Sheet Silicates n n n Abundant and common

Sheet Silicates n n n Abundant and common minerals throughout upper 20 km of crust Felsic to intermediate igneous, metamorphic, and sedimentary rocks All are hydrous Contain H n Bonded to O to form OHn n n Z/O ratio of 2/5 2 Major groups: Micas & Clays

Groupings n n Based on structure Two kinds of “layers” within the “sheets” n “T” layers – tetrahedral layers n n Tetrahedral coordination of Si and Al “O” sheets – octahedral layers n Octahedral coordination of mostly Al and Mg, occasionally Fe

n T and O layers bonded to form sheets The sheets are repeated in vertical direction n The spaces between the sheets may be: n Vacant n Filled with interlayer cations, water, or other sheets n n Primary characteristic - basal cleavage Single perfect cleavage n Occurs because bonds between sheets are very weak n

Construction of T-O-T Sheets n n n Octahedral layers: Two planes of OH- anionic groups Cations are two types: Divalent (Fe 2+ or Mg 2+) n Trivalent (Al 3+ or Fe 3+) n Mg and Al most common n

n Divalent cations fill 3 of 3 sites Form trioctahedral sheets n Ideal formula is Mg 3(OH)6 n This formula is brucite n n A hydroxide, not a silicate mineral All sites filled with divalent cations Charge neutral

n Trivalent cations fill 2 of 3 sites Form dioctrahedral sheets n Ideal formula is Al 2(OH)6 n Mineral called gibbsite n n A hydroxide, not silicate mineral 2/3 of sites filled with trivalent cations Charge neutral
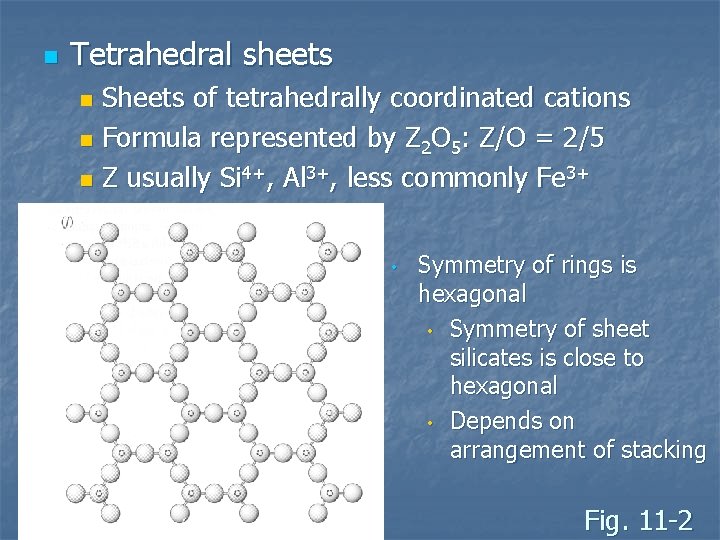
n Tetrahedral sheets Sheets of tetrahedrally coordinated cations n Formula represented by Z 2 O 5: Z/O = 2/5 n Z usually Si 4+, Al 3+, less commonly Fe 3+ n • Symmetry of rings is hexagonal • Symmetry of sheet silicates is close to hexagonal • Depends on arrangement of stacking Fig. 11 -2

n Tetrahedron are meshes of 6 -fold rings Three basal oxygen on each tetrahedron shared by adjacent tetrahedron n The fourth, unshared oxygen is the apical oxygen n n Tetrahedral layers are two oxygen thick

• Tetrahedral sheet composition is Si 2 O 52 • May have Al 3+ or Fe 3+ substitute for Si 4+ • Increases net negative charge Fig. 13 -1

