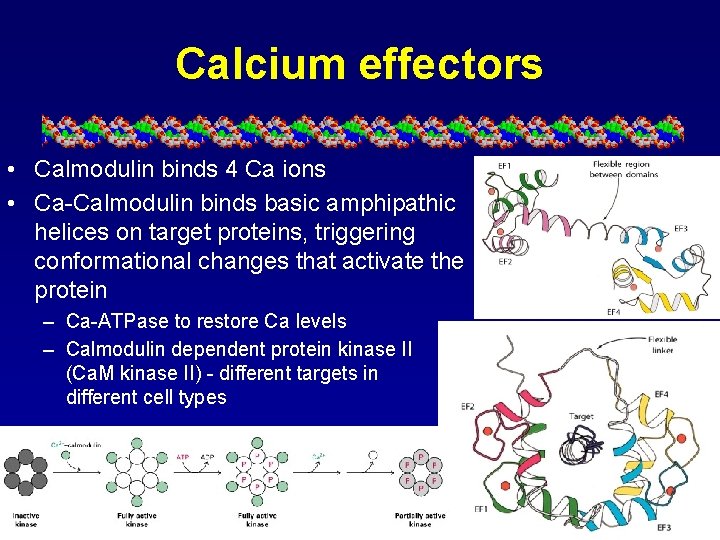
Calcium effectors • Calmodulin binds 4 Ca ions • Ca-Calmodulin binds basic amphipathic helices on target proteins, triggering conformational changes that activate the protein – Ca-ATPase to restore Ca levels – Calmodulin dependent protein kinase II (Ca. M kinase II) - different targets in different cell types

G-protein families
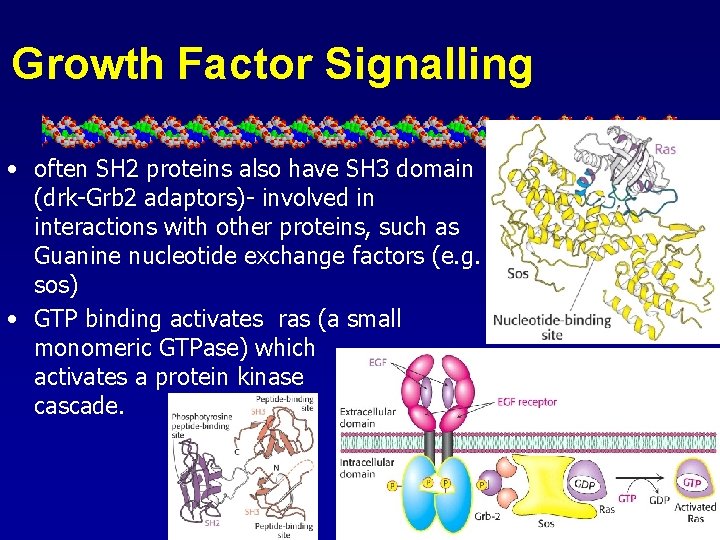
Growth Factor Signalling • often SH 2 proteins also have SH 3 domain (drk-Grb 2 adaptors)- involved in interactions with other proteins, such as Guanine nucleotide exchange factors (e. g. sos) • GTP binding activates ras (a small monomeric GTPase) which activates a protein kinase cascade.
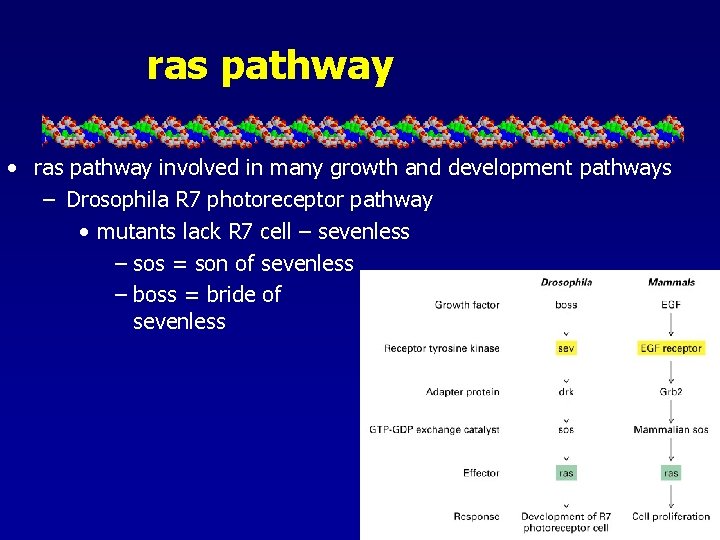
ras pathway • ras pathway involved in many growth and development pathways – Drosophila R 7 photoreceptor pathway • mutants lack R 7 cell – sevenless – sos = son of sevenless – boss = bride of sevenless
