Sickle Cell Anemia Scott Hebert MD April 24
Sickle Cell Anemia Scott Hebert, MD April 24, 2017

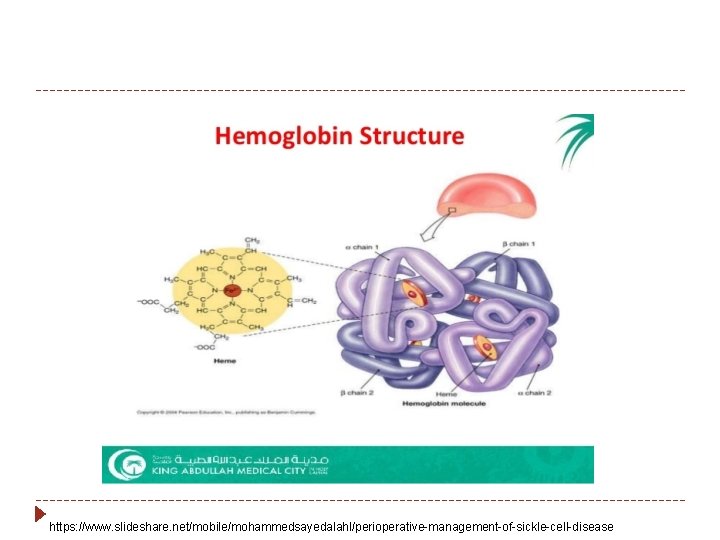
What is SSD? � SSD is an autosomal recessive disorder of a point mutation in the beta globulin chain in hemoglobin.
https: //www. slideshare. net/mobile/mohammedsayedalahl/perioperative-management-of-sickle-cell-disease

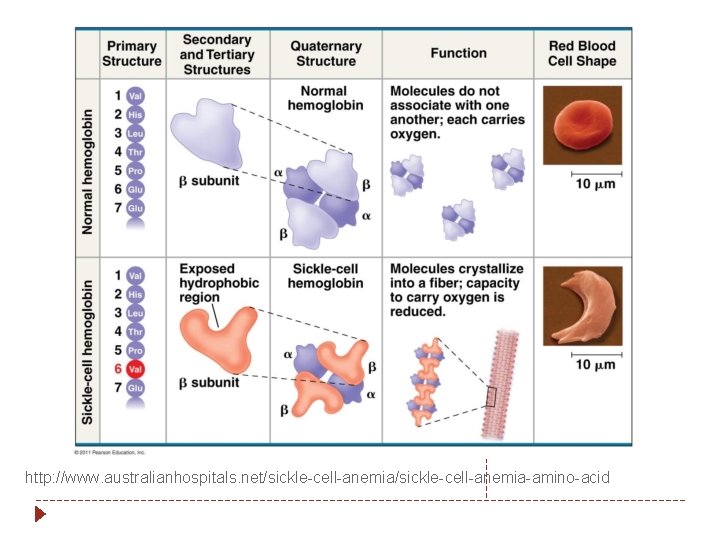
What is SSD? � The point mutation in SSD causes a substitution of valine for glutamic acid as the 6 th amino acid in the beta globin chain {Hb. S (a 2 b. S 2[6 Glu->Val])}. � This results in the tetramer (A 2/B S 2), becoming poorly soluble when deoxygenated, resulting in rope like, crystallized fibers that elongate, decreasing oxygen carrying capacity and cause distortion of the erythrocyte into the crescent or sickle shape.
http: //www. australianhospitals. net/sickle-cell-anemia-amino-acid

� The hallmark symptom of this distortion is a vasoocclusive crisis due to � decreased oxygen access to heme (due to stacking) � sickling of the erythrocyte (microvascular obstruction) � increased adherence to endothelium � free plasma hemoglobin that scavenge nitric oxide (causing vasoconstriction).

