Signal Transduction 2 BL 4010 12 05 Signalling

Signal Transduction 2 BL 4010 12. 05

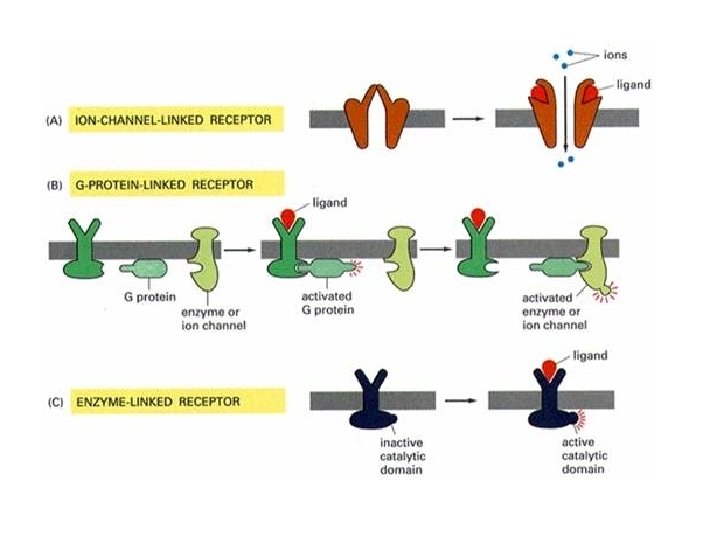
Signalling vocabulary • • • Signal/stimulus Effector Receptor Messenger Ligand Cascade
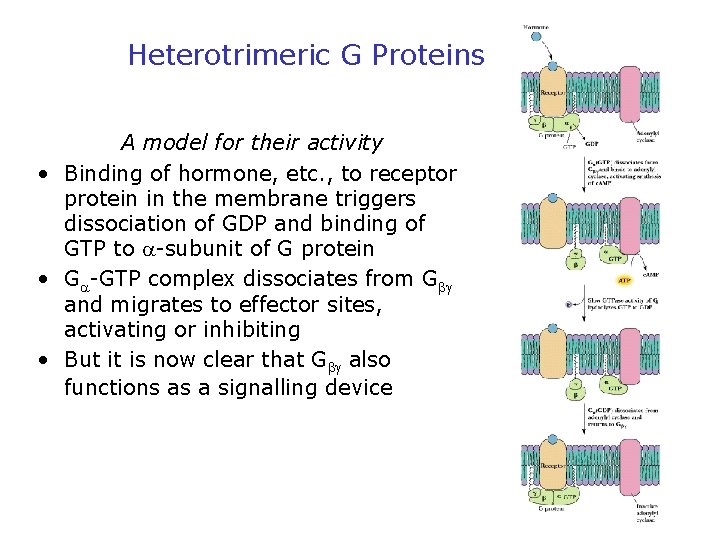
Heterotrimeric G Proteins A model for their activity • Binding of hormone, etc. , to receptor protein in the membrane triggers dissociation of GDP and binding of GTP to -subunit of G protein • G -GTP complex dissociates from G and migrates to effector sites, activating or inhibiting • But it is now clear that G also functions as a signalling device

GTPase activating proteins (GAPs)
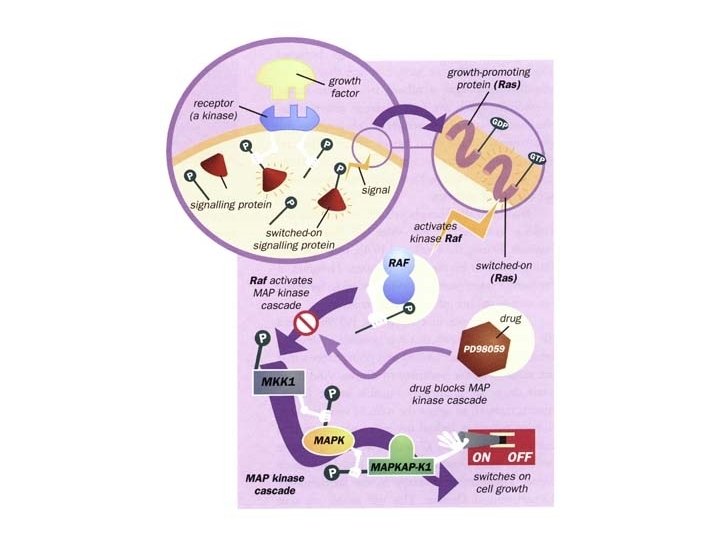
MAP Kinase Cascade
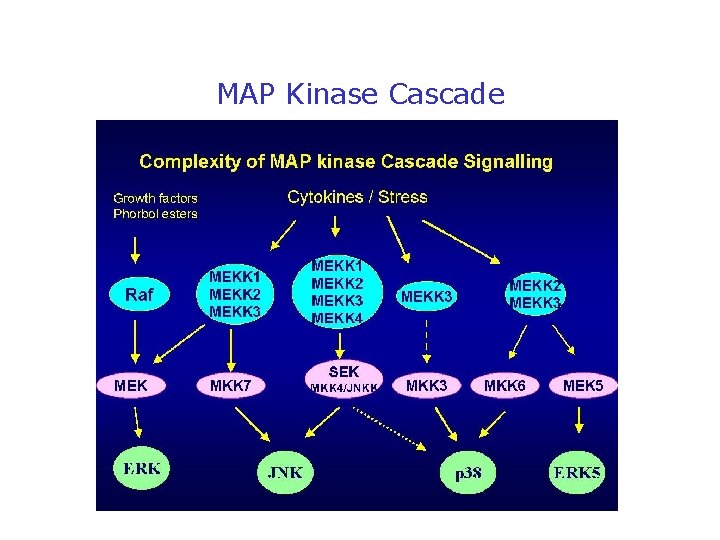
MAP Kinase Cascade
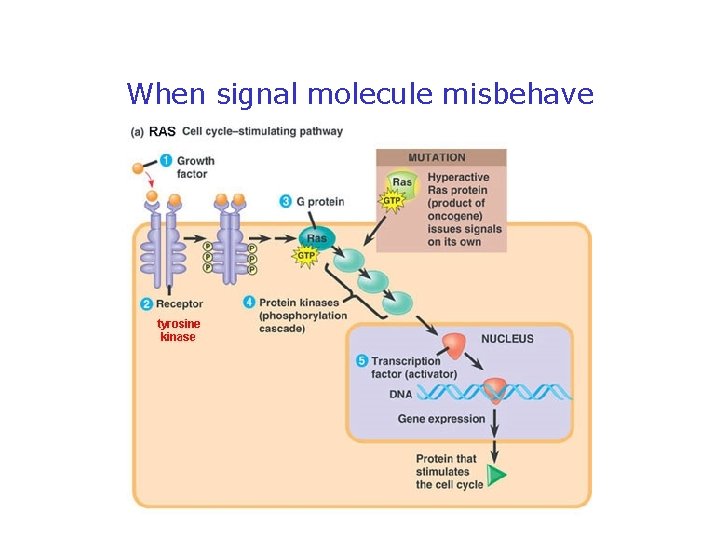
When signal molecule misbehave