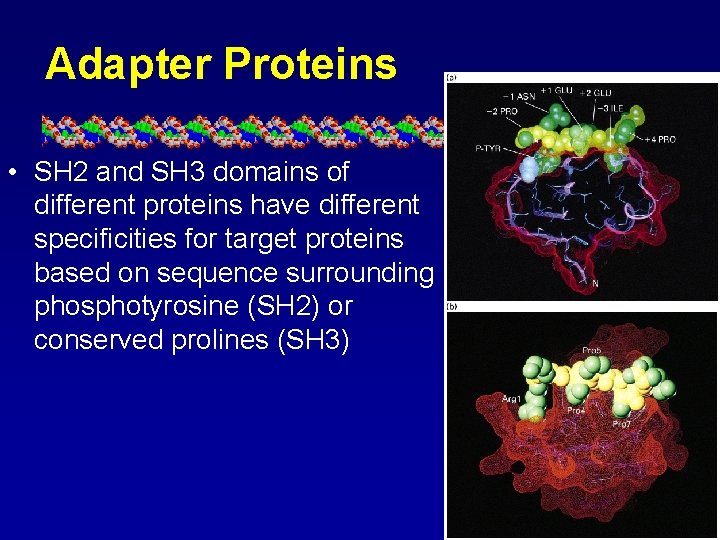
Adapter Proteins • SH 2 and SH 3 domains of different proteins have different specificities for target proteins based on sequence surrounding phosphotyrosine (SH 2) or conserved prolines (SH 3)
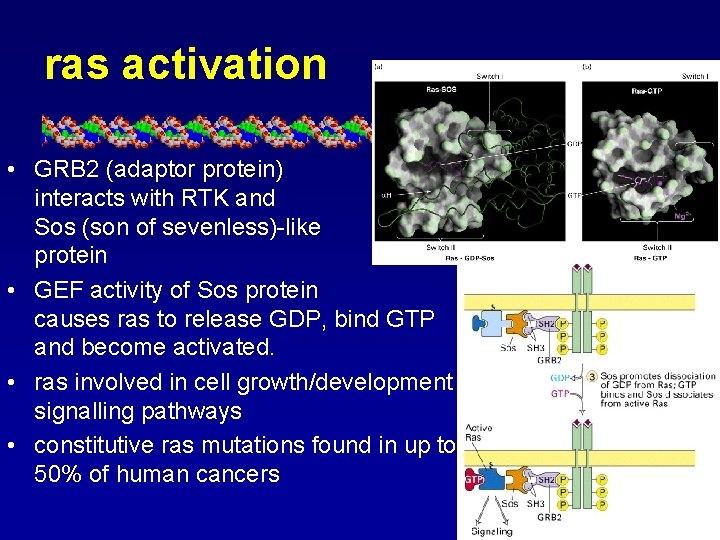
ras activation • GRB 2 (adaptor protein) interacts with RTK and Sos (son of sevenless)-like protein • GEF activity of Sos protein causes ras to release GDP, bind GTP and become activated. • ras involved in cell growth/development signalling pathways • constitutive ras mutations found in up to 50% of human cancers
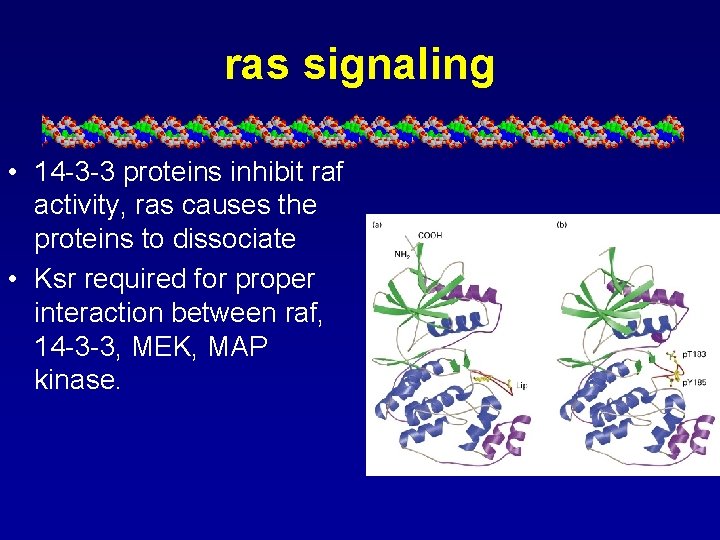
ras signaling • 14 -3 -3 proteins inhibit raf activity, ras causes the proteins to dissociate • Ksr required for proper interaction between raf, 14 -3 -3, MEK, MAP kinase.