Diagnostic testing � Methods of diagnosis of sickle cell disease vary with the age of the patient � Prenatal: � DNA-based testing � chorionic villus sampling at 8 to 10 weeks gestation � PCR based testing currently of interest in vitro

Diagnostic testing � Methods of diagnosis of sickle cell disease vary with the age of the patient � After birth: Separate hemoglobin species according to amino acid composition � hemoglobin electrophoresis- REVIEW PATTERN � thin layer isoelectric focusing (similar concept to electrophoresis, but the proteins migrate to their p. H neutral regions. Patterns based on this. Capable of higher resolution with proteins where a single band can be fractionanted) � solubility testing (Hb. S less soluble and forms crystals/cloudy appearance when cells are lysed then mixed with a reagent) � peripheral blood smear �
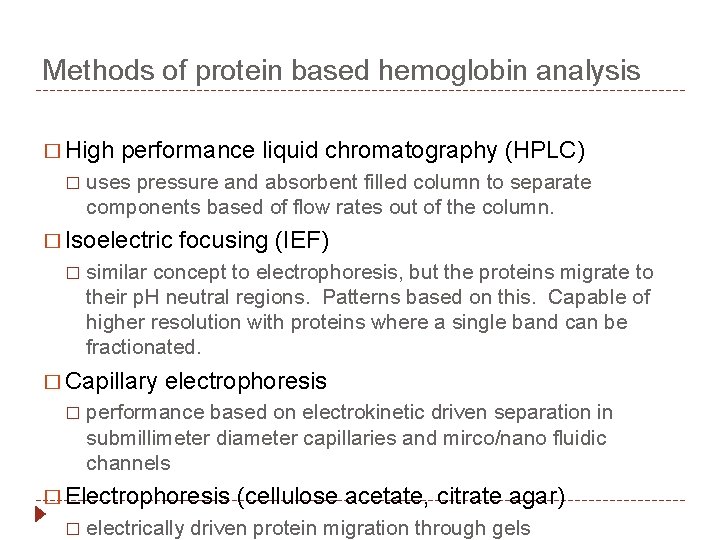
Methods of protein based hemoglobin analysis � High � performance liquid chromatography (HPLC) uses pressure and absorbent filled column to separate components based of flow rates out of the column. � Isoelectric � similar concept to electrophoresis, but the proteins migrate to their p. H neutral regions. Patterns based on this. Capable of higher resolution with proteins where a single band can be fractionated. � Capillary � focusing (IEF) electrophoresis performance based on electrokinetic driven separation in submillimeter diameter capillaries and mirco/nano fluidic channels � Electrophoresis � (cellulose acetate, citrate agar) electrically driven protein migration through gels

Protein based hemoglobin analysis advantages vs disadvantages

Methods of DNA based hemoglobin analysis � GAP � PCR amplification using oligo-primer flanking deletion breakpoints � Traditional � determining precise order of nucleotide within a DNA molecule � Multiplex � DNA sequencing ligation-dependant probe amplification uses forward and reverse probe oligonuclotides that hybridize and ligate together to form one target. Unbound probes are separated out before amplification. � Next-generation � comprehensive analysis of genome � Allele-specific � oligonucleotide testing targets specific mutation using complimentary DNA to the target sequence. � Restriction � DNA sequencing fragment length polymorphism testing measurement of fragments after digestion of DNA samples with restriction endonucleases.

DNA based hemoglobin analysis advantages vs disadvantages
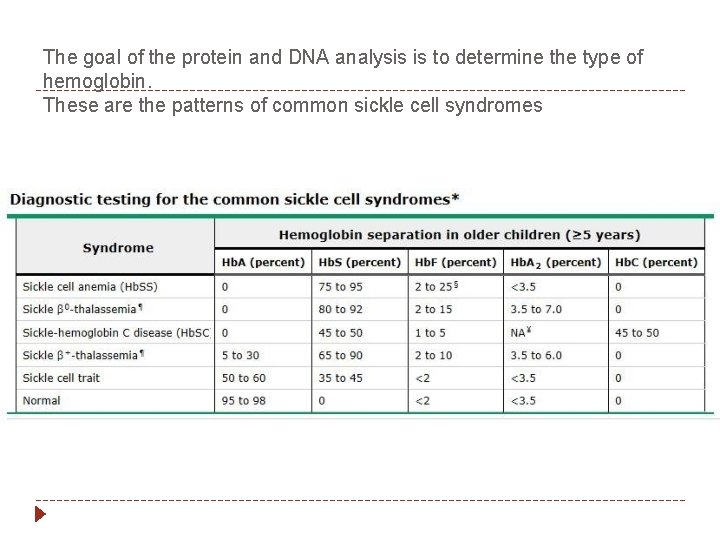
The goal of the protein and DNA analysis is to determine the type of hemoglobin. These are the patterns of common sickle cell syndromes