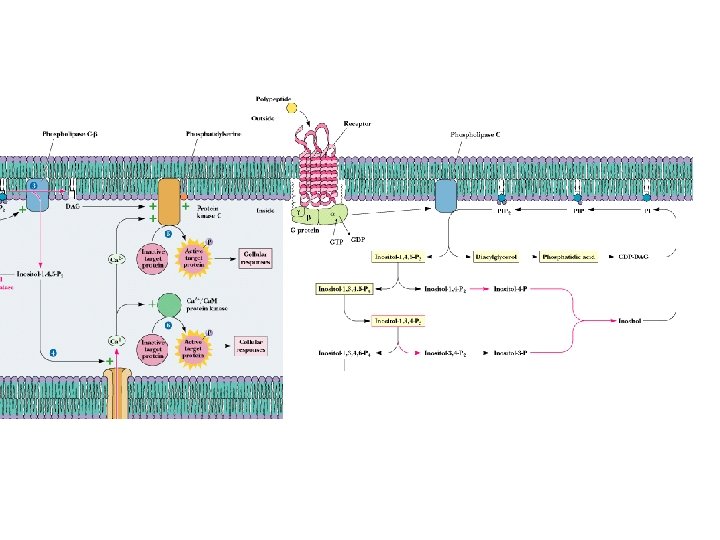
Phospholipases Release Second Messengers • Inositol phospholipids yield IP 3 and DAG • PLC is activated by 7 -TMS receptors and G proteins • PLC is activated by receptor tyrosine kinases (via phosphorylation) • Note PI metabolic pathways and the role of lithium
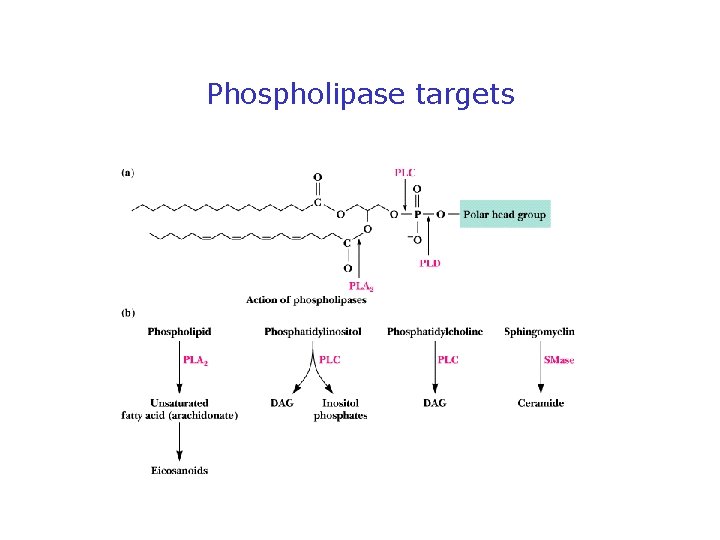
Phospholipase targets
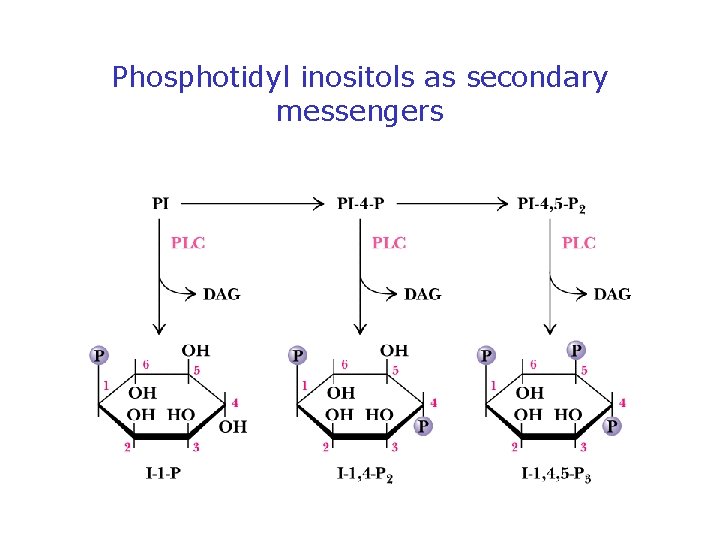
Phosphotidyl inositols as secondary messengers
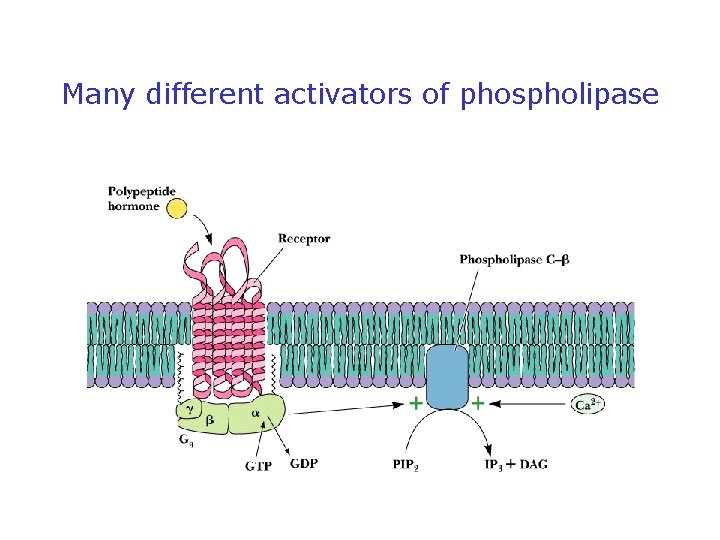
Many different activators of phospholipase
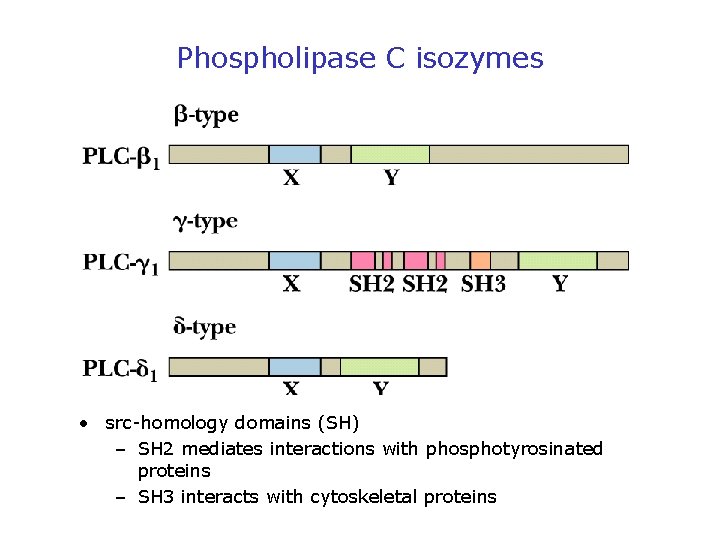
Phospholipase C isozymes • src-homology domains (SH) – SH 2 mediates interactions with phosphotyrosinated proteins – SH 3 interacts with cytoskeletal proteins

Other Lipids as Messengers Recent findings - lots more to come • More recently than for PI, other phospholipids have been found to produce second messengers! • Phosphatidyl choline can produce prostaglandins, diacylglycerol and/or phosphatidic acid • Sphingomyelin and glycosphingolipids also produce signals such as ceramide, a trigger of apoptosis - programmed cell death

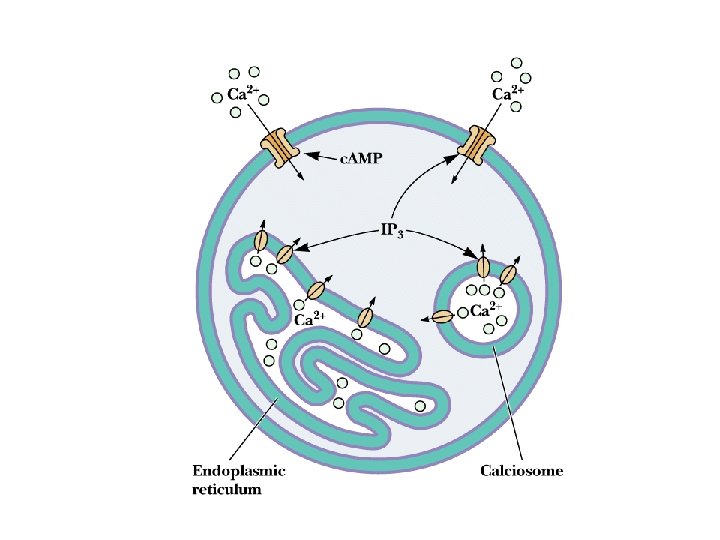
Ca 2+ as a Second Messenger Several sources of Ca 2+ in cells! • [Ca 2+] in cells is normally very low: < 1 M • Calcium can enter cell from outside or from ER and calciosomes (plants store Ca 2+ in oxalate crystals) • CICR - Calcium-Induced Calcium Release – see animation
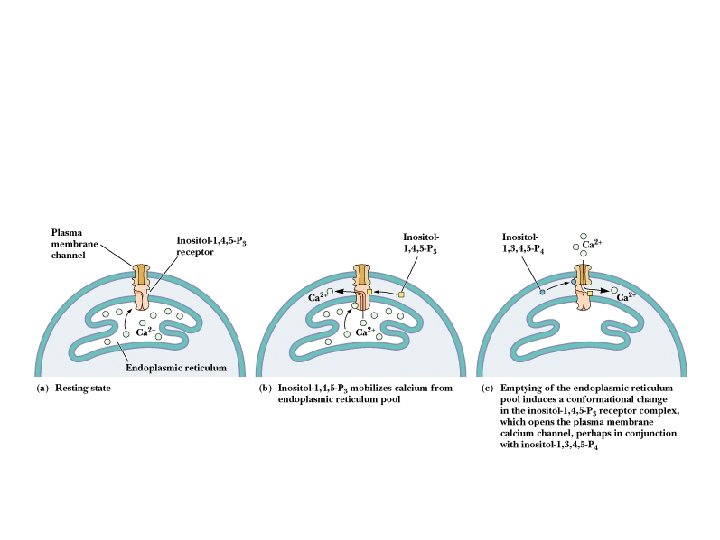