n Tetrahedral and octahedral sheets always joined Apical oxygen of tetrahedral sheets formed part of octahedral sheets n Apical oxygen replaces one of the OH- in the octahedral sheets n n Sheets joined in two ways TO layers, called 1: 1 layer silicates n TOT layers, called 2: 1 layer silicates n
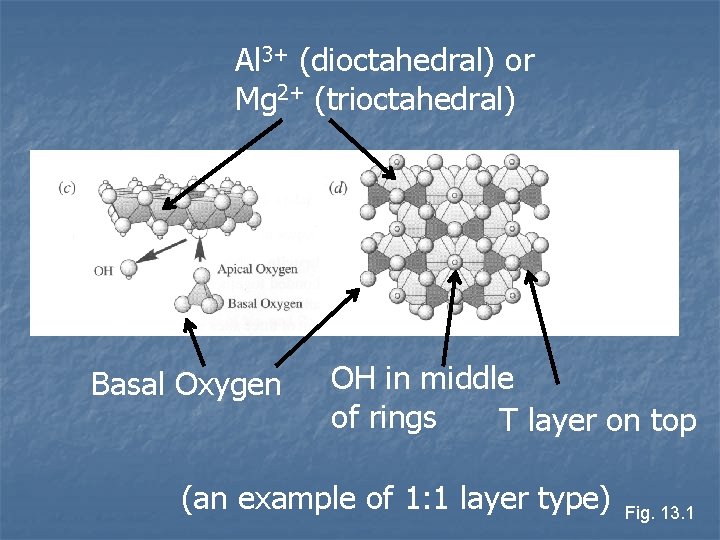
Al 3+ (dioctahedral) or Mg 2+ (trioctahedral) Basal Oxygen OH in middle of rings T layer on top (an example of 1: 1 layer type) Fig. 13. 1

1: 1 layer summary n n Consists of 3 planes of anions One plane is basal plane of shared tetrahedral oxygen Other side is the OH- anionic group of the octahedral sheet Middle layer is the OH- anionic group with some OH- replaced by oxygen
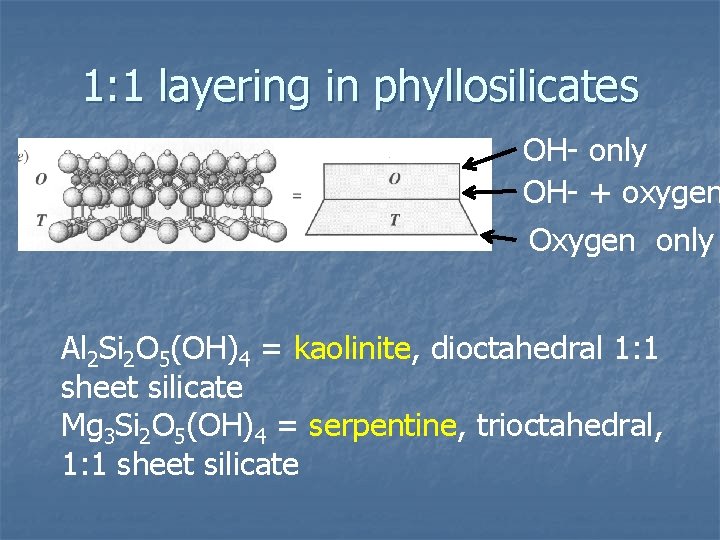
1: 1 layering in phyllosilicates OH- only OH- + oxygen Oxygen only Al 2 Si 2 O 5(OH)4 = kaolinite, dioctahedral 1: 1 sheet silicate Mg 3 Si 2 O 5(OH)4 = serpentine, trioctahedral, 1: 1 sheet silicate

2: 1 layer silicates n n 2 tetrahedral layers on both sides of octahedral layer TOT structure has 4 layers of anions Both sides (outermost) are planes of basal, shared oxygen Middle planes contain original OH- from octahedral layers and apical oxygen from tetrahedron
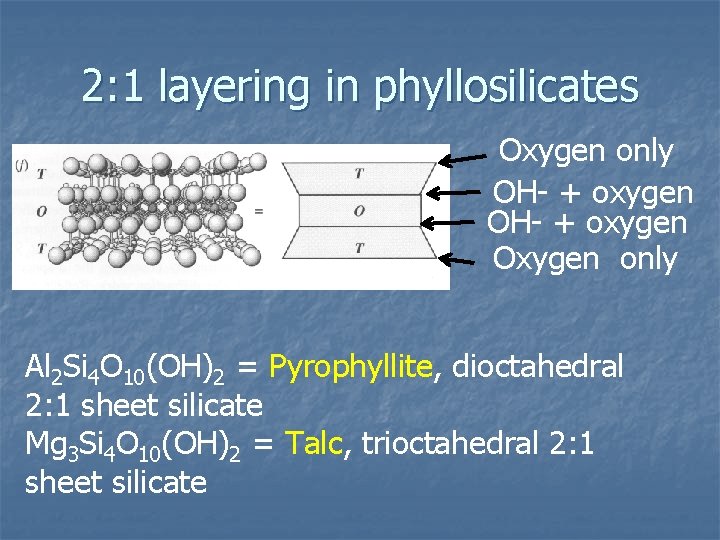
2: 1 layering in phyllosilicates Oxygen only OH- + oxygen Oxygen only Al 2 Si 4 O 10(OH)2 = Pyrophyllite, dioctahedral 2: 1 sheet silicate Mg 3 Si 4 O 10(OH)2 = Talc, trioctahedral 2: 1 sheet silicate