SSD Presentation � Patients in the adult population will likely present with a history of sickle cell disease as symptoms begin early in life.
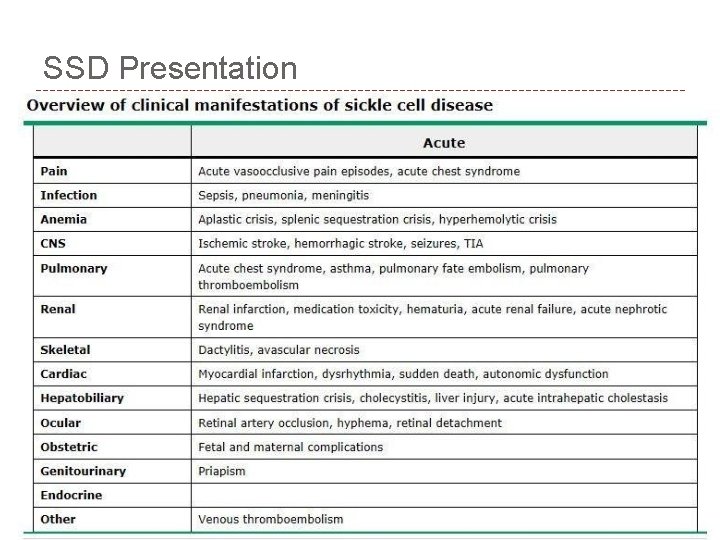
SSD Presentation
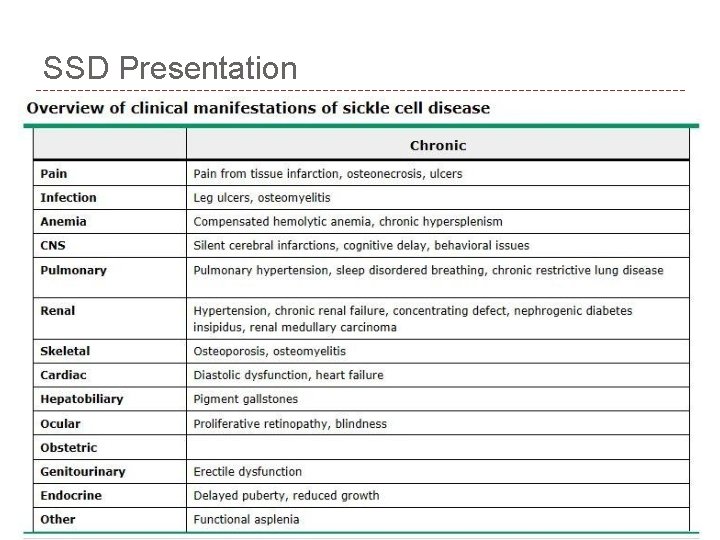
SSD Presentation

SSD Presentation Acute pain crisis � Most common types of vasoocclusive event. Not always a crisis � Pain can be a SCD complication in and of itself or pain can co-occur with/mask other potentially lifethreatening complications � Pain episodes can begin as early as six months of age and typically last throughout life

SSD Presentation Acute pain crisis � Patient’s report is the gold standard for assessment, as there is no combination of physical findings or laboratory tests that can be used to determine/confirm pain. � Placebo should never be used as it undermines the physician-patient relationship and lengthens the duration of pain. � Pain typically is managed by the patient or family at home

SSD Presentation Acute pain crisis � ED presentations usually when pain can no longer be managed at home. � Pain events are commonly associated with: � Acute chest syndrome � Acute multi-organ failure � Sudden death syndrome � Acute surgical abdomen (eg, cholecystitis) � Acute papillary necrosis � Delayed hemolytic transfusion reaction � Acute splenic or hepatic sequestration crises � Opioid withdrawal

SSD Presentation Chronic Pain � Experienced by a large percentage of patients � Mechanisms differs from acute pain and may include � bone and joint damage � chronic ulcers � central sensitization � Hyperalgesia � altered opioid metabolism � Frequent pain may generate feelings of despair, depression, and apathy that interfere with daily life and promote an existence that revolves around pain.