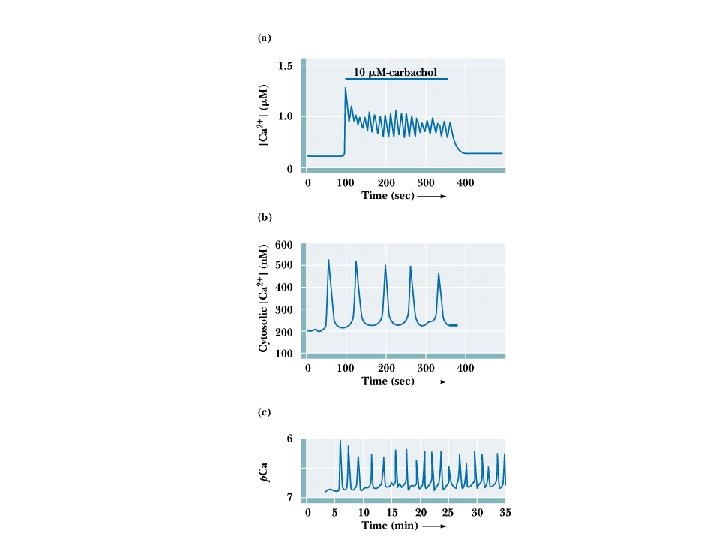
Calcium Oscillations! M. Berridge's model of Ca 2+ signals • Ca 2+ was once thought to merely rise in cells to signal and drop when the signal was over • Berridge's work demonstrates that Ca 2+ levels oscillate in cells! • The purpose may be to protect cell components that are sensitive to high calcium, or perhaps to create waves of Ca 2+ in the cell
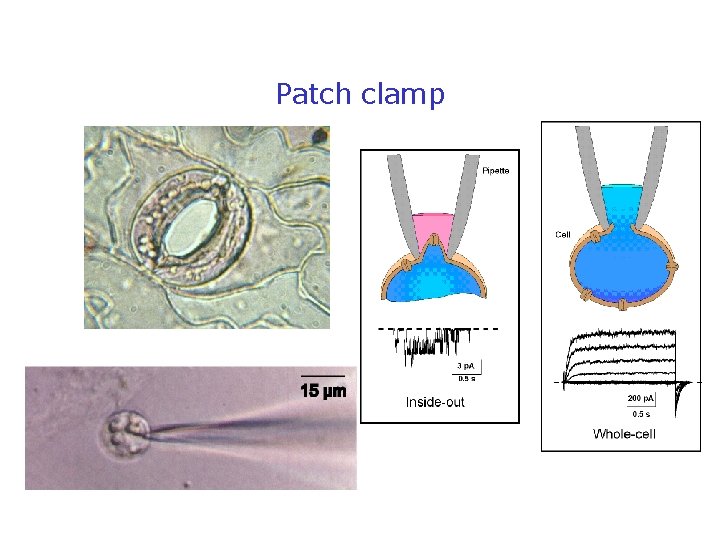
Patch clamp

Ca 2+-Binding Proteins • • Mediators of Ca 2+effects in cells Many cellular proteins modulate Ca 2+ effects 3 main types: protein kinase Cs, Ca 2+-modulated proteins and annexins Kretsinger characterized the structure of parvalbumin, prototype of Ca 2+-modulated proteins "EF hand" proteins bind BAA helices
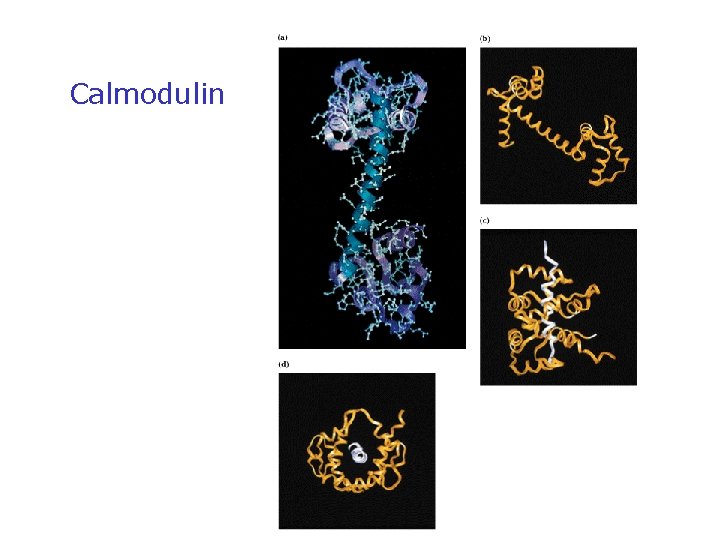
Calmodulin

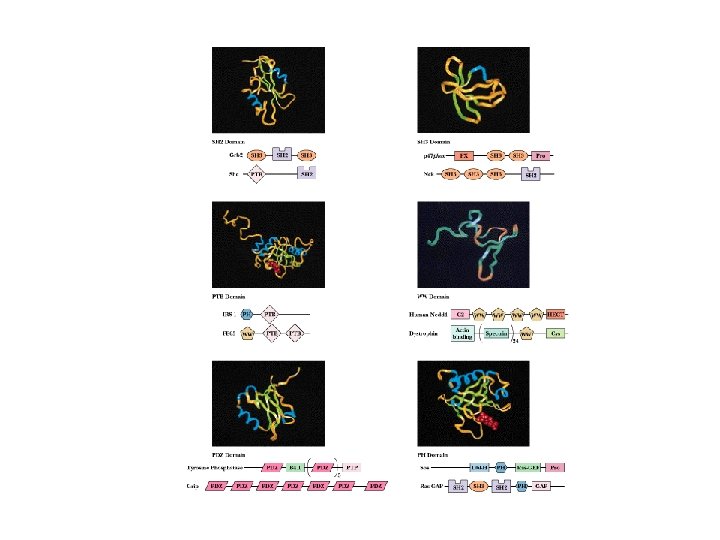
Protein Modules in Signal Transduction • Signal transduction in cell occurs via protein and protein-lipid interactions based on protein modules • Most signaling proteins consist of two or more modules • This permits assembly of functional signaling complexes

Transduction of two second messenger signals • • PKC is activated by DAG and Ca 2+ Most PKC isozymes have several domains, including ATPbinding domain, substrate-binding domain, Ca-binding domain and a phorbol ester-binding domain Phorbol esters are apparent analogues of DAG Cellular phosphatases dephosphorylate target proteins Read about okadaic acid
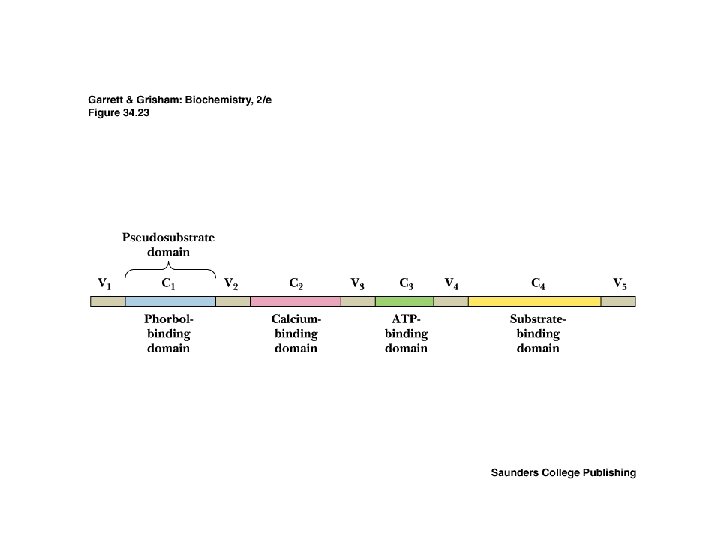
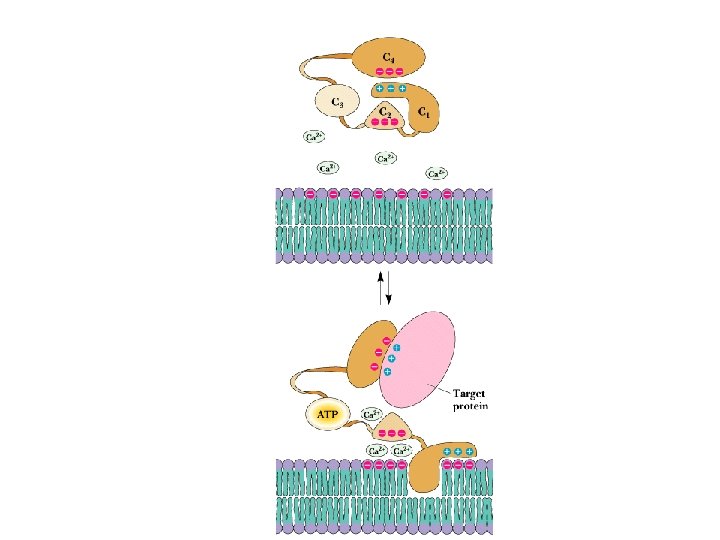

Localization of Signaling Proteins • Adaptor proteins provide docking sites for signaling modules at the membrane • Typical case: IRS-1 (Insulin Receptor Substrate-1) – N-terminal PH domain – PTB domain – 18 potential tyrosine phosphorylation sites – PH and PTB direct IRS-1 to receptor tyrosine kinase - signaling events follow!
Lipids Rafts • first hypothesized in 1988 • nice review: Cary, L. A. & Cooper, J. A. (2000) Molecular switches in lipid rafts. Nature. 404, 945 -947 • Moffett, S. , Brown, D. A. & Linder, M. E. (2000) Lipid-dependent targeting of G proteins into rafts. J. Biol. Chem. 275, 2191 -2198.
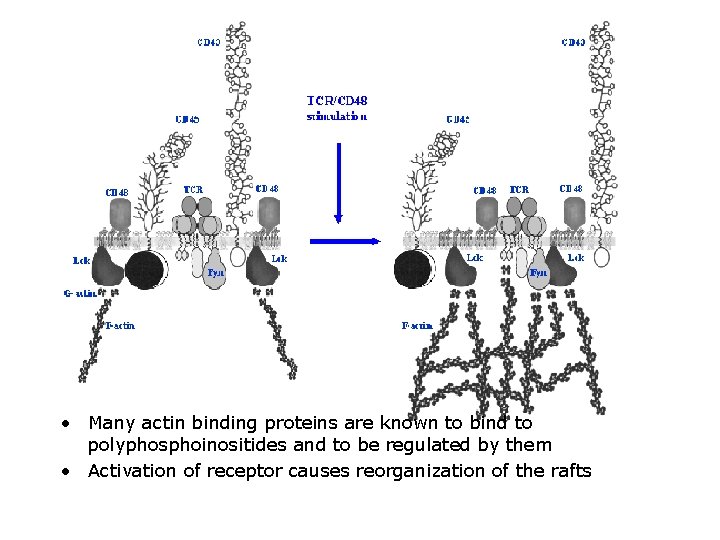
• Many actin binding proteins are known to bind to polyphosphoinositides and to be regulated by them • Activation of receptor causes reorganization of the rafts