How are sheets stacked? 1: 1 layer I. …T-O…T-O… I. 2: 1 layer II. III. § § Kaolinite (dioctahedral) Serpentine (trioctahedral) Pyrophyllite (dioctahedral) Talc (trioctahedral) …T-O-T…T-O-T… c…T-O-T…c…T-O-T…c… O…T-O-T…O Four types of layers, each dioctahedral or trioctahedral Each one may be dioctahedral or trioctahedral

1: 1 layer silicates n n Kaolinite and Serpentine Bonding between sheets very weak Electrostatic bonds – van der Waals and hydrogen n Results in very soft minerals n Thickness of TO layers around 7 Å n
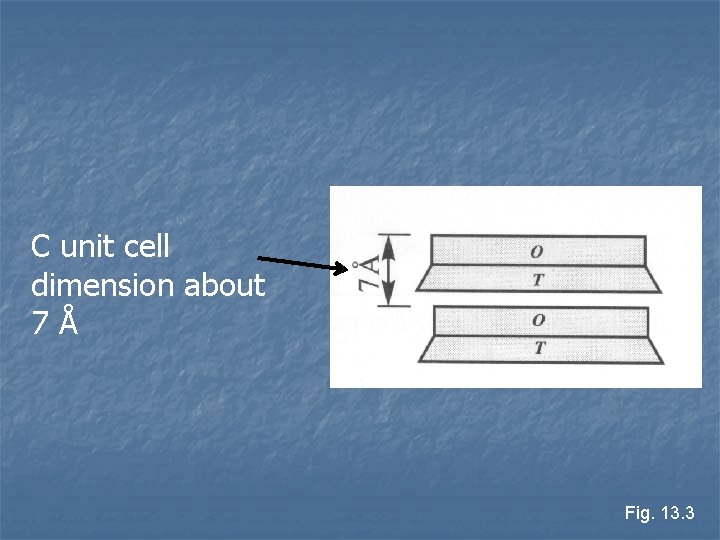
C unit cell dimension about 7Å Fig. 13. 3

2: 1 layer silicates n Unit structure is repeating TOT layers, two ways: n (1) TOT layers can be electrically neutral Nothing in between sheets van der Waal forces n Pyrophyllite & Talc n n (2) substitution in TOT layers can give a net charge Most common substitution is Al 3+ for Si 4+ in tetrahedral layers n Charge balance maintained with substitution between the sheets n

n TOT structure If there is only Si 4+ in T layers (no Al 3+ or Fe 3+) n Electrically neutral, no interlayer cations n TOT layers weakly bonded by van der Waal and hydrogen bonds n Soft (Talc), greasy feel n
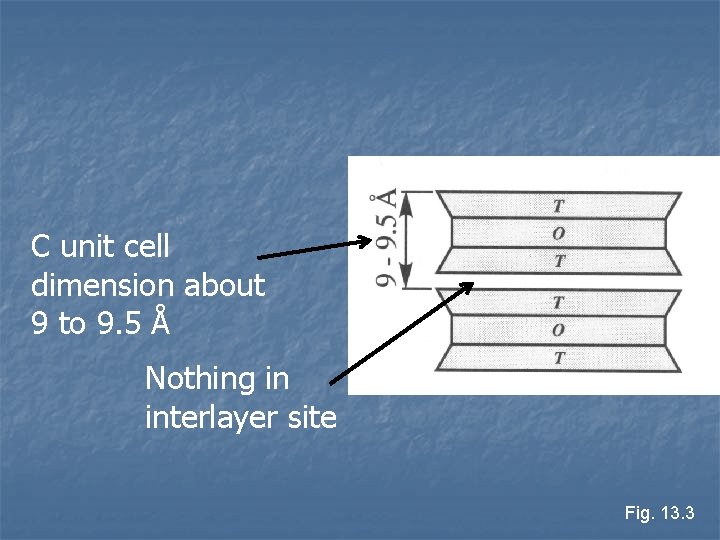
C unit cell dimension about 9 to 9. 5 Å Nothing in interlayer site Fig. 13. 3