SSD Presentation Infection � Viral infections may be more virulent (eg, parvovirus, H 1 N 1 influenza, Zika virus). � Malaria common cause of morbidity and mortality in children worldwide � Infections mortality are a major cause of morbidity and

SSD Presentation Infection � Mechanisms � functional include hyposplenism/asplenism � increases risk of encapsulated organisms. � reduced tissue perfusion � presence of an indwelling catheter/central line � hypoventilation
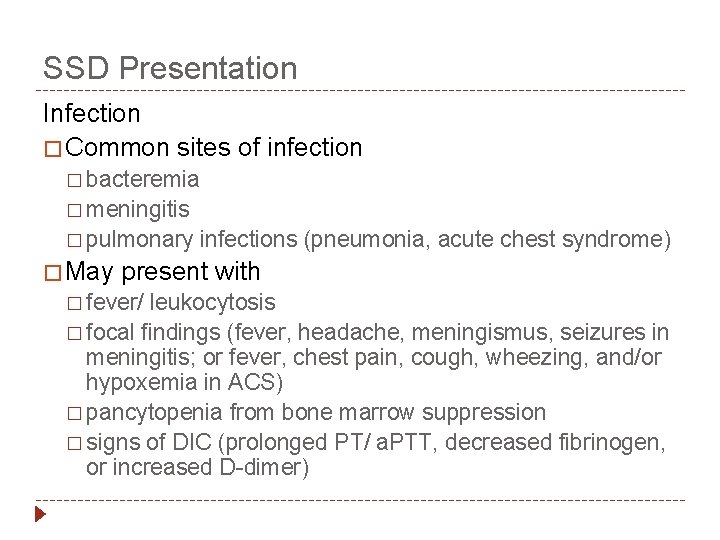
SSD Presentation Infection � Common sites of infection � bacteremia � meningitis � pulmonary � May infections (pneumonia, acute chest syndrome) present with � fever/ leukocytosis � focal findings (fever, headache, meningismus, seizures in meningitis; or fever, chest pain, cough, wheezing, and/or hypoxemia in ACS) � pancytopenia from bone marrow suppression � signs of DIC (prolonged PT/ a. PTT, decreased fibrinogen, or increased D-dimer)

SSD Presentation Multiorgan failure � Life-threatening complication in which multiple organ systems are affected by ischemia and/or infarction. � Typically seen in the setting of an acute painful episode � The mechanism is incompletely understood. � Management with prompt and aggressive exchange transfusion therapy

SSD Presentation Anemia � Chronic, compensated hemolytic anemia with episodes of acute worsening � Contributed by inappropriately low serum erythropoietin and/or folate or iron deficiency. � Major causes of an acute drop in hemoglobin level that are life threatening � aplastic crisis � splenic sequestration crisis � hyperhemolytic crisis

SSD Presentation Chronic compensated hemolytic anemia � Sickled cells undergo hemolysis with a lifespan of approximately 17 days � Compensatory increases in RBC production and adaptation to a lower hemoglobin level � Typical findings hemoglobin 8 to 10 g/d. L � hematocrit 20 to 30 percent � polychromasia and reticulocytosis (retic 3 -15%) � sickled cells, Howell Jolly bodies � mildly increased WBC count � normal RBC indices (unless coexistence of thal/iron def � � Markers may be less severe in pts receiving transfusion therapy or hydroxyurea

SSD Presentation Aplastic crisis � Acute drop in hemoglobin level caused by a transient arrest of erythropoiesis � Managed with transfusion

SSD Presentation Splenic sequestration crisis � Acute drop in hemoglobin 2/2 vasoocclusion within the spleen and splenic pooling of RBCs which can lead to hypovolemic shock and death. � Occurs in spleens that have not yet become fibrotic due to repeated splenic infarction. Infants/sickle variants most often affected. � Rapidly enlarging spleen, decreased hemoglobin despite persistent reticulocytosis � Splenectomy after the first acute event to prevent recurrence

SSD Presentation Neurologic complications � Stroke and TIA � without intervention, 11 percent patients with clinically apparent stroke by 20 yrs, 25% by 45. � ischemic stroke is more children; hemorrhagic more common in adults � primary prevention to reduce the risk of a first stroke based on the use of regular transcranial doppler measurements for risk stratification � previous stroke warrants chronic prophylactic transfusions to prevent recurrent stroke.