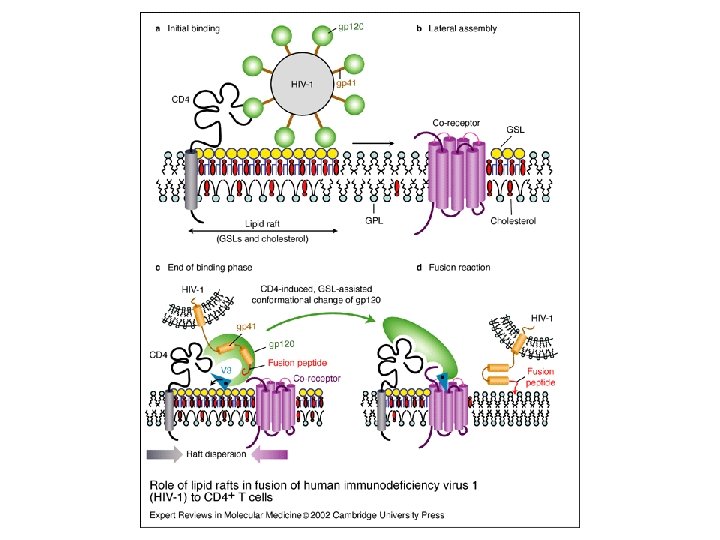
Simons, K. et al. J. Clin. Invest. 2002; 110: 597 -603 J. Fantini, N. Garmy, R. Mahfoud and N. Yahi Lipid rafts: structure, function and role in HIV, Alzheimer’s and prion diseases Expert Reviews in Molecular Medicine: 20 December 2002
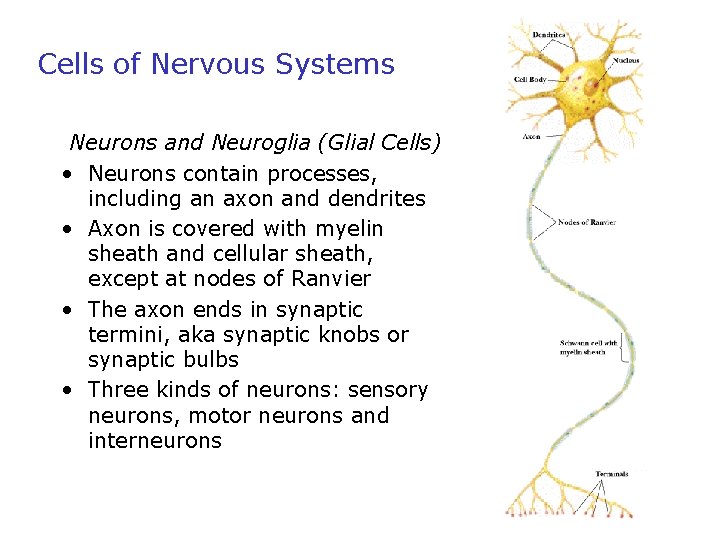
Cells of Nervous Systems Neurons and Neuroglia (Glial Cells) • Neurons contain processes, including an axon and dendrites • Axon is covered with myelin sheath and cellular sheath, except at nodes of Ranvier • The axon ends in synaptic termini, aka synaptic knobs or synaptic bulbs • Three kinds of neurons: sensory neurons, motor neurons and interneurons