n c…T-O-T…c These are the mica minerals n Also less common are “brittle micas” n Structure is TOT layers with 1 out of 4 tetrahedral sites occupied by Al 3+ n

n Micas Al/Si ratio in the tetrahedral layer is 1/3 n Dioctahedral TOT layer = Al 2(Al. Si 3 O 10)(OH)21 n n n Remember Pyrophyllite: Al(Si 2 O 5)(OH)2 Trioctahedral TOT layer = Mg 3(Al. Si 3 O 10)(OH)21 n Remember Talc: Mg 3(Si 2 O 5)(OH)2 Negative charge balance by large monovalent cation, usually K+ n Bonds are ionic, fairly strong, harder minerals n
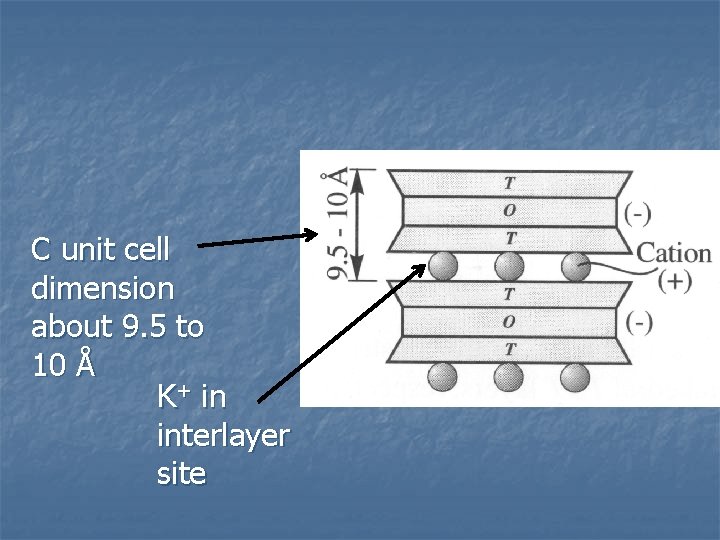
C unit cell dimension about 9. 5 to 10 Å K+ in interlayer site

n Dioctahedral mica – muscovite n n KAl 2(Al. Si 3 O 10)(OH)2 Trioctahedral mica – Phlogopite n KMg 3(Al. Si 3 O 10)(OH)2

Brittle Micas n n Similar to micas, but more Al 3+ substitution Charge balanced by Ca 2+ Margarite – half of tetrahedral sites have Al 3+ substitution Clintonite – ¾ of tetrahedral sites have Al 3+ substitution

n Margarite Dioctahedral n Ca. Al 2(Al 2 Si 2 O 10)(OH)4 n n Clintonite Trioctahedral n Ca. Mg 2 Al(Al 3 Si. O 10)(OH)2 n n Now charge balance in part from Al substitution in octahedral layer