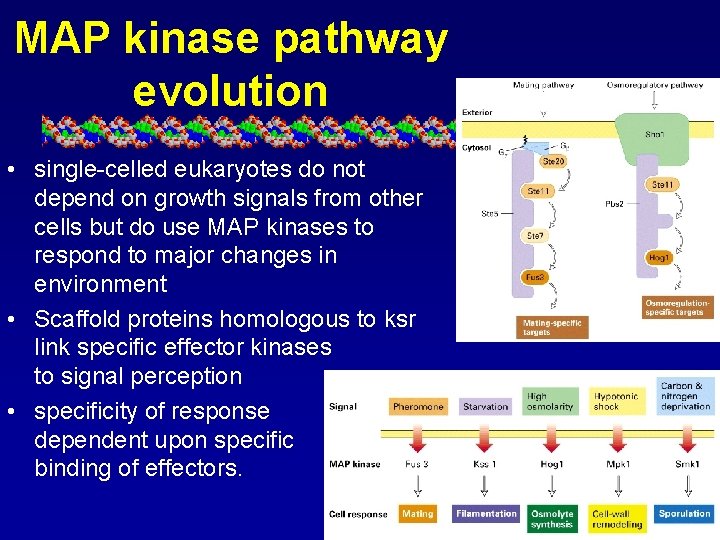
MAP kinase pathway evolution • single-celled eukaryotes do not depend on growth signals from other cells but do use MAP kinases to respond to major changes in environment • Scaffold proteins homologous to ksr link specific effector kinases to signal perception • specificity of response dependent upon specific binding of effectors.
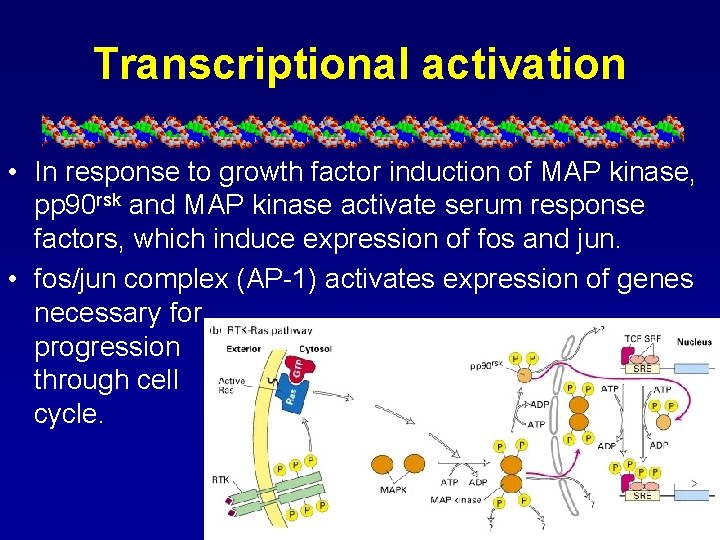
Transcriptional activation • In response to growth factor induction of MAP kinase, pp 90 rsk and MAP kinase activate serum response factors, which induce expression of fos and jun. • fos/jun complex (AP-1) activates expression of genes necessary for progression through cell cycle.
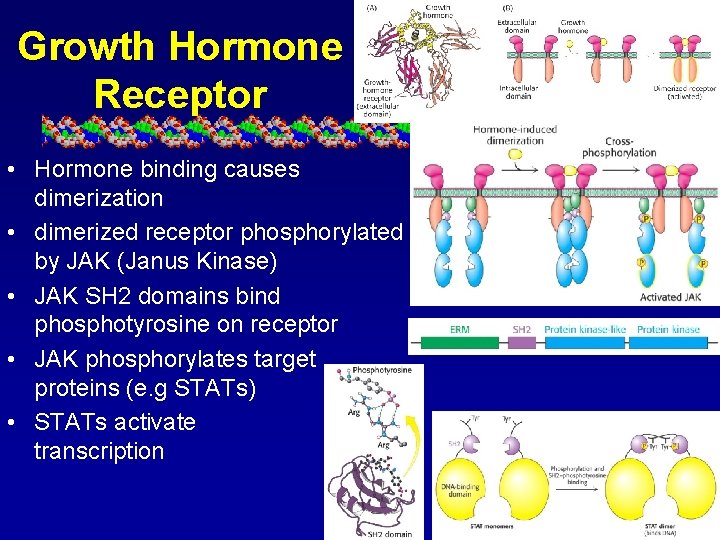
Growth Hormone Receptor • Hormone binding causes dimerization • dimerized receptor phosphorylated by JAK (Janus Kinase) • JAK SH 2 domains bind phosphotyrosine on receptor • JAK phosphorylates target proteins (e. g STATs) • STATs activate transcription

Signal Amplification • Signal transduction cascades amplify a signal • ligand-receptor complex can activate many G • each G can activate adenyl cyclase such that many c. AMP are produced • each c. AMP activates c. APK which can phosphorylate multiple proteins • each protein can then affect multiple downstream effectors
- Slides: 33