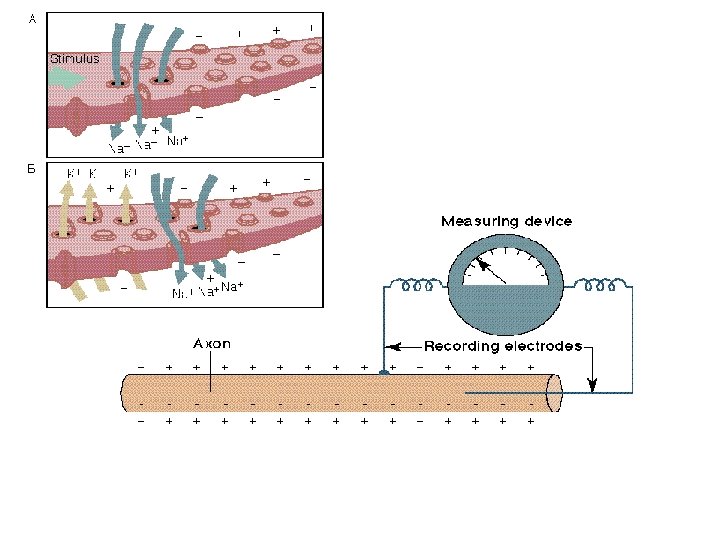
Ion Gradients The source of electrical potentials in neurons • Nerve impulses consist of electrical signals that are transient changes in the electrical potential differences (voltages) across neuron membrane • Difference between Nernst potential and actual potential represents a thermodynamic push
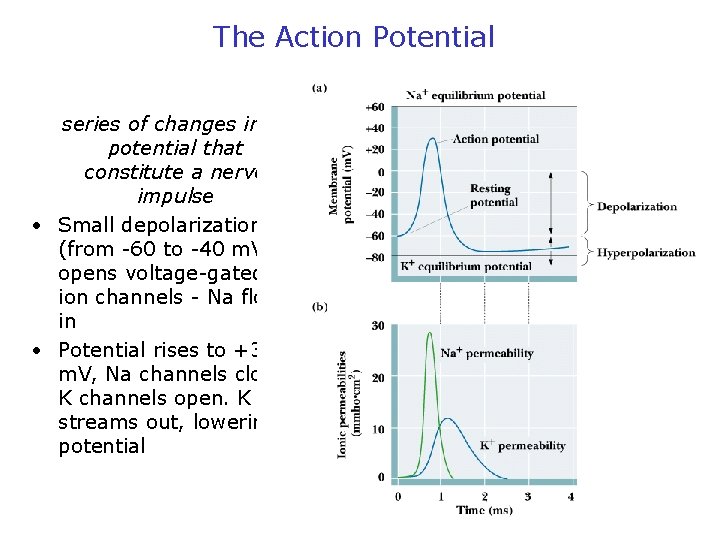
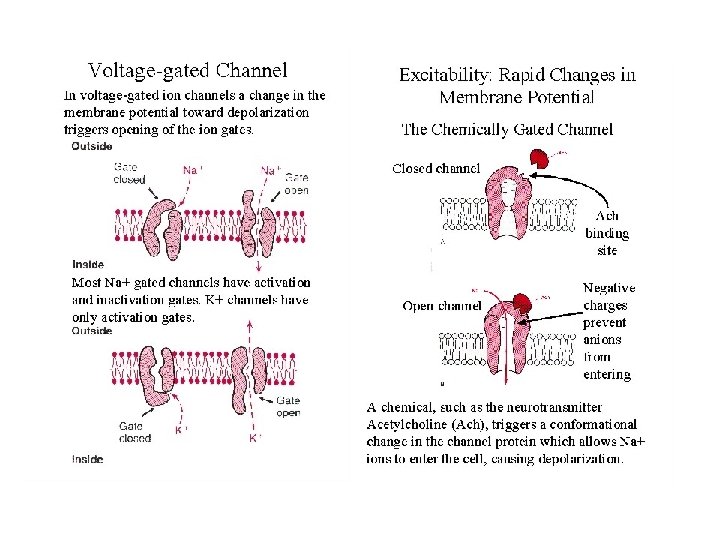
The Action Potential series of changes in potential that constitute a nerve impulse • Small depolarization (from -60 to -40 m. V) opens voltage-gated ion channels - Na flows in • Potential rises to +30 m. V, Na channels close, K channels open. K streams out, lowering potential
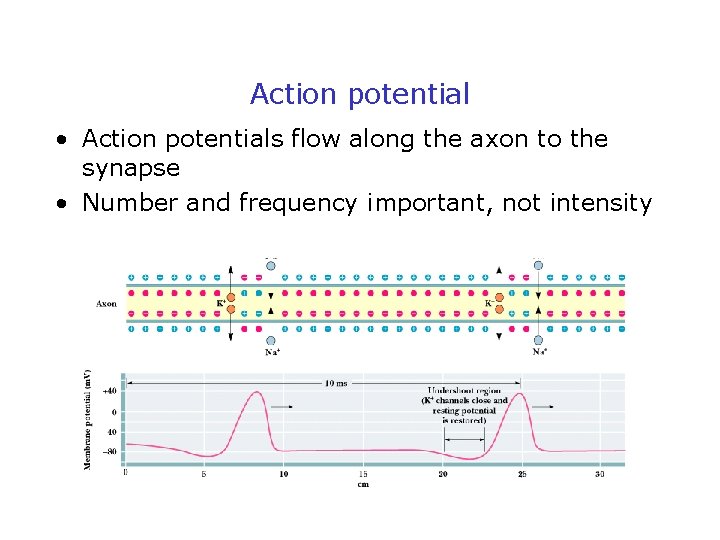
Action potential • Action potentials flow along the axon to the synapse • Number and frequency important, not intensity
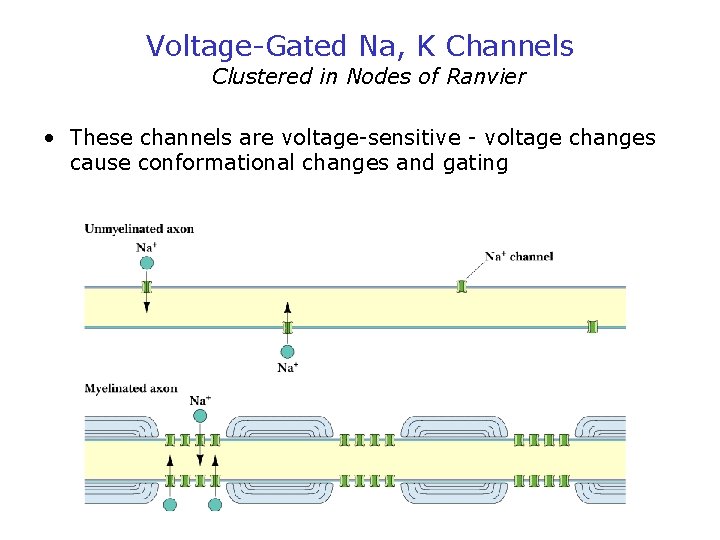
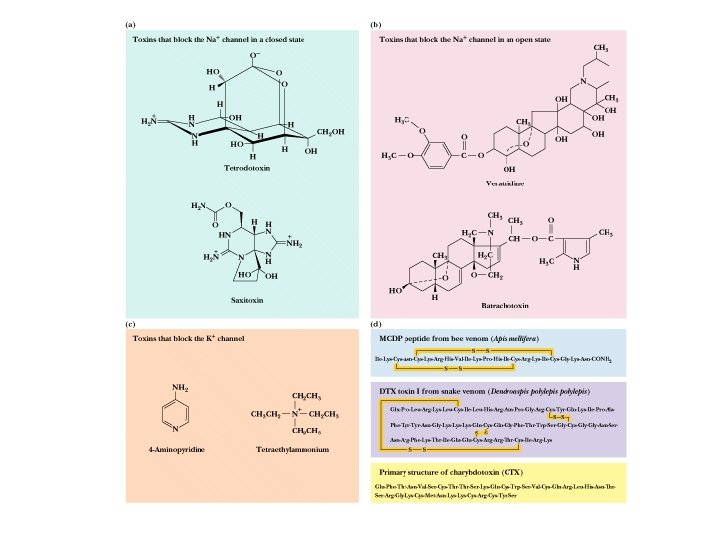
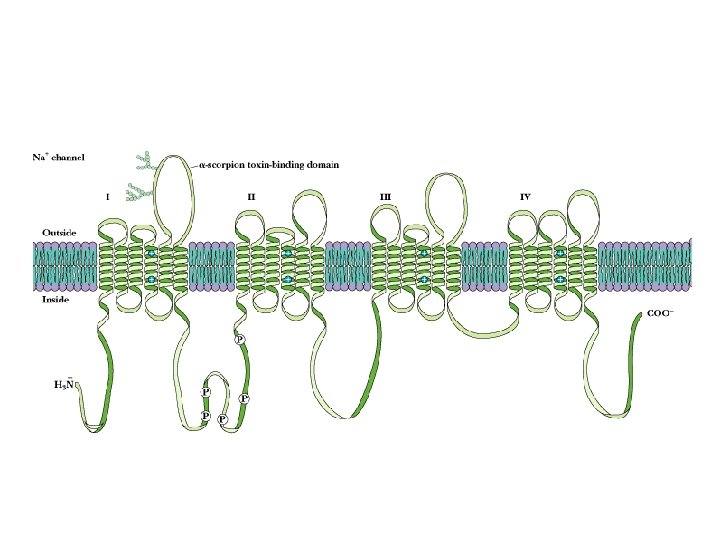
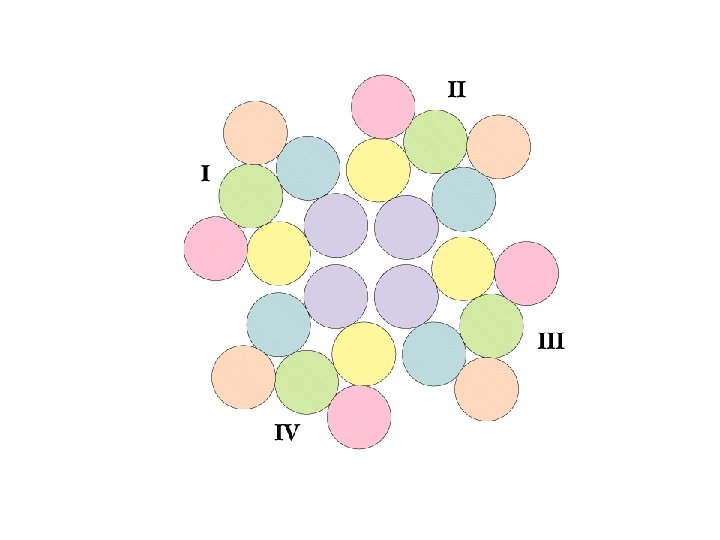
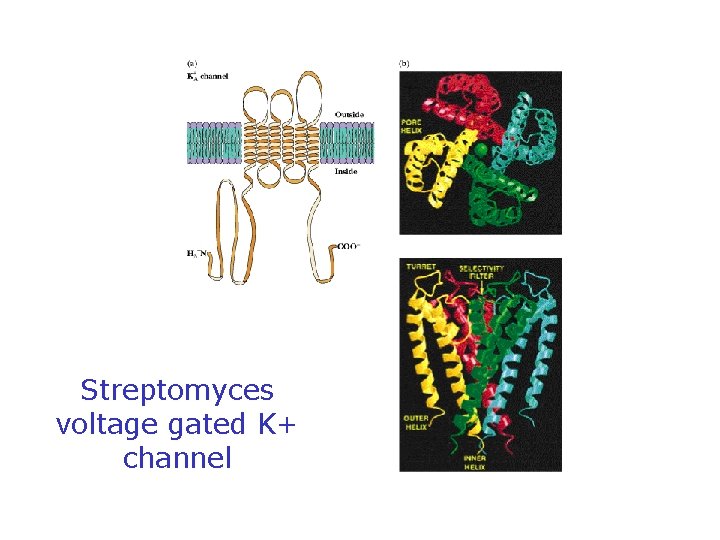
Voltage-Gated Na, K Channels Clustered in Nodes of Ranvier • These channels are voltage-sensitive - voltage changes cause conformational changes and gating
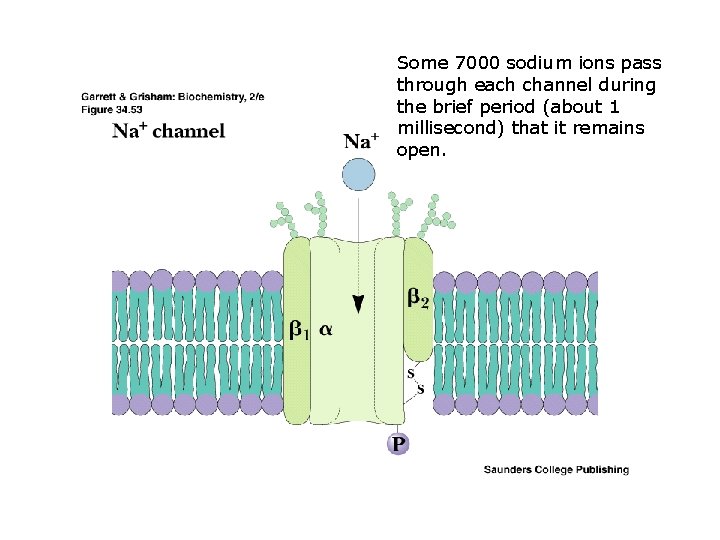
Some 7000 sodium ions pass through each channel during the brief period (about 1 millisecond) that it remains open.
Streptomyces voltage gated K+ channel

Communication at the Synapse • • A crucial feature of neurotransmission Ratio of synapses to neurons in human forebrain is 40, 000 to 1! Chemical synapses are different from electrical Neurotransmitters facilitate cell-cell communication at the synapse Note families of neurotransmitters in Table 34. 6