n …O…T-O-T…O… Most common members are in the chlorite group n Structure like Talc, but with brucite (Mg 3(OH)6) interlayer n n T layers with small negative charge n n O layers often have net positive charge n n Substitute small amounts of Al 3+ for Si 4+ Substitute Al 3+ or Fe 3+ for divalent cations Minerals harder than expected
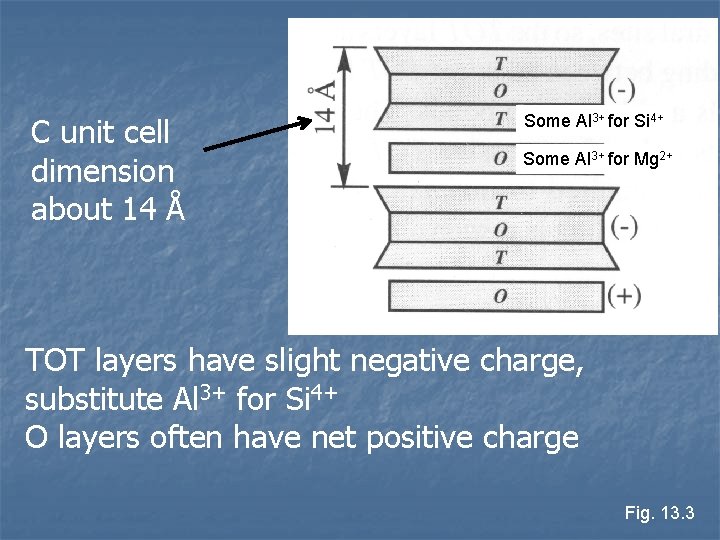
C unit cell dimension about 14 Å Some Al 3+ for Si 4+ Some Al 3+ for Mg 2+ TOT layers have slight negative charge, substitute Al 3+ for Si 4+ O layers often have net positive charge Fig. 13. 3

Varieties of sheet silicates n TO structures Serpentine (var. Antigorite, Chrysotile, Lizardite) n All are trioctahedral n Trioctahedral sheets, a = 5. 4 Å; b = 9. 3 Å n Tetrahedral sheets, a = 5 Å; b = 8. 7 Å n Mismatched size leads to variations n
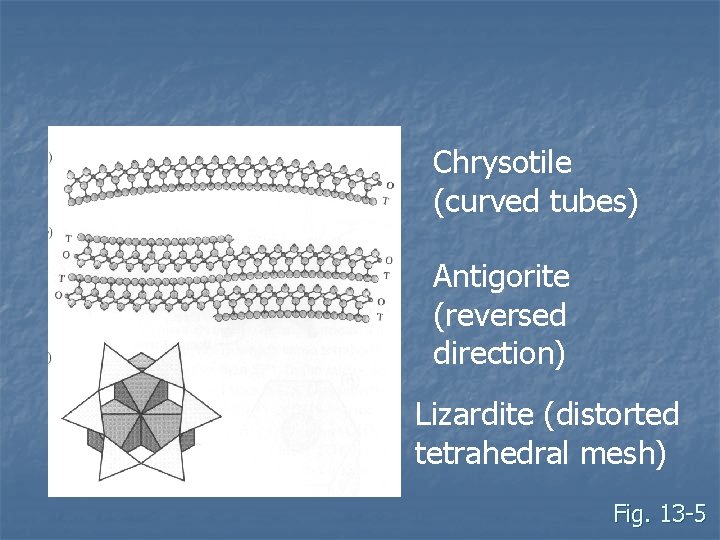
Chrysotile (curved tubes) Antigorite (reversed direction) Lizardite (distorted tetrahedral mesh) Fig. 13 -5

Clay Minerals n Clay has two meanings: Particles < 1/256 mm, or 0. 0039 mm n A group of sheet silicate minerals (not micas) that are commonly clay-sized n n n Original description from not being able to identify small grain size material Now can use X-ray diffraction to determine clays

Terminology n n Clay: Sediment composed of particles that are < 0. 002 mm Claystone: Rock composed of clay-sized particles Clay minerals: 1: 1 and 2: 1 Phyllosilicate minerals without K+ or Ca 2+ bonding sheets (those are Micas) Argillaceous: Rock or sediment containing large amounts of clay and clay minerals

n Problems Clay size fraction can contain other minerals (quartz, carbonates, zeolites etc. ) n “Clay” used to define size fraction – size not mineralogical n Several clay minerals can be larger than the size requirements n

Clay classification n 1: 1 layer clays 7 Å type n TO layers n Kaolinite (dioctahedral) n Serpentine (trioctahedral) n

n 2: 1 layer clays: n End members: 10 Å – Pyrophyllite (dioctahedral) & talc (trioctahedral) n 10 Å – Charge imbalance: with K as interlayer: Mica: Muscovite (trioctahedral) & Biotite (dioctahedral) n 14 Å type (Chlorite) n

n Intermediate 2: 1 clays Have net negative charge, but less than one per formula n Requires less interlayer cations to charge balance n n Mixed layer clays – combined 1: 1 and 1: 2

n Three types of intermediate 10 Å clays Low charge imbalance – smectite clays n High charge imbalance – illite clays n Intermediate charge imbalance – vermiculite n n Charge imbalance controlled by “interlayer” cations n They move in and out – Cation Exchange Capacity (CEC) n Surface adsorption n

Low charge n n Smectite approximately = Ca 0. 17(Al, Mg, Fe)2(Si, Al)4 O 10(OH)2 • n. H 2 O Net negative charge is 0. 2 to 0. 6 per formula unit, typically 0. 33 Ca and Na are typical interlayer ions n n n Exchangable May be dioctahedral or trioctahedral Charge results from Al substitution for Si in tetrahedron n Mg for Al in octahedron (in dioctahedral) n

n Low charge means water and cations (Na, K, Ca, Mg) easily move in and out of interlayer sites No water = 10 Å n One water layer = 12. 5 Å n Two water layer = 15. 2 Å n n Water moves in and out depending on moisture in environment

High charge n Illite/glauconite Approximately = K 0. 8 Al 2(Al 0. 8 Si 3. 2)(OH)2 n Net negative charge of 0. 8 to 1 per formula n Very similar to muscovite – called mica-like n Mostly substitute of Al 3+ for Si 4+ n All are dioctahedral; Glauconite has Fe 3+ n Interlayer ion is K+ n High K concentration means strong bond n Difficult for water to enter n Non-swelling clay n

Intermediate charge n Vermiculite Approximately = (Mg, Ca)0. 3(Mg, Fe 2+, Fe 3+, Al)3(Si, Al)4)O 10(OH)2 n About 0. 6 charge per formula unit n Comes from oxidation of Fe 2+ to Fe 3+ in biotite n n Reduces the negative charge on TOT layer from -1 to -0. 6 Less K+ than mica, can exchange for Ca 2+ and Mg 2+ and water n Swell clay n With water interlayer spacing is 14. 4 Å n

Mixed layer clays n n Natural clays rarely similar to the end members Typically contain parts of different types of clays Actually mixtures at unit cell level – not physical mixtures Nomenclature – combined names n Illite/smectite or chlorite/smectite

7Å 1: 1 layer clays 10 Å 2: 1 layer clays – low charge, smectite 2: 1 layer clays – high charge, illite 14 Å 2: 1 layer clays – Chlorite gp Mixed layer Figure 13 -15

Burial Diagenesis n n n Smectite converts to illite with burial Most conversion at 50 to 100º C Conversion requires K, usually comes from dissolution of K spar
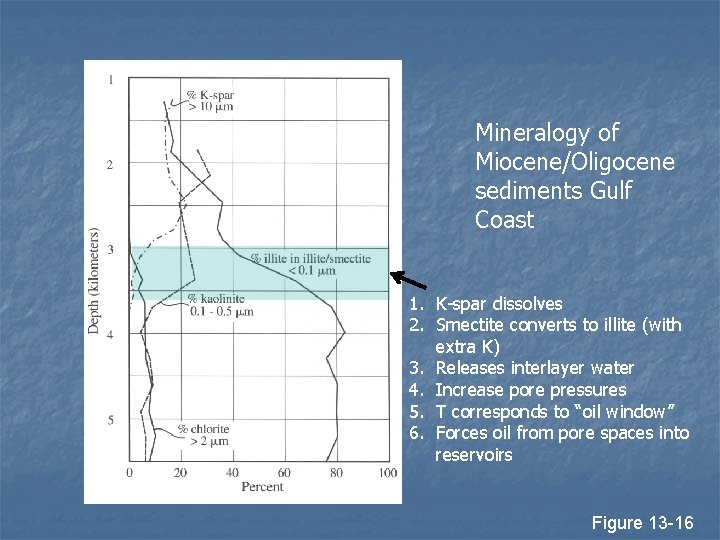
Mineralogy of Miocene/Oligocene sediments Gulf Coast 1. K-spar dissolves 2. Smectite converts to illite (with extra K) 3. Releases interlayer water 4. Increase pore pressures 5. T corresponds to “oil window” 6. Forces oil from pore spaces into reservoirs Figure 13 -16
- Slides: 46