SSD Presentation Neurologic complications � Seizures � 2 -3 x more common in SCD � Posterior � less reversible encephalopathy common, can mimic stroke.
SSD Presentation Pulmonary complications � Facilitate sickling with low oxygen tension and low flow � Acute and chronic complications � acute chest syndrome � fever, chest pain, hypoxemia, wheezing, cough, or respiratory distress � cause is often multifactorial and includes infection, vasoocclusion, hypoventilation, atelectasis, thrombosis or thromboembolism, and in some cases fat embolism.

SSD Presentation Pulmonary complications � Management includes analgesia, oxygen, incentive spirometry, bronchodilators, antibiotics, and transfusion � Preventive approaches to reducing the risk of ACS include � prophylactic antibiotics and immunizations � Hydroxyurea � transfusions for recurrent ACS episodes despite hydroxyurea � consideration of hematopoietic cell transplantation for those with an available donor

SSD Presentation Pulmonary complications � Asthma � Sleep disordered breathing � Pulmonary fibrosis � Thromboembolic disease � Pulmonary hypertension � 6 to 10 percent of individuals with SCD � screening q 1 -3 yrs

SSD Presentation Skeletal complications � Dactylitis in infants/children � Osteoporosis � Avascular necrosis � Osteomylitis

SSD Presentation Cardiac complications � Cardiomyopathy � p. HTN � chronic anemia � iron overload, � HTN and heart failure due to

SSD Presentation Hepatobiliary complications � Acute ischemia � Cholestasis � Hepatic sequestration crisis � Transfusional iron overload � Pigment gallstones � Hepatitis C virus

SSD Presentation Leg ulcers � Due to compromised blood flow, endothelial dysfunction, thrombosis, inflammation, and delayed healing � More common in males than females � Post traumatic, potentially related to superinfection � Preventative strategies important

SSD Presentation Retinopathy � Annual screening after age 10

SSD Presentation Pregnancy complications � Fetal � intrauterine growth restriction, fetal death, and low birthweight � Maternal � acute chest syndrome, infections, preeclampsia, and thromboembolic events � Management � reproductive counseling, birth control, folic acid, screening for bactiuria and fetal growth, cord blood harvesting, prophylactic transfusion, postpartum anticoagulation in cesarean delivery

SSD Presentation Priapism � Common, serious, and under diagnosed

SSD Presentation VTE � Hypercoaguable state � Thromboprophylaxis in hospitalized patients

SSD Presentation Growth and development � Frequently underweight with normal height � Often delayed puberty

SSD Presentation Psychosocial issues � Most are well adjusted � Increased risk of chronic medical condition promoting � low self-esteem � social isolation � poor family relationships � withdrawal from normal daily living due to inappropriate pain coping strategies, reduced quality of life, anxiety, depression, and neurocognitive impairment

Management � Regular clinician follow up � Prevention of complications with early penicillin prophylaxis started in the newborn period, appropriate immunizations, and blood transfusions for those at risk for stroke.

Management: Hydroxyurea � used to reduce sickle hemoglobin by increasing the production of fetal hemoglobin � Increased Hb. F production up to 2 fold via shift in gene expression at the beta globin locus. � Effects: � reduce sickeling � increased RBC lifespan � improved RBC hydration � decreased hemolysis and vascular endothelium adhesion � improved microcirculation blood flow � decreased vasooclusive events.

Management: Hydroxyurea � Other mechanisms: � decreased hemolysis = less nitric oxide scavenging and vasodilation � decrased density with more mature RBC’s and easier passage through microcirculation � decreased WBC count and neutrophil adhesiveness decreasing inflammatory pain � Hydroxyurea should be started in symptomatic patients � Baseline Hb. F should be measured then q 2 -3 m during titration

Management: Hydroxyurea � initial dosing and monitoring oral dose with a creatinine clearance >60 m. L/min is 15 to 20 mg/kg per day. � increase dose every 8 weeks, max dose 35 mg/kg daily or 2500 mg daily � adjust dose based on Maximal Tolerated Dose (MTD)

Management: Hydroxyurea � MTD parameters � ANC >1500 -3000/micro. L � platelet count >80, 000/micro. L � hemoglobin >8 g/d. L off transfusion � reticulocyte cocunt >80, 000 -100, 000/micro. L

Management: Hydroxyurea � Adverse effects � Myelosuppression � GI upset (direct pt to take at night) � oral ulcers � Hyperpigmentation � Rash � nail changes � Hydroxyurea can be continued during admission, but does not need to be initiated while hospitalized

Management: Hydroxyurea � Contraceptive � Discontinue � Risk counseling required 3 months prior to pregnancy vs benefit during breast feeding � Males should discontinue 6 m prior to conception

Management: Hydroxyurea � Lack of response � carefully assess compliance � failure to increase Hb. F or MCV >100 � 20 -30% adults with true lack of efficacy � no benefit in continuing therapy in true lack of response

Management Preventative measures � � Hydroxyurea � Appropriate vaccinations with PCV 13 � PPSV 23 � Influenza � Meningococcus, � standard childhood vaccinations, including those against hepatitis A and B; measles, mumps, and rubella; varicella; rotavirus; Haemophilus influenzae; tetanus, diphtheria, and pertussis; and poliovirus in countries where it is still endemic � �

Management � Screening for HTN, cerebral complications, retinopathy, lung disease, p. HTN, renal disease, birth control, family planning, leg ulcers, bone health, � Infection management: early follow up if febrile, empiric abx, blood cultures � Nutrition � folic acid, MTV, Vit D

Management: Transfusion Rational for transfusion � Dilution of Hgb. S with Hgb. A blood � Suppression of erythropoietin release thus reducing the production of new Hgb. S-containing cells � Decreased percentage of Hgb. S-containing cells due to longer lifespan of Hgb. A-containing cells � Increase in Hgb oxygen saturation levels by approximately 1 to 6 percent, which increases oxygen delivery to the tissues

Management: Transfusion Indication for transfusion � Acute therapeutic Transfusions used acutely for the treatment of vasoocclusive phenomena accompanied by severe anemia that cannot be adequately compensated by increased red cell production. � Acute stroke � Acute chest syndrome � Acute multi-organ failure � Acute symptomatic anemia � A drop in baseline reticulocyte count � Hepatic or splenic sequestration �

Management: Transfusion � Prophylaxis � Reduces perioperative complications in patients undergoing surgery and reduces the incidence of vasoocclusive complications. � Transfusions used acutely for the treatment of vasoocclusive phenomena accompanied by severe anemia that cannot be adequately compensated by increased red cell production.

Management: Transfusion � Indications for Prophylactic transfusions: � Stroke � Silent cerebral infarcts in children with Hb. SS or Hb. Sbeta 0 thal � Recurrent acute chest syndrome despite hydroxyurea therapy � Vasoocclusive pain episodes that are severe, frequent, and not responsive to maximum tolerated doses of hydroxyurea � Recurrent priapism � Pulmonary hypertension with progressive clinical symptoms/increasing PA pressure that has not benefitted/candidate for hydroxyurea

Management: Transfusion Simple vs exchange transfusion �Simple blood transfusion � uncomplicated drop in Hgb levels resulting in signs or symptoms of decreased oxygen delivery, where the immediate need is to restore oxygen carrying capacity rather than to decrease sickling � If the Hgb is <5 g/d. L and the patient is critically ill, simple transfusion can be used with a goal to increase the Hgb level to 10 to 11 g/d. L. � Preoperative transfusion to reduce complications of surgery

Management: Transfusion Simple vs exchange transfusion �Simple blood transfusion �Consider � Hgb avoiding if is near baseline (simple transfusions do not rapidly reduce the percentage of Hgb. S cells) � Risk of hyperviscosity syndrome

Management: Transfusion Simple vs exchange transfusion � Exchange blood transfusion � For acute emergencies, when the patient is acutely ill and deteriorating quickly (eg, multi-organ failure, suspected stroke, respiratory compromise, acute chest syndrome) � For regularly scheduled transfusions used in the prevention of stroke, acute chest syndrome, and recurrent painful episodes. � In general, automated apheresis is preferred over manual exchange because it can be done faster and causes fewer volume shifts [ � acute illness: consider lowering the Hgb. S level into the range of 15 to 20 percent, and raising the total Hgb to the range of 10 to 12 g/d. L. � avoids positive iron balance

Management Acute pain management �Adequate, early, aggressive treatment of pain is essential �Every patient with should have � established, individualized plan for treating pain, � education, medication, and contact info for scripts and uncontrolled pain symptoms �Assessment of pain quality and intensity

Management Acute pain management �Failure to adequately assess/appreciate the intensity/causes of pain may lead to unnecessary withholding of opioid analgesics, possibly worsen pain and coping mechanisms �Trust between the patient and clinicians providing care is essential for management. �Interventions to minimize and treat opioid complications: constipation, sedation/hypoventilation, pruritus
Management Chronic pain � Treatment of acute and chronic pain overlap � Hydroxyurea to reduce frequency of vasoocclusive pain episodes � Inhibation of mast cell activation using imatinib, or elimination of mast cells by genetic manipulation, both reduced nociceptive pain and decreased pathologic measures of tissue inflammation. � Treatment with the mast cell stabilizer cromolyn increases the analgesic efficacy of low-dose morphine in treating hypoxia-induced vasoocclusive pain. � APAP, NSAIDS, short and long acting opioids

Management: Hospitalization � Hydration � Pain management � Management of acute event (infection, infarction, etc) � Oxygen � Diphenhydramine � Antiemetics, bowel regimen � Mass cell stabilizers (cromolyn, 5 -HT 3 receptor antagonist) � Simple vs exchange transfusion � Thromboembolism prophylaxis � Continuation of hydroxyurea � Pruritis: continuous infusion of low-dose naloxone (0. 25 mcg/kg per hour) has been very helpful in decreasing pruritus without requiring opioid adjustment � Do not use meperidine or ketorolac due to potential toxicities and lack of additional benefit

Management Hematopoietic cell transplantation � Potentially curative � Used most often in patients <16 � Barriers: clinical course of the disease is quite variable � Patient eligibility is limited because of the presence of advanced stage symptomatic disease, often involving severe pulmonary and neurologic vasculopathy. � Serious concerns about transplantation-related mortality, as well as the potential for treatment-induced malignancy �

Sickle cell disorders � cconditions in which the patient carries at least 1 Hb. S gene mutation. � Hb. SC disease � Sickle-Beta thal � Sickle- Alph thal � Sickle – fetal Hb � Sickle- Delta Beta (0) thal � Sickle – Hb lepore � Sickle- Hb. D � Sickle Hbo Arab � Sickle Hb. E

Sickle cell disorders: Hb. C disease: � Substitution of lysine for glutamic acid at 6 th amino acid in beta globin chain {Hb. C (a 2 b. C 2[6 Glu->Lys])}. � Similar to Hb. S, the Hb. C is less soluble. � Hb. C causes crystal formation potentially leading to increased blood viscosity and cellular rigidity, shortening red cell survival. � Hb. C does not cause sickling of cells alone, but in Hb. SC disease, sickling happens less severely than in Hb. SS. � Anemias and reticulocytosis are more mild due to extended time in circulation. Hb. SC 27 days, Hb. SS 17 days. � Less pain episodes � Lower rate of focal segmental glomerulosclerosis � Lower rate of leg ulcers
Sickle cell disorders: Hb. C � Later development of ostenecorsis � Life expectancy 64 vs 45 � Less pregnancy complication � Higher incidence of peripheral retinopathy (possibly due to higher hematocrit) � Functional asplenia occurs in 45% Hb. SC. Persistent splenic function means that splenic infarction and splenic sequestration crisis can happen at any age.
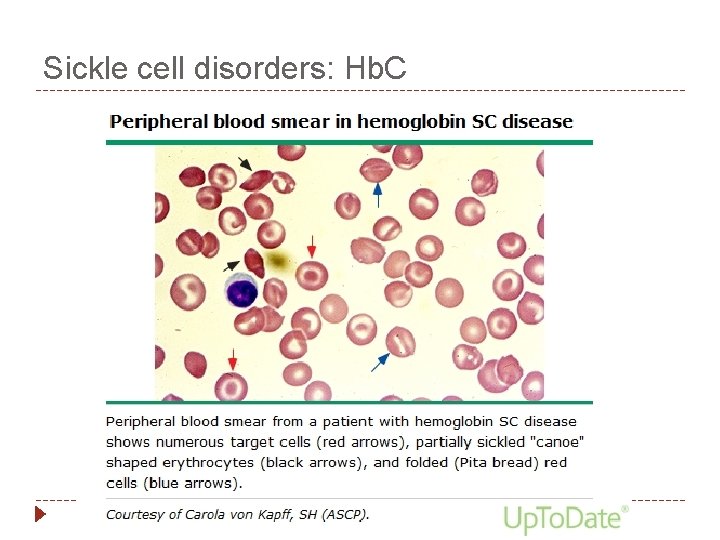
Sickle cell disorders: Hb. C

Sickle cell disorders Sickle-Beta Thalassemia � Thalassemia: spectrum of diseases characterized by reduced or absent production of one or more globin chains. � Beta Thal: impaired production of beta globin chains, leading to relative excess of alpha globin chains. � Incidence in African Americans 0. 004. � Divided into sickle cell-beta 0 thalassemia and sickle cell-beta+ thalassemia � based upon the complete absence of beta globin or the presence of reduced amounts of beta globin, determining level of Hb. A

Sickle cell disorders Sickle-Beta Thalassemia � Clinical manifestations — Severity inverse proportional to quantity of Hb. A � Sickle cell-beta 0 thalassemia (ie, no Hb. A production) have a clinical course as severe as Hb. SS � Often no not undergo rapid splenic infacrction and have enlargement into adulthood, causing continued risk of acute sequestration crisis � Sickle cell-beta+ thalassemia: more benigh course due to low Hb. A production

Sickle cell disorders Sickle-Alpha Thalassemia (-a/aa) or (-a/-a) � Impared alpha globin chain, leading to excess beta globin chain � Clinical manifestations generally less severe � Milder anemia than SCD � Fewer sickle forms

Sickle cell disorders Sickle-hereditary persistence of Fetal Hb � Pancellular: 10 -40% Hb. F expression � Hypocellular: up to 5% � 10 -20% Hb. F expression is suggested as the threshold for diminished clinical severity

Sickle cell disorders Sickle-Delta beta Thalassemia � Hb. S, F, A 2 � 15 -25% Hb. F � Anemia and reticulocytosis are mild � Clinical complications are infrequent
Sickle cell disorders Sickle-Hb Lepore disease � Crossover fusion produce of delta and beta globin genes � Resemble sickle cell traid on electrophoresis with 12% mutant hemoglobin present � Microcytosis, hypochromia, irreversibly sickle cells � Vasoocclusive complications and splenomegaly common � More mild than sickle cell beta thal

Sickle cell disorders Sickle-Hb. D Disease � Glutamic acid to glutamate substitution at codon 121 of beta globin � Similar to SCD, but with persistent splenomegaly being more common � Electrophoresis looks like SCD. Isoelectric focusing can separate distinguish from SCD
Sickle cell disorders Sickle-Hb. O Arab disease � Hemoglobin O: glutamic acid to lysine substitution at codon 121 of beta globin � Resembles Hb. C on electrophoresis. � Moderately severe hemolytic anemia, anisocytosis, poikilocytosis, irregersibly sickle cells � Decreased oxygen affinity compared to SCD � Similar clinical manifestations to SCD

Sickle cell disorders Sickle- Hb. E disease � Hb. E glutamic acid to lysine substitution at codon 26 of beta globin � Mild hemolysis, no vasooclusive complications, no remarkable red cell morphology � Reported cases of splnic infarct with air travel, hematuria, acute chest, reversible bone marrow from parvo

Clinic To Do list � � � � Pneumococcal. 13 then 23. 23 q 5 yrs Annual ophtho exam Bone healt: Ca++d, q 3 yr dexa. Vit D: Screening echo for p. HTN Folic acid Last transfusion: Cumulative transfusions: Baseline h/h: Ferritin: Pain regimen: Pain contract: Last admission: Osteonecrosis screening: Stem cell transplant? Annual transcranial Doppler HCV screening
- Slides: 79