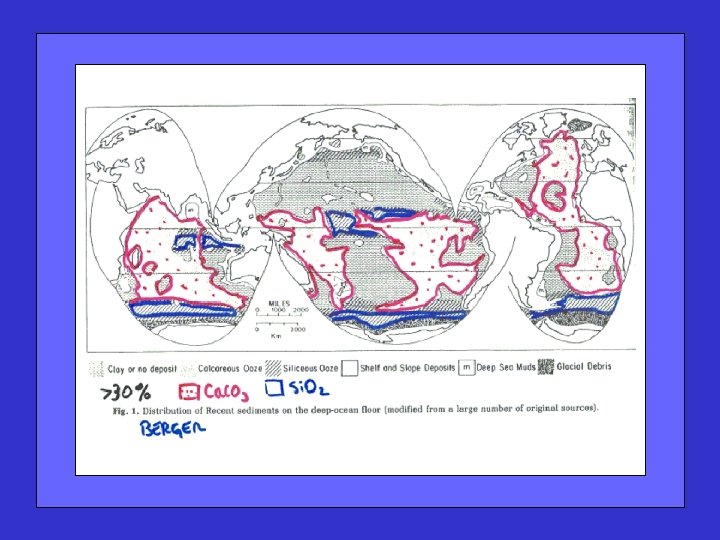
Shallow water carbonate sedimentation Including partial reviews of

Shallow water carbonate sedimentation Including partial reviews of : Carbonate chemistry (solubility, saturation state) Metabolic dissolution (impact of OM decomposition) Stoichiometry and flux balance (predictions, checks) Non-diffusive transport(s) (often important) And introducing Ocean Acidification
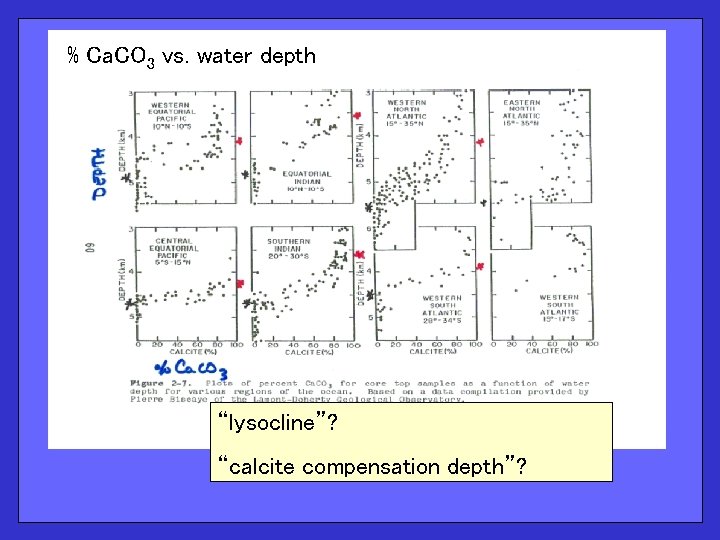
% Ca. CO 3 vs. water depth “lysocline”? “calcite compensation depth”?
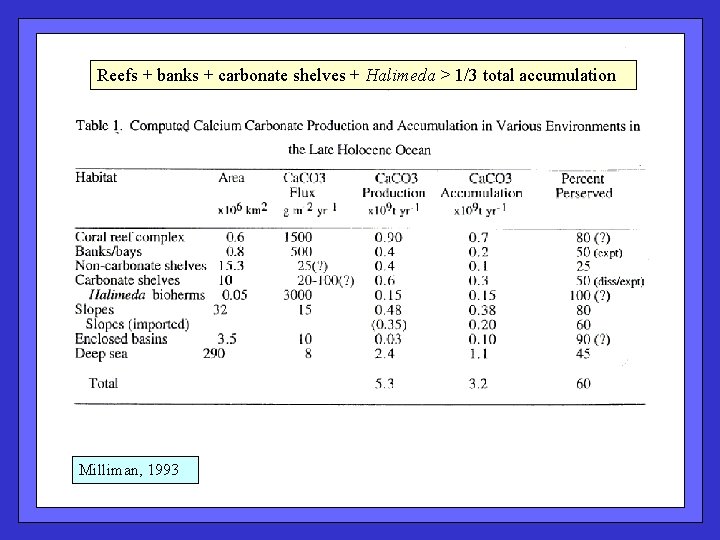
Reefs + banks + carbonate shelves + Halimeda > 1/3 total accumulation Milliman, 1993

Substantial carbonate dissolution in shallow water carbonate sediments. Bottom water supersaturated with likely carbonate phases (calcite, aragonite, high-Mg calcite); the dissolution must be “metabolic”.
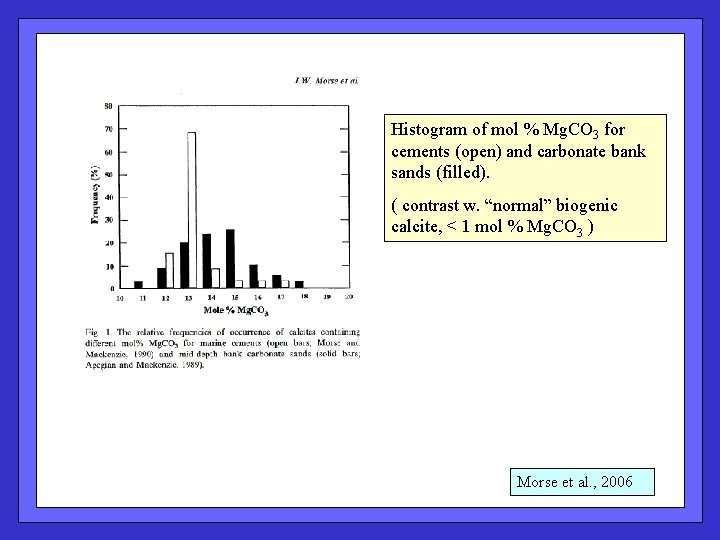
