Side effects of commonly used glaucoma medications comparisonoftolerability


Side effects of commonly used glaucoma medications: comparisonoftolerability, chance ofdiscontinuation, and patient satisfaction • วารสาร : Graefe’s Archive for Clinical and Experimental Ophthalmology (2008) 246: 1485– 1490 • ผจดทำ : Henny J. M. Beckers, Jan S. A. G. Schouten, Carroll A. B. Webers , Rikkert van der Valk and Fred Hendrikse • แหลงเงนทน : Dutch Health Care Insurance Council

Background • To compare the tolerability of commonly prescribed topical glaucoma medications • determining • frequency and bother of side effects • patient satisfaction with their medication • chance of discontinuation of eye drops.

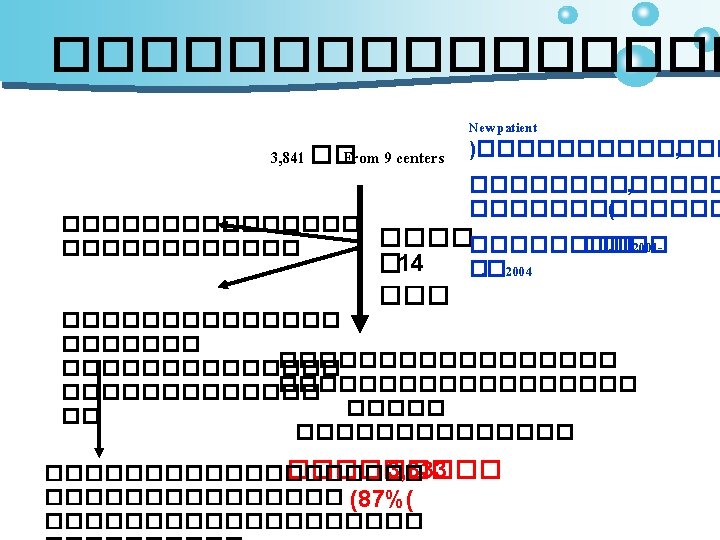
Methods • studied in glaucoma patients from 9 hospitals • The frequency and severity of side effects • patient satisfaction with the medication • probability to change medication due to side effects • To register side effects in a questionnaire based on “the Comparison of Ophthalmic Medications for Tolerability” (COMTOL)

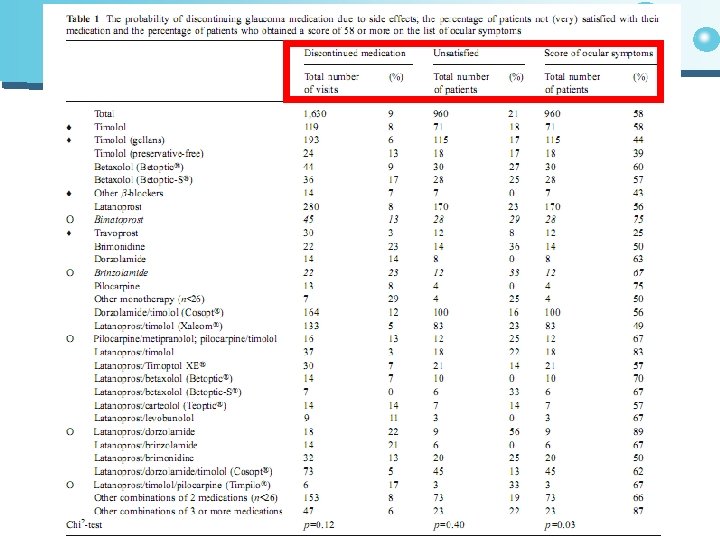
Results • The number of patients responding was 3, 333(87%) 3841/ • Most patients (79%) were satisfied with their eye medication • The median score for ocular side effects was 58 on a scale ranging from 0 to 320 • The probability that medication would be changed by the ophthalmologist was 9% • The most frequently prescribed drugs were • Timolol • latanoprost • dorzolamide/timolol (Cosopt®) • latanoprost/timolol (Xalcom®) Onlysmall differences in tolerability were found between these drugs

Conclusions • Patients are satisfied with their glaucoma medication and have a low chance of discontinuation of eye drops due to side effects.
Add Your Company Slogan ���������� Company Logo



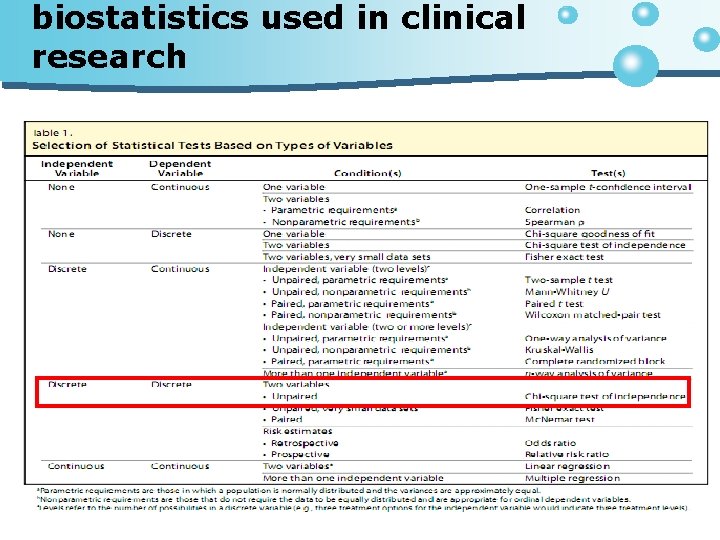
������� • Descriptive • Observational study n Cross-sectional studies

Questionnaire • • • Comparison of Ophthalmic Medications for Tolerability (COMTOL) ���������������������������� Farmacotherapeutisch Kompas ���������� the Dutch Health Care Insurance Board �������������� 2 �� ����������������� subjective data ��������� objective data ������� systemic

Questionnaire The ocular symptoms listed were burning, stinging, conjunctival hyperaemia, itching, ocular secretion, ocular pain, tearing, brow ache, dryness, foreign body sensation, eyelid redness, eyelid oedema, blurred vision, visual acuity loss, accommodation difficulties, and night vision problems

• Side effect frequency of effects 6 -point ‘Did not experience’, ‘One day’, ‘Several days’, ‘About half of days’, ‘Almost every day’ and ‘Everyday’ the intensity of bother 5 -point ‘Not at all bothered’, ‘A little bothered’, ‘Quite bothered’, ‘Much bothered’ and ‘Extremely bothered’. • level of satisfaction 7 -point ‘Very unsatisfied‘, ‘Unsatisfied’, ‘A little unsatisfied, ‘Not unsatisfied nor satisfied’, ‘A little satisfied’, ‘Satisfied’ and ‘Very satisfied’.

����� Questionnaire • �������� • Ocular symtomp n frequency of effects n intensity of bother • ������





biostatistics used in clinical research



Thank you
- Slides: 27