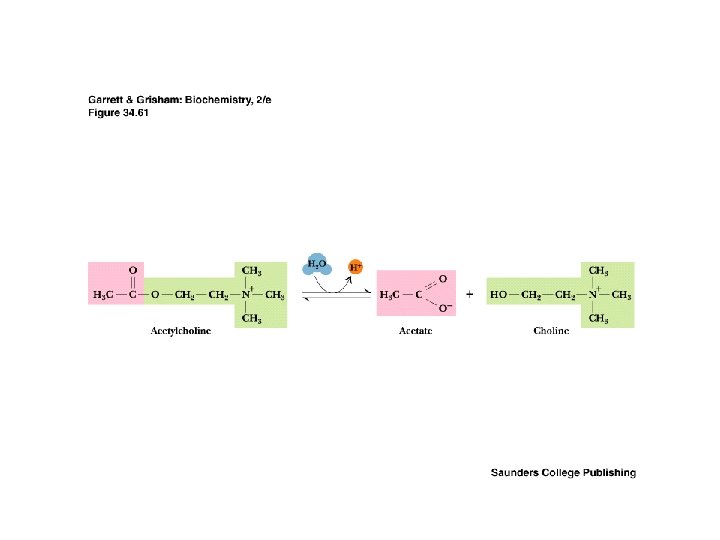
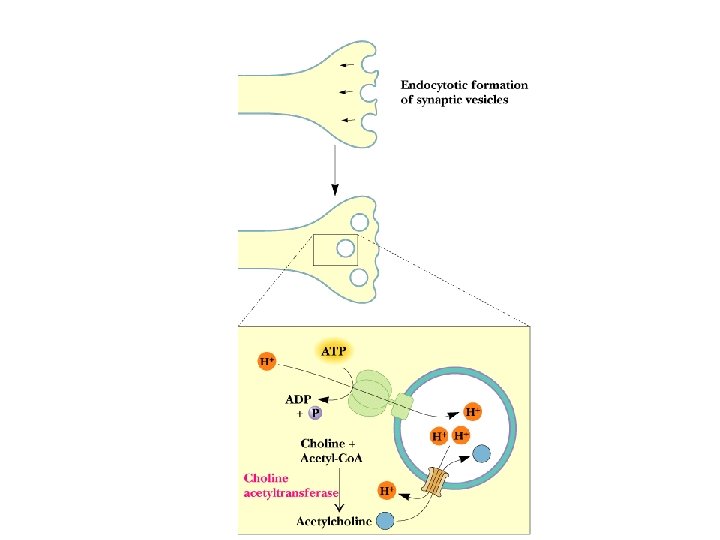
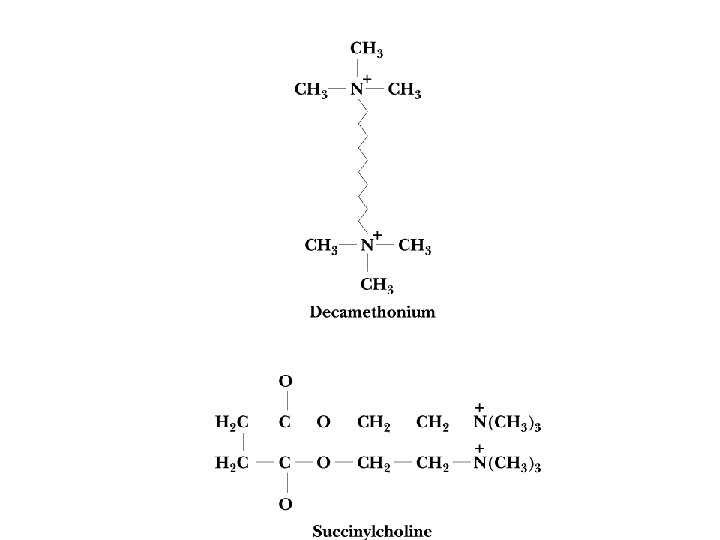
The Cholinergic Synapse • • A model for many others Synaptic vesicles in synaptic knobs contain acetylcholine (10, 000 molecules per vesicle) Arriving action potential depolarizes membrane, opening Ca channels and causing vesicles to fuse with plasma membrane Acetylcholine spills into cleft, migrates to adjacent cells and binds to receptors Toxin effects: botulism toxin inhibits Ac-choline release, black widow's latrotoxin protein overstimulates
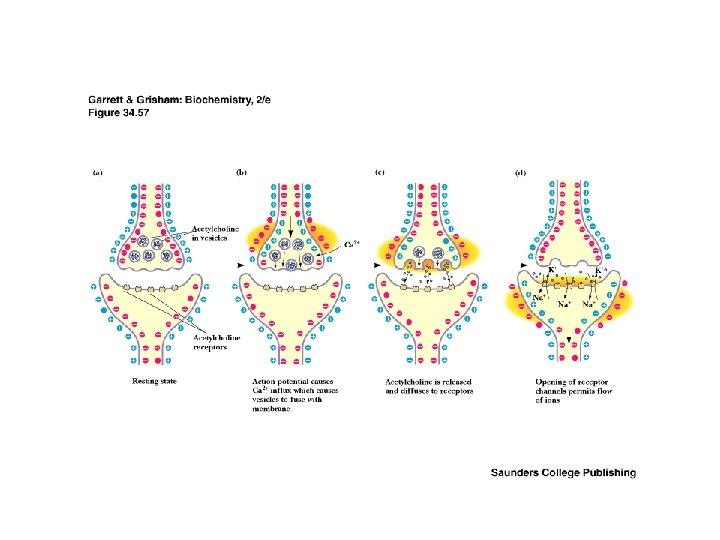

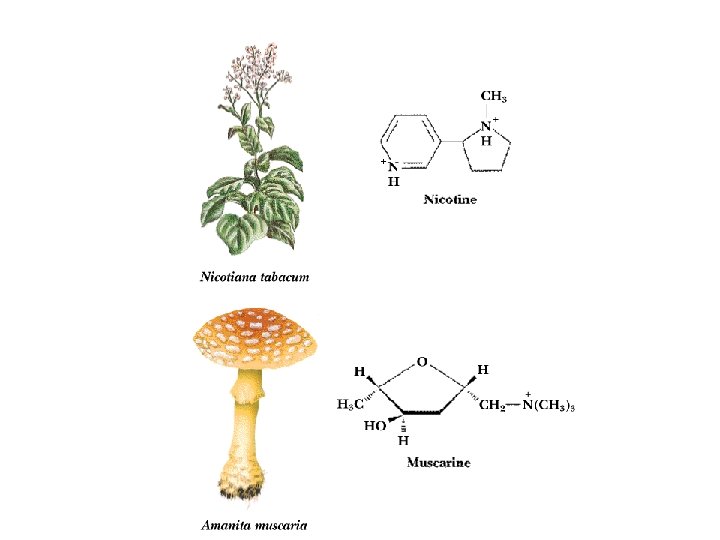
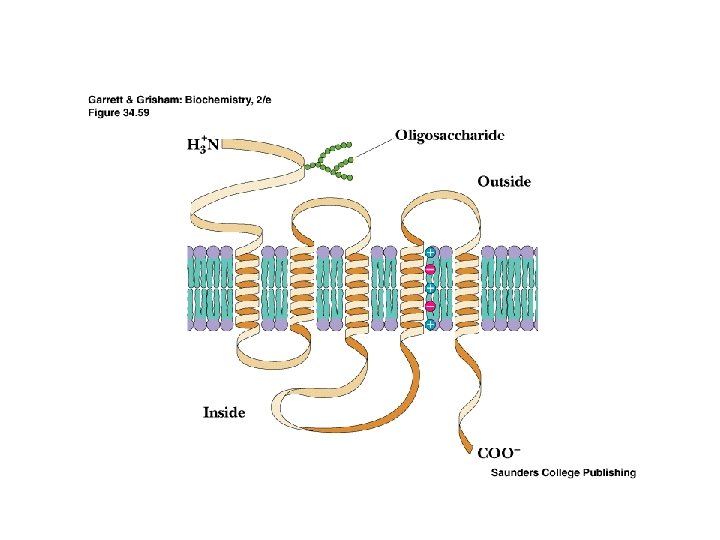
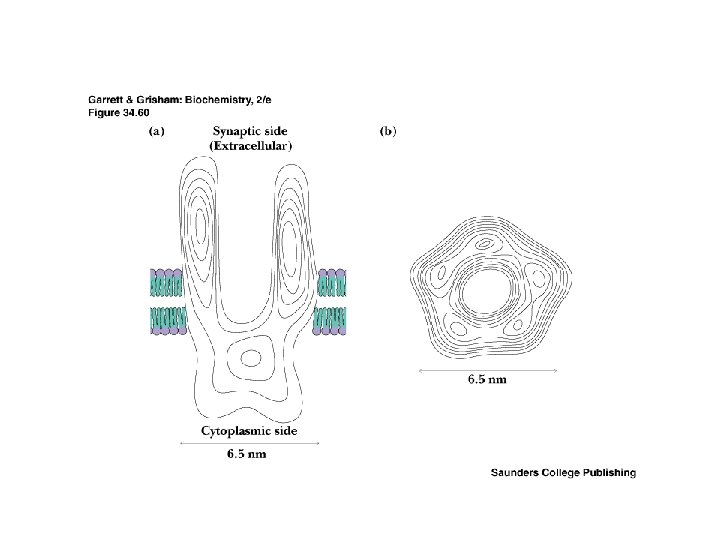
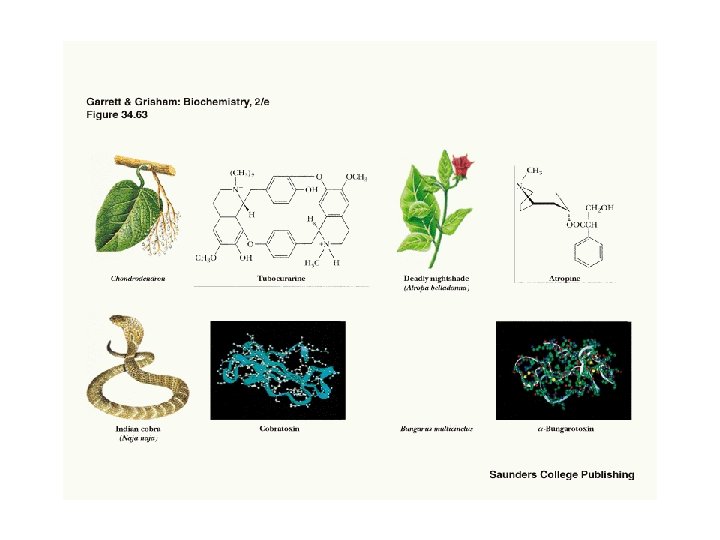
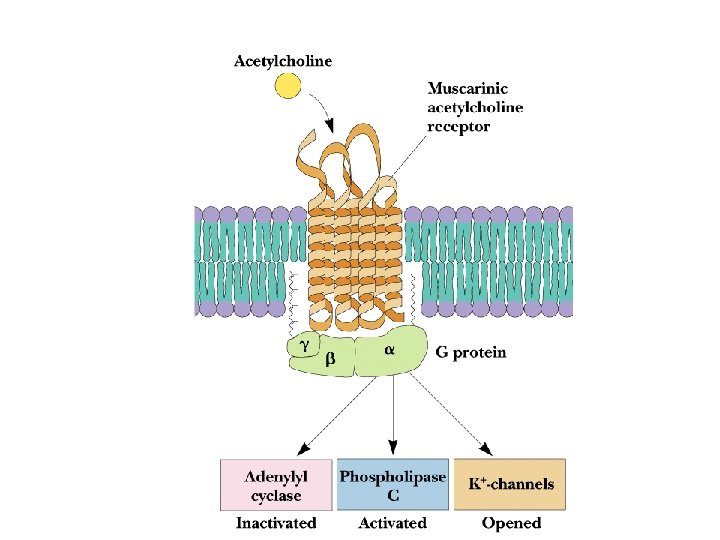
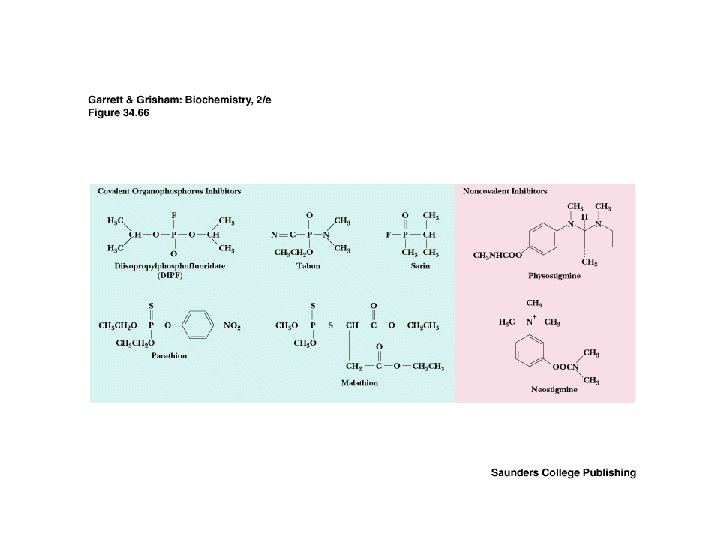
Two Classes of Ac-Ch Receptor • • • Nicotinic and muscarinic As always, toxic agents have helped to identify and purify hard-to-find biomolecules Nicotinic Ac-Ch receptors are voltage-gated ion channels Muscarinic Ac-Ch receptors are transmembrane proteins that interact with G proteins Acetylcholinesterase degrades Ac-Ch in cleft Transport proteins and V-type H+-ATPases return Ac-Ch to vesicles - called reuptake

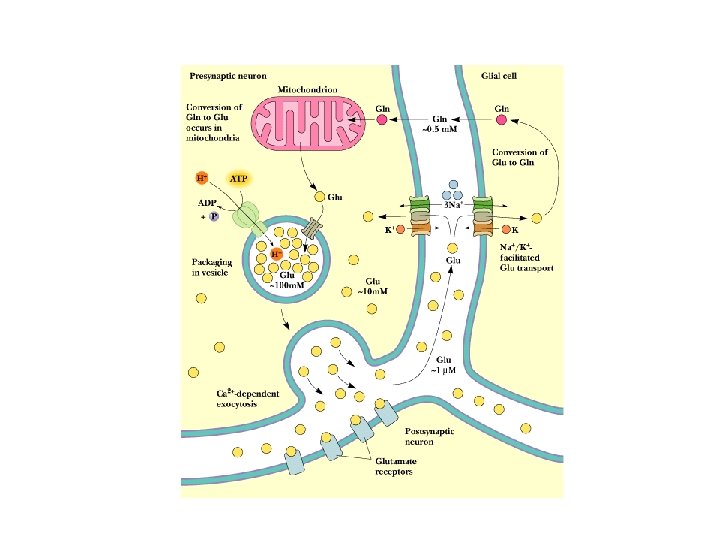
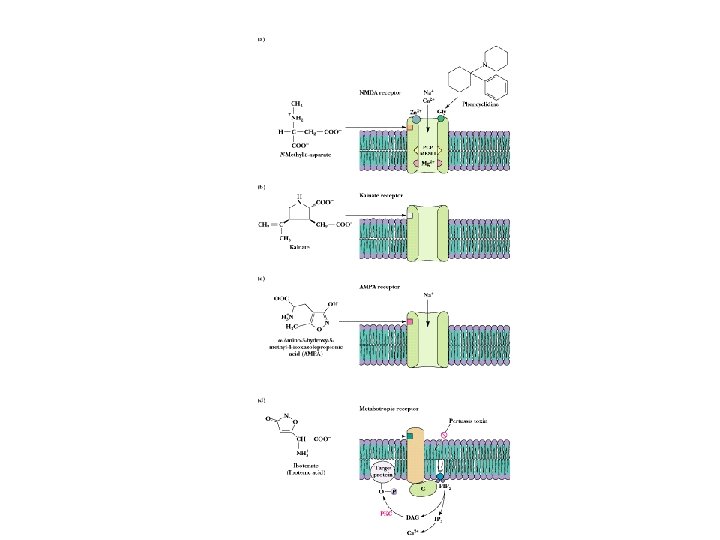
Other Neurotransmitters • • • Excitatory and inhibitory Glutamate is good example: nerve impulse triggers Cadependent exocytosis of glutamate Glutamate is either returned to neuron, or carried into glial cells, converted to Gln and taken back to the neuron from which it was released See 4 types of glutamate receptors in Fig. 34. 68 NMDA receptor is best understood for now Note phencyclidine (angel dust) story

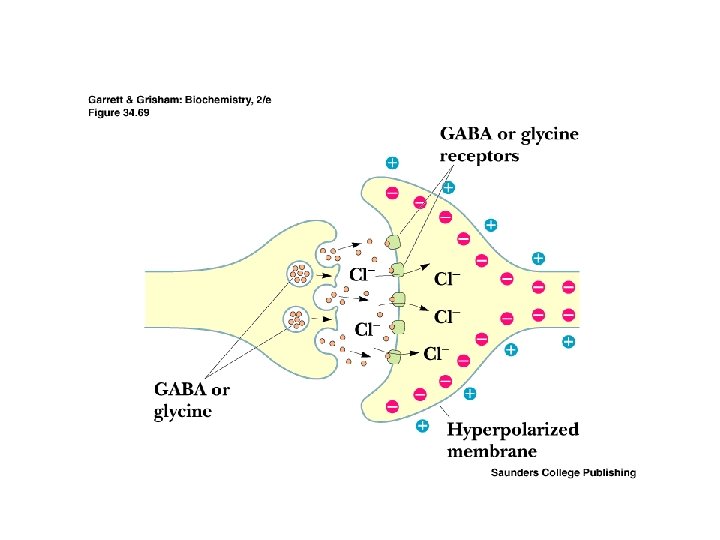
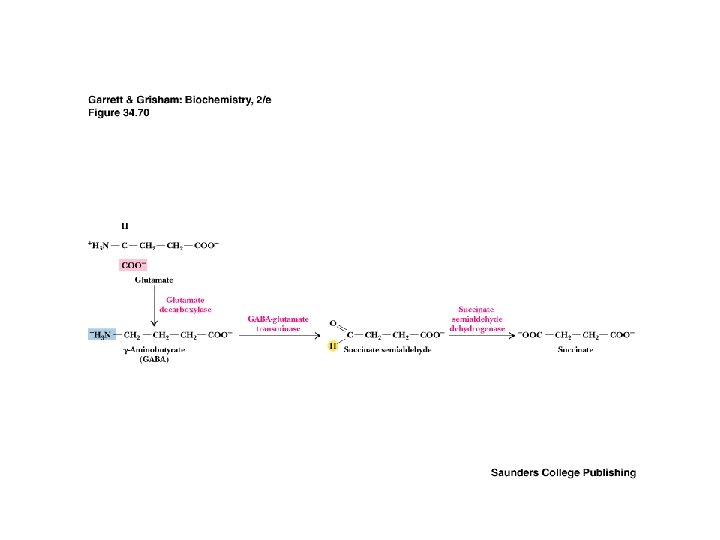
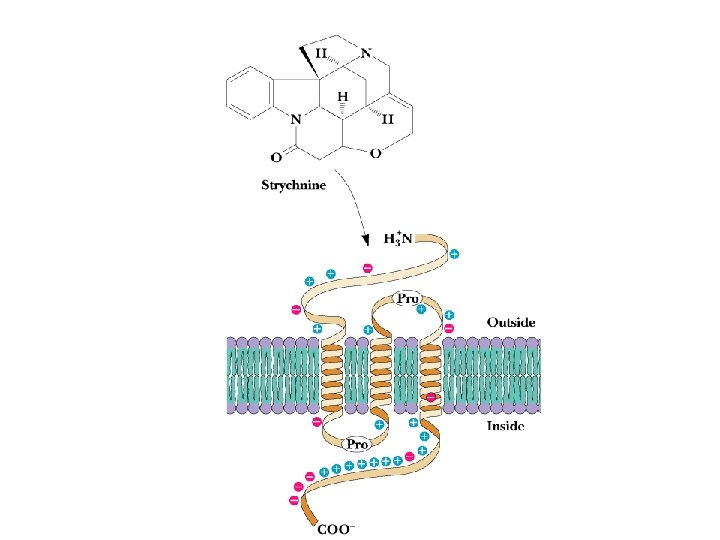
GABA and Glycine • • • Inhibitory Neurotransmitters Inhibitory neurotransmitters diminish the actions of activating neurotransmitters See Figure 34. 70 for glutamate degradation Excitatory glutamate is broken down to inhibitory GABA, which is broken down to non-signals GABA & glycine receptors are chloride channels Glycine receptor is site of action of strychnine

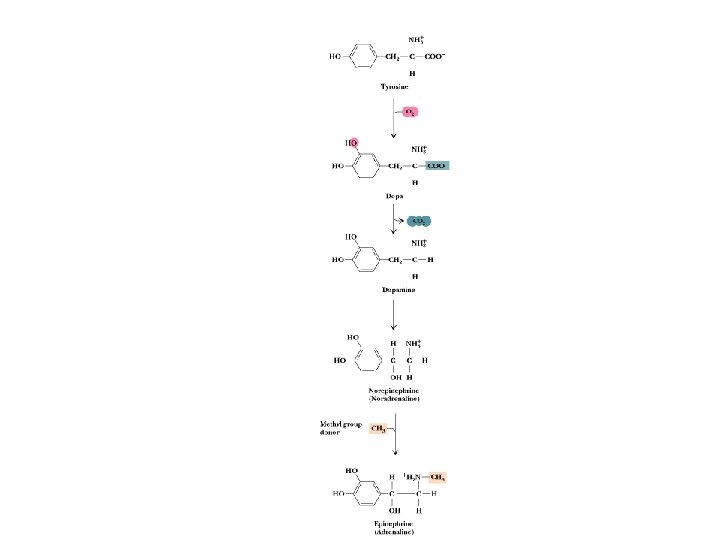
Catecholamine Neurotransmitters • Epinephrine, norepinephrine, dopamine and L-dopa are all neurotransmitters • Synthesized from tyrosine - see Fig. 34. 72 • Excessive production of dopamine (DA) or hypersensitivity of DA receptors produces psychotic symptoms and schizophrenia • Lowered production and/or loss of DA neurons are factors in Parkinsonism

Neurological Disorders • • Depression, Parkinsonism, etc. Defects in catecholamine processing are responsible for many neurological disorders Norepinephrine and dopamine systems are keys Breakdown of NE and DA by catechol-O-methyl transferase and monoamine oxidase Reuptake by specific transport proteins MAO inhibitors are antidepressants Tricyclics - antidepressants that block reuptake Cocaine blocks reuptake, prolongs effects of DA

Peptide Neurotransmitters • • • Lots more to learn here! Likely to be many peptide NTs Concentrations are low; purification is hard Roles are complex Endorphins and enkephalins are natural opioids Endothelins affect smooth muscle contraction, vasoconstriction, mitogenesis, tissue changes Vasoactive intestinal peptide stimulates AC (to make c. AMP) via G proteins, and its effects are synergistic with those of other neurotransmitters
Sensory Transduction • Similarities between sight, taste, smell • Specialized sensory cells translate stimulus into electrical signals in adjacent neurons • Vision is the paradigm system • Absorption of light quanta by rhodopsin isomerizes retinal (11 -cis to all-trans) • Light is absorbed by rhodopsin in the outer segments of rod and cone cells

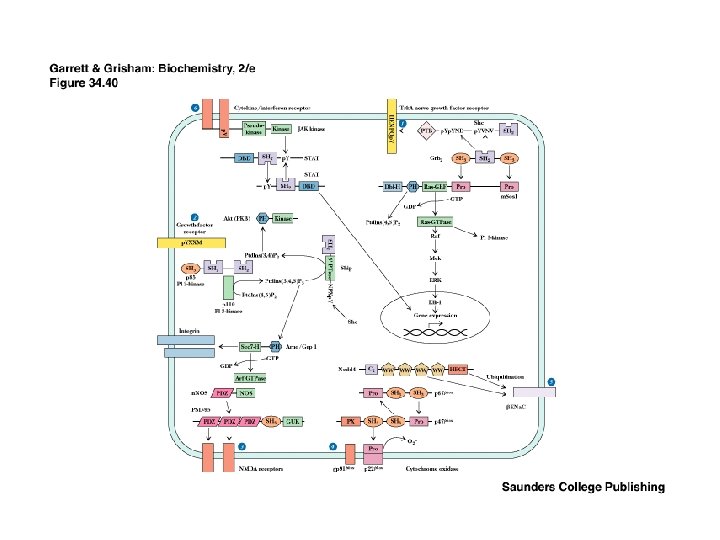
Recap: Signaling Pathways from Membrane to the Nucleus • The complete path from membrane to nucleus is understood for a few cases • Signaling pathways are redundant • Signaling pathways converge and diverge • This is possible with several signaling modules on a signaling protein
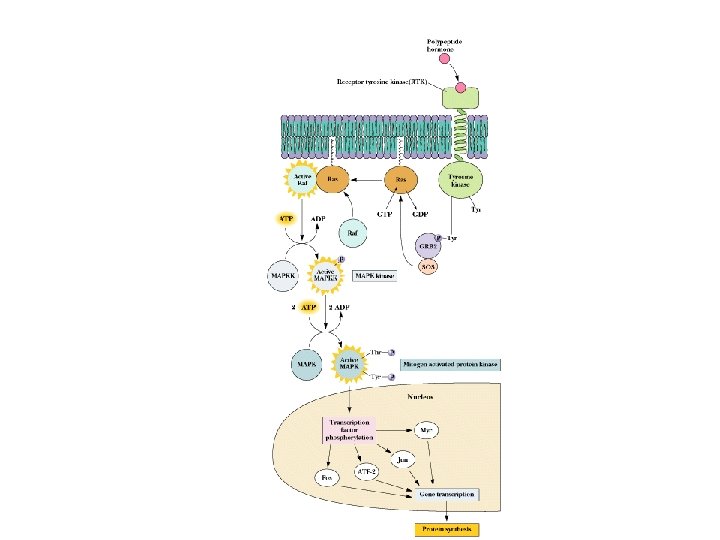
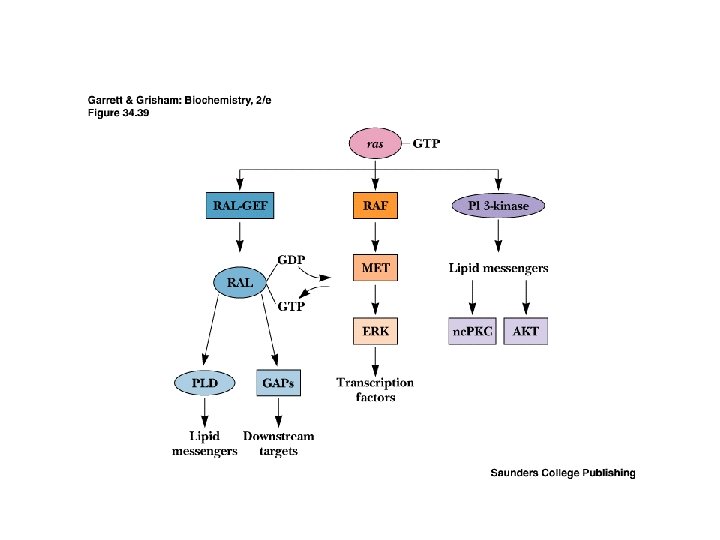

Module Interactions Rule! • The interplay of multiple modules on many signaling proteins permits a dazzling array of signaling interactions • We can barely conceive of the probable extent of this complexity • For example, it is estimated that there are more than 1000 protein kinases in the typical animal cell - all signals!
- Slides: 75