Sigma and Pi Bonds Bonding in Ethylene Bonds

Sigma ( ) and Pi ( ) Bonds
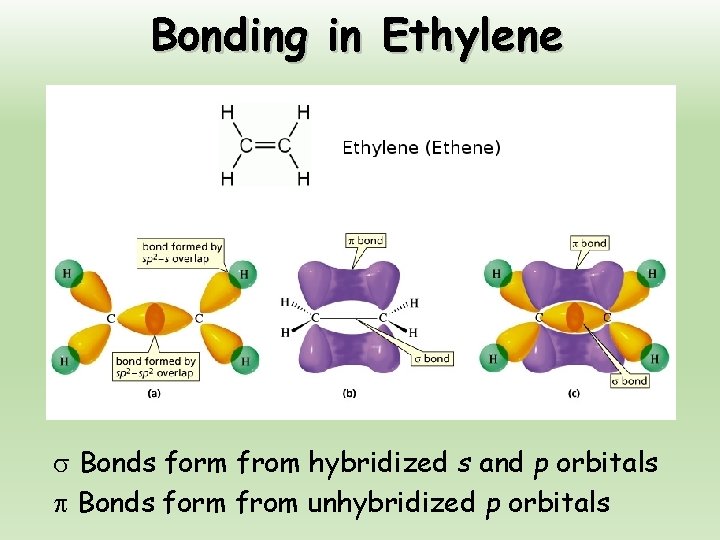
Bonding in Ethylene Bonds form from hybridized s and p orbitals Bonds form from unhybridized p orbitals
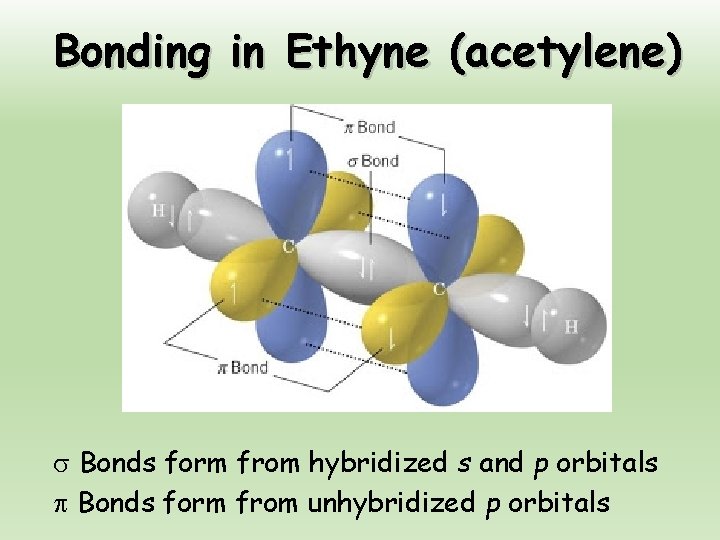
Bonding in Ethyne (acetylene) Bonds form from hybridized s and p orbitals Bonds form from unhybridized p orbitals
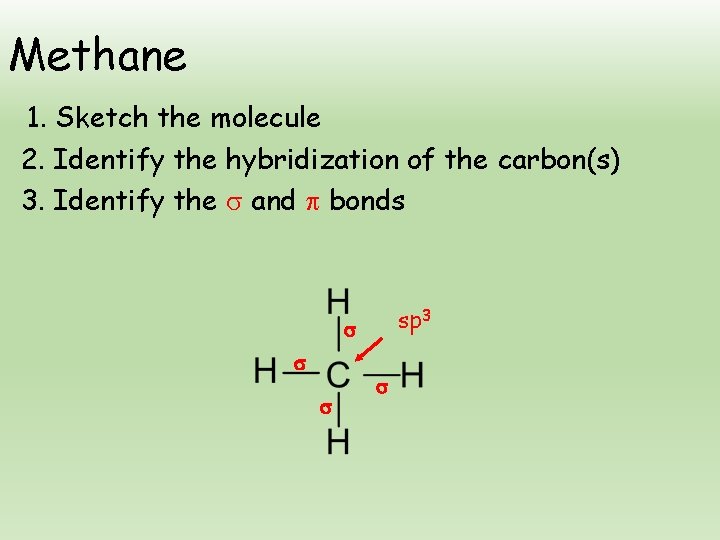
Methane 1. Sketch the molecule 2. Identify the hybridization of the carbon(s) 3. Identify the and bonds sp 3
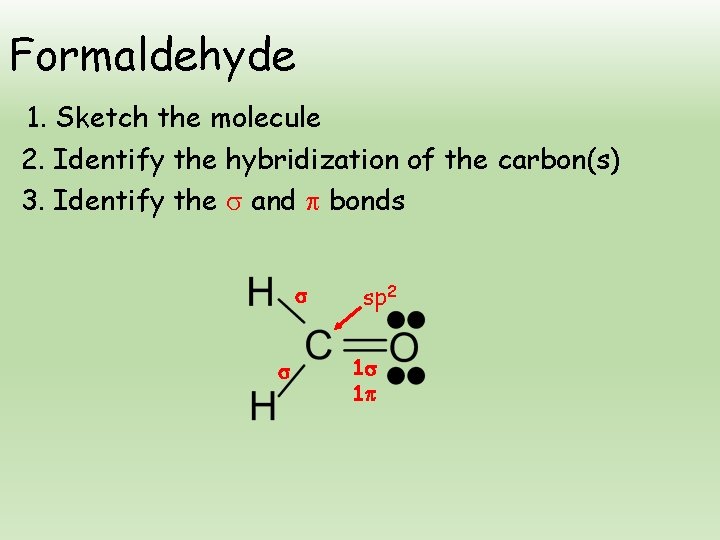
Formaldehyde 1. Sketch the molecule 2. Identify the hybridization of the carbon(s) 3. Identify the and bonds sp 2 1 1
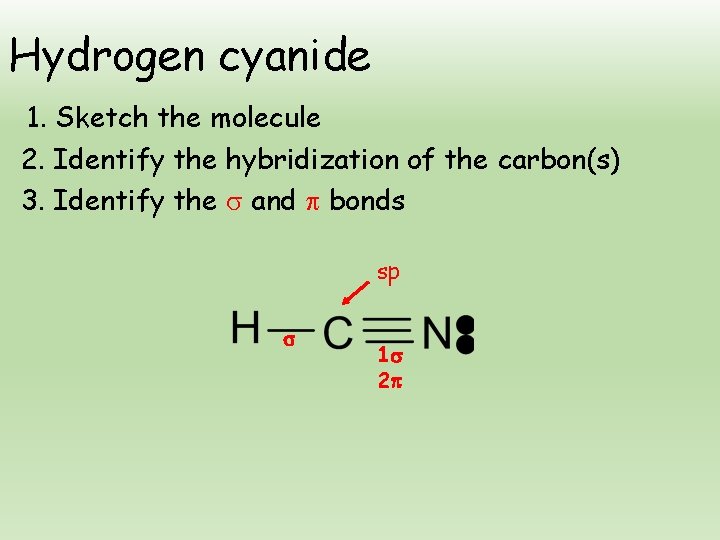
Hydrogen cyanide 1. Sketch the molecule 2. Identify the hybridization of the carbon(s) 3. Identify the and bonds sp 1 2
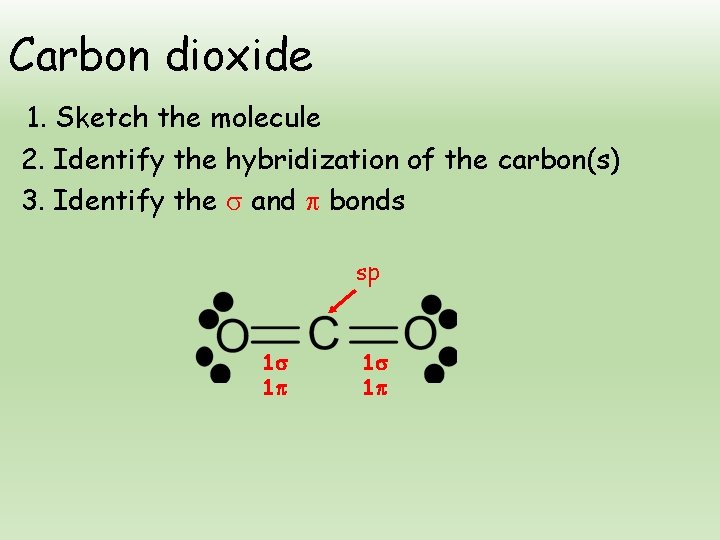
Carbon dioxide 1. Sketch the molecule 2. Identify the hybridization of the carbon(s) 3. Identify the and bonds sp 1 1
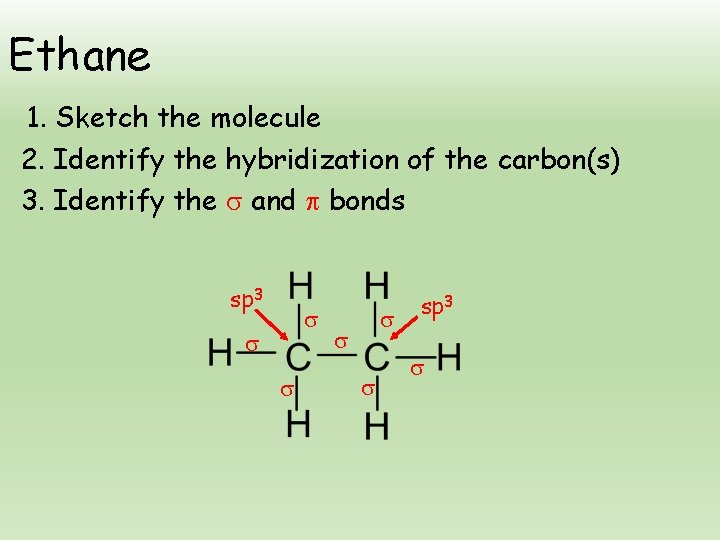
Ethane 1. Sketch the molecule 2. Identify the hybridization of the carbon(s) 3. Identify the and bonds sp 3
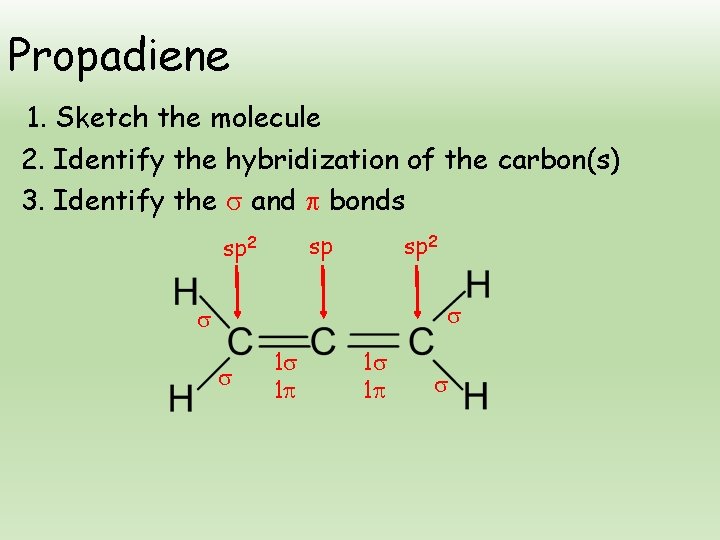
Propadiene 1. Sketch the molecule 2. Identify the hybridization of the carbon(s) 3. Identify the and bonds sp sp 2 1 1
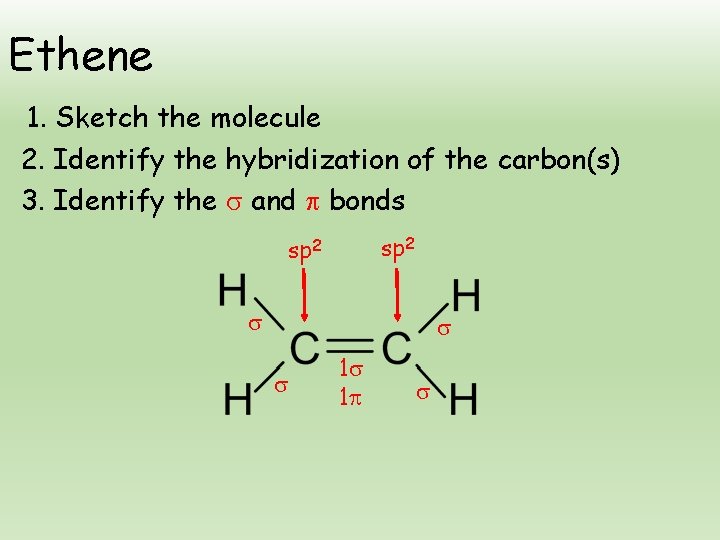
Ethene 1. Sketch the molecule 2. Identify the hybridization of the carbon(s) 3. Identify the and bonds sp 2 1 1
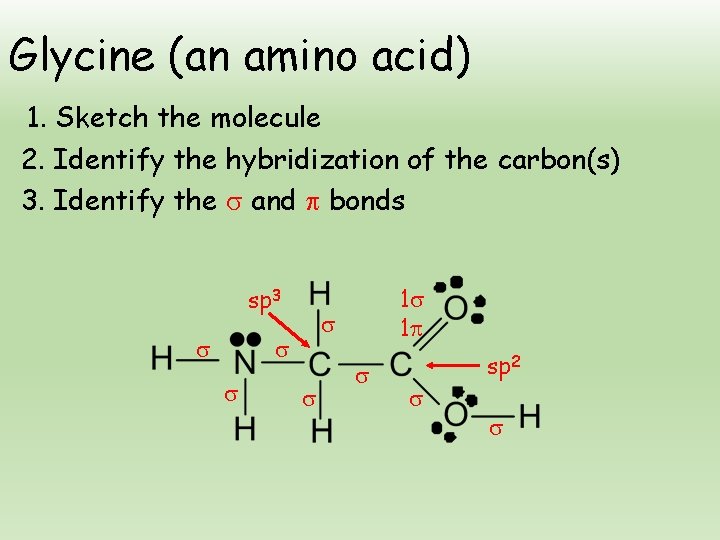
Glycine (an amino acid) 1. Sketch the molecule 2. Identify the hybridization of the carbon(s) 3. Identify the and bonds sp 3 1 1 sp 2
- Slides: 11