Shell Tsinghua Chair Professorship Natural Gas Hydrates Jakob

Shell – Tsinghua Chair Professorship Natural Gas Hydrates Jakob de Swaan Arons Professor Royal Dutch Shell Chair Chemical Engineering Department Tsinghua University, Beijing, China 19 th September 2006

Royal Dutch Shell Transport and Trading Company (British) and Royal Dutch Petroleum Company (Dutch)

Contents What are gas hydrates Ø Models, thermodynamics and phasebehavior Ø Applications Ø Conclusions Ø
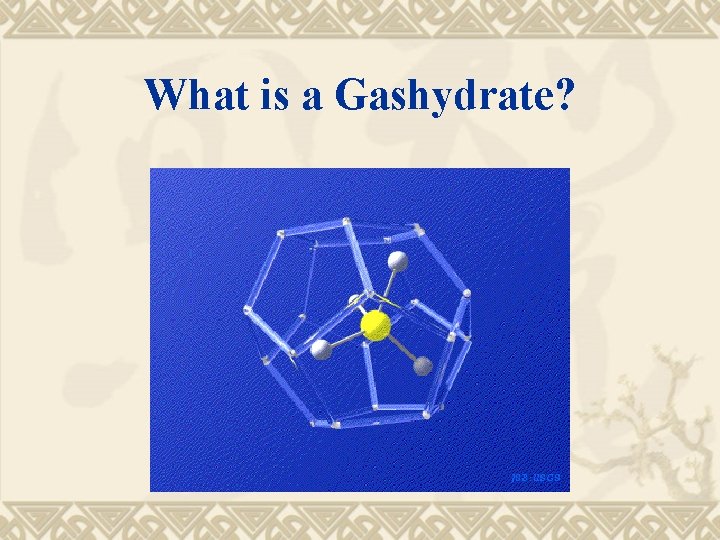
What is a Gashydrate?

Gashydrates Crystalline structures of water with cavities of molecular size, containing (hosting) molecules of compounds (guest) with boiling points mainly below and sometimes above room temperature.
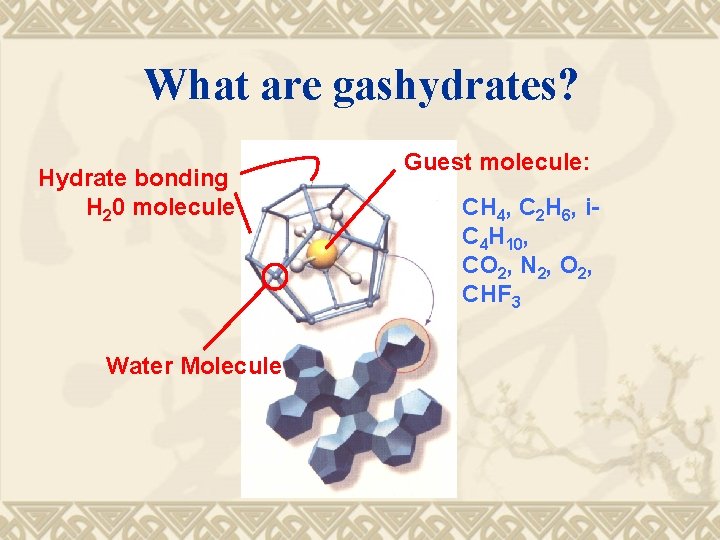
What are gashydrates? Hydrate bonding H 20 molecule Water Molecule Guest molecule: CH 4, C 2 H 6, i. C 4 H 10, CO 2, N 2, O 2, CHF 3
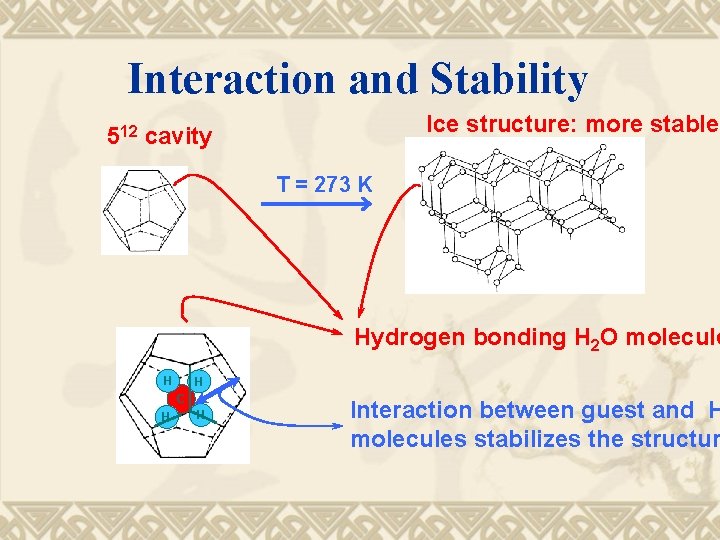
Interaction and Stability Ice structure: more stable 512 cavity T = 273 K Hydrogen bonding H 2 O molecule H H C H H Interaction between guest and H molecules stabilizes the structur
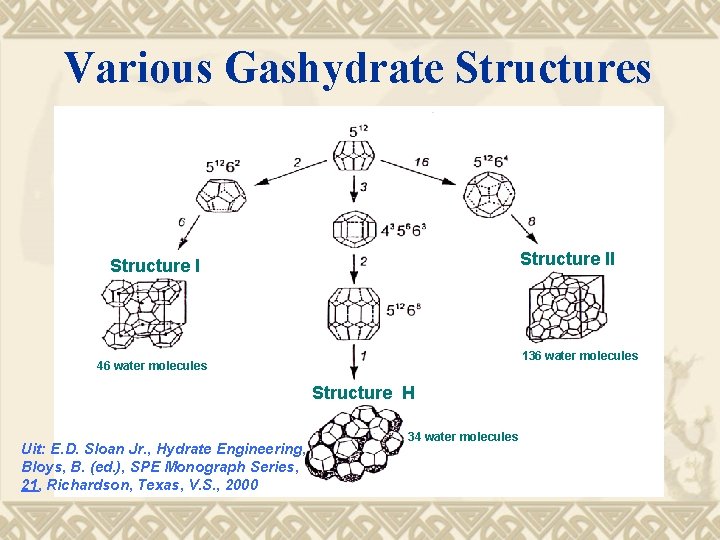
Various Gashydrate Structures Structure II Structure I 136 water molecules 46 water molecules Structure H Uit: E. D. Sloan Jr. , Hydrate Engineering, Bloys, B. (ed. ), SPE Monograph Series, 21, Richardson, Texas, V. S. , 2000 34 water molecules
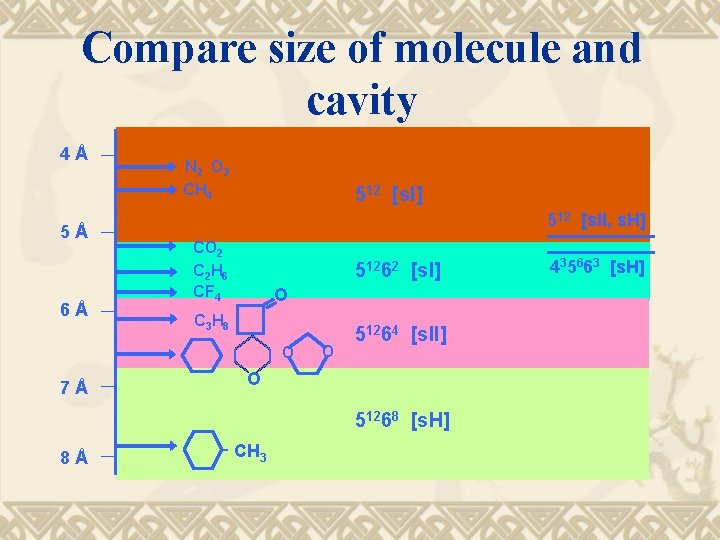
Compare size of molecule and cavity 4Å 5Å 6Å N 2 O 2 CH 4 512 [s. I] 512 [s. II, s. H] CO 2 C 2 H 6 CF 4 51262 [s. I] O C 3 H 8 O 7Å O 51264 [s. II] O 51268 [s. H] 8Å CH 3 435663 [s. H]

Gashydrate flame

Importance Ø Ø Nuisance Blessing ? Separations Scientific
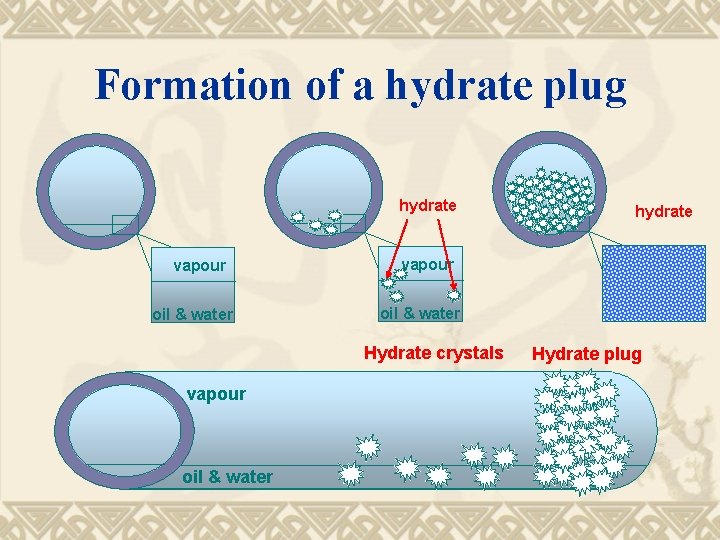
Formation of a hydrate plug hydrate vapour oil & water Hydrate crystals vapour oil & water hydrate Hydrate plug

They resemble ice, we find them in Nature. Gas hydrate

Natural gas hydrate reservoirs Blake Ridge Barents Zee Noorse Zee Hydrate Ridge Zee van Okhotsk Prudhoe Bay Mc. Kenzie Delta Other 3. 780 Fossil 5. 000 Hydrate 10. 000 Locates Gas Hydrate: Zeebodem Permafrost K. A. Kvenvolden, A Primer on the Geological Occurrence of Gas Hydrate, in: Gas Hydrates – Relevance to World Margin Stability and Climate Change, Henriet, J. -P. , Mienert, J. (eds. ), Geol. Soc. Special Publ. , 137, Geological Society, Londen, GB, p. 9 -30, 1998
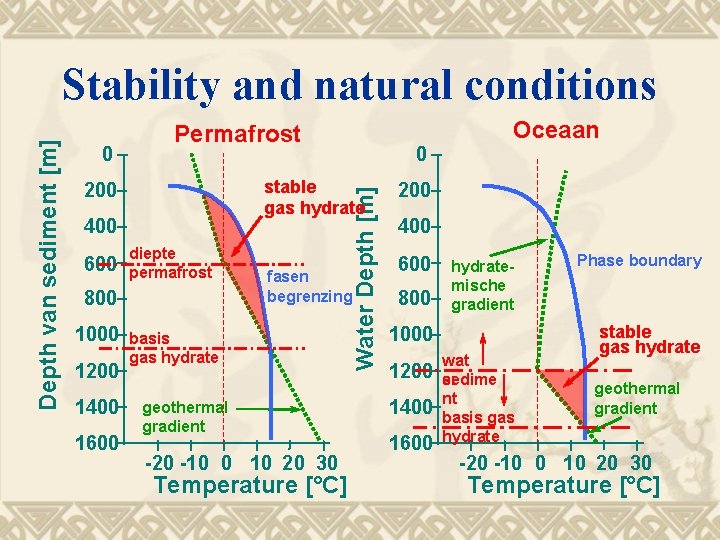
0 Permafrost stable gas hydrate 200 400 600 diepte permafrost 800 1000 1200 1400 1600 0 Water Depth [m] Depth van sediment [m] Stability and natural conditions fasen begrenzing basis gas hydrate geothermal gradient -20 -10 0 10 20 30 Temperature [°C] Oceaan 200 400 600 800 hydratemische gradient 1000 1200 1400 1600 wat er sedime nt basis gas hydrate Phase boundary stable gas hydrate geothermal gradient -20 -10 0 10 20 30 Temperature [°C]

Industrial question Dutch natural gas may contain up to 14% N 2. Could hydrates act as a good separation agent?

Scientific importance As we will see later, gas hydrates offer an extremely interesting example of a large family of so called inclusion compounds made up of host- and guest- molecules. Water l Urea l Hydroquinone l

Question What has the subject of Natural Gas Hydrates (NGH) to do with a course in Advanced Chemical Engineering Thermodynamics?

Answer In dealing with NGH we can demonstrate the power and beauty of Applied, Molecular and Statistical Thermodynamics.

Classical thermodynamics Presents broad relationships between macroscopic properties but it is not concerned with quantitative prediction of these properties. Example John M. Prausnitz

Statistical thermodynamics Seeks to establish relationships between macroscopic properties and intermolecular forces and other molecular properties. Example John M. Prausnitz

Molecular thermodynamics Seeks to overcome some of the limitations of both classical and statistical thermodynamics. It is an engineering science, based on classical thermodynamics but relying on molecular physics and statistical thermodynamics ……. . In application it is rarely exact and has an empirical flavour. John M. Prausnitz

Van der Waals – Platteeuw model for gashydrates These former colleagues at Shell Research International developed a wonderful model, back in the 1950 -ies, that since then has seen many small modifications but still “stands as a rock”. Johan van der Waals and Joost Platteeuw Adv. Chem. Phys. 2, 1 -57 [1959]

Assumptions for the model 1. 2. 3. 4. 5. Guest molecules don’t affect the cavity structure At most one guest molecule/cavity No interactions between guest molecules Guest molecule can rotate freely in cavity Lennard- Jones type potential for interaction between guest and cavity
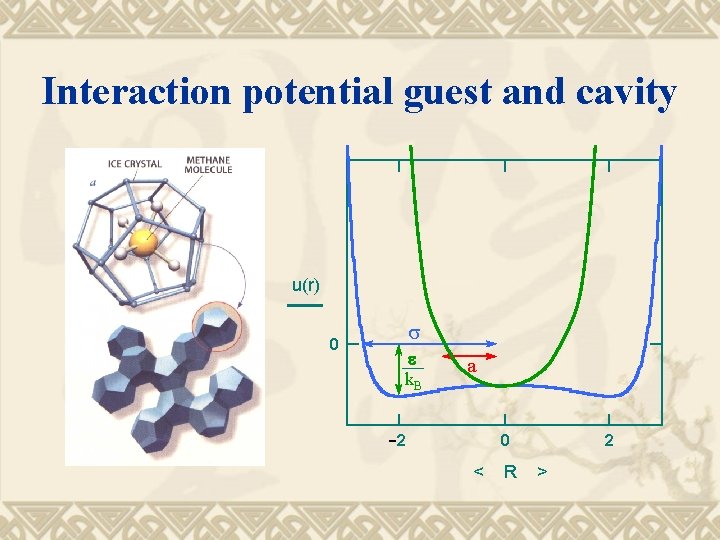
Interaction potential guest and cavity u(r) 0 s e k. B a 2 0 < R 2 >

Intermolecular potential (1) a = radius guest R = radius cavity r = variable distance from center cavity = r value for which potential is 0 = potential at maximum attraction

Intermolecular potential (2) The most successful potential has been proposed by the Japanese scientist Kihara. Its parameters have been optimized from experimental data on hydrate phase equilibria.

The “ Langmuirconstant” This constant can be expressed for guest k in cavity of type i by

Guest k in cavity i expresses the fraction of cavity type i occupied by guest k. In case of CH 4 the fugacity is approximately the total pressure P

Cavity occupancy and host water “Langmuir” “Raoult” In case of water CH 4 : the higher the gas pressure, the higher the cavity occupancy, the more stable the hydrate vi= number of cavities type i

Analogies The equations for thermodynamic potential or fugacity of solutes (guests) and solvent (host) show a remarkable resemblance with those for adsorption (Langmuir) and solvency (Raoult).
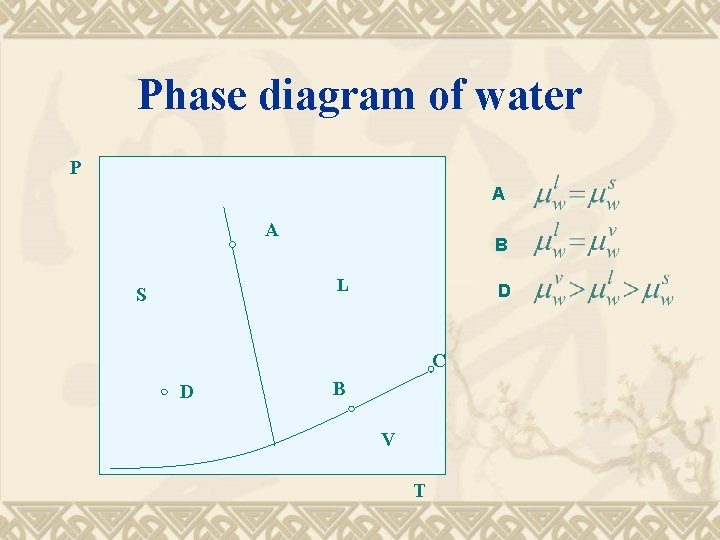
Phase diagram of water P A A B L S D C D B V T
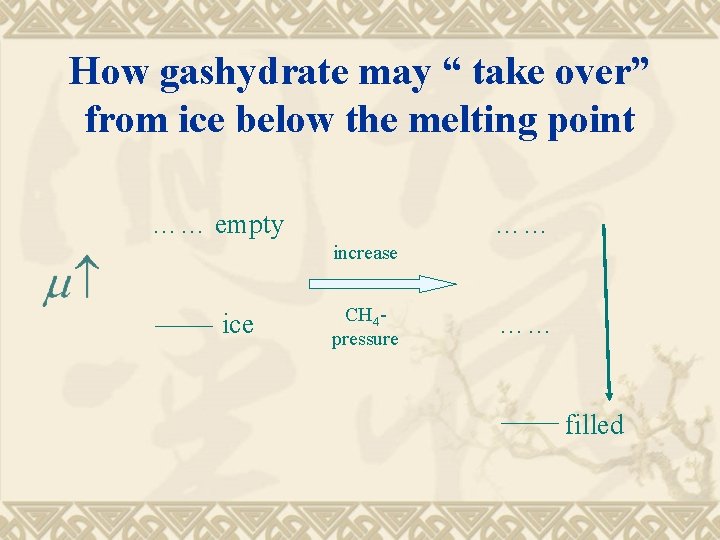
How gashydrate may “ take over” from ice below the melting point …… empty …… increase ice CH 4 pressure …… filled

How gashydrate may form from liquid water above the melting point …… empty …… ice …… water Increasing CH 4 - pressure …… filled
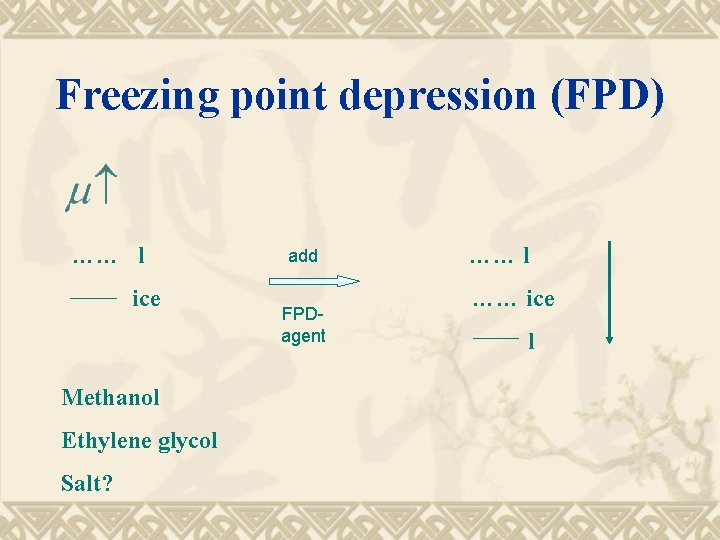
Freezing point depression (FPD) …… l ice Methanol Ethylene glycol Salt? add FPDagent …… l …… ice l

Hydrate inhibition Just like an FPD- agent is effective in suppression of ice formation it may suppress hydrate formation …… empty H …… …… liquid …… …… ice …… filled H …… liquid A costly “ affair”

Hydrate promoters (1) Certain molecules, like tetrahydrofuran (THF), may promote hydrate formation by assisting in filling the vacancies.

Hydrate Promoters (2) These, usually non-volatile, promoters may produce the hydrate structure in which the gas molecules can be included although their pressure is too low to achieve this by themselves. (e. g. H 2)

Solution? It is my impression that these days industry employs inhibitors that don't suppress hydrate formation but suppress hydrate crystal growth producing some kind of “hydrate milk” that does not block pipeline operation. “If you can’t beat the enemy, join them……”

Prediction (1) In the oil-and gas industry one likes to know when hydrate formation can be expected, especially at temperatures above 0 °C. Possible phases Hydrate H Liquid W aqueous Liquid non-aqueous Vapour V

Prediction (2) Components of natural gas: C 1 C 2 C 3 …… N 2 CO 2 …… For example: where is the location of the HLw. Vequilibrium curve? k = 1, 2, ……N Models required for the various phases

Prediction (3) These days the large oil- and gas companies make use of powerful software to allow them to predict not only all possible hydrate phase diagrams but also the effect of inhibitors (by including for example methanol in the calculation programme).

Equilibrium conditions for different hydrocarbons Pressure I-H-V H-Lw-L red CH 4 blue C 2 H 6 green C 3 H 8 magenta i-C 4 H 10 Temperature [K]

Equilibrium conditions for some other gases Pressure I-H-V H-Lw-L H-L-V red N 2 blue CO 2 magenta H 2 S Temperature [K]
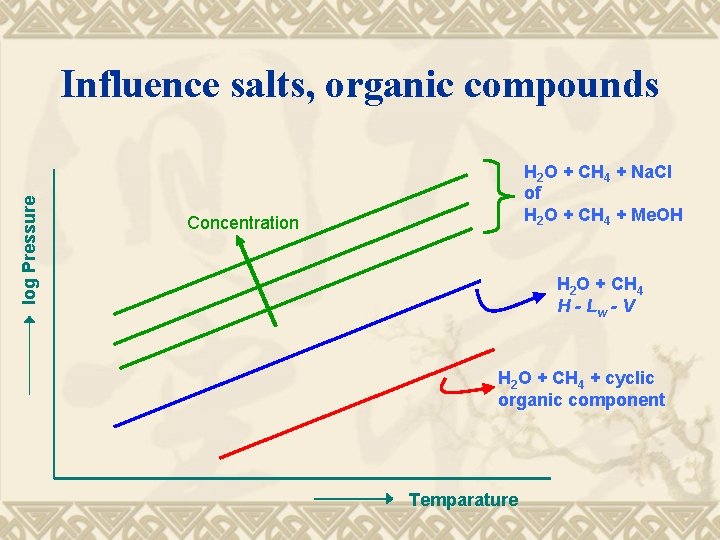
log Pressure Influence salts, organic compounds H 2 O + CH 4 + Na. Cl of H 2 O + CH 4 + Me. OH Concentration H 2 O + CH 4 H - Lw - V H 2 O + CH 4 + cyclic organic component Temparature
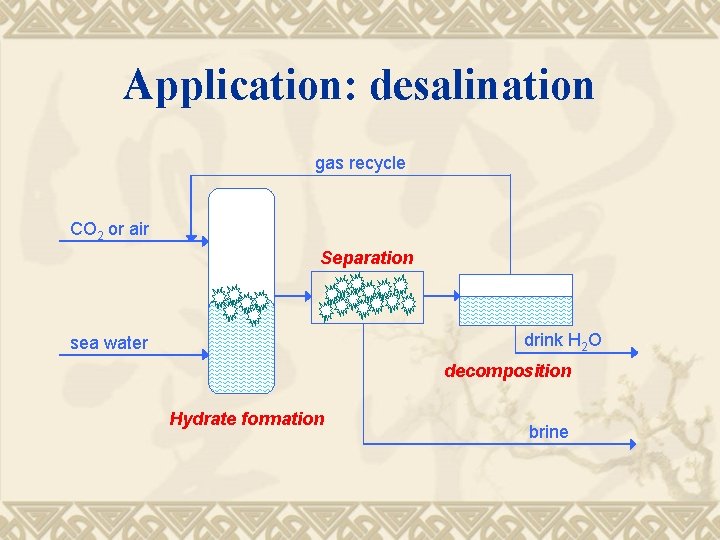
Application: desalination gas recycle CO 2 or air Separation drink H 2 O sea water decomposition Hydrate formation brine

Threat CH 4 is a much more serious contributant to greenhouse effect than CO 2. So with the Earth warming up, natural gas hydrates may start dissociating and we may face a “runaway” greenhouse effect. Also: Leaking pipelines in former Soviet-Union
![Living on NGH ? US Geological Survey [1998] Living on NGH ? US Geological Survey [1998]](http://slidetodoc.com/presentation_image_h/a5b60e423ac481baab0b17ebc60a2132/image-48.jpg)
Living on NGH ? US Geological Survey [1998]

May I introduce myself ?

Acknowledgment I wish to acknowledge the contributions of my former Ph. D. student Miranda Mooijer- Van den Heuvel, who is now with Shell Global Solutions International. She graduated on a thorough study of how certain compounds can promote hydrate formation.
- Slides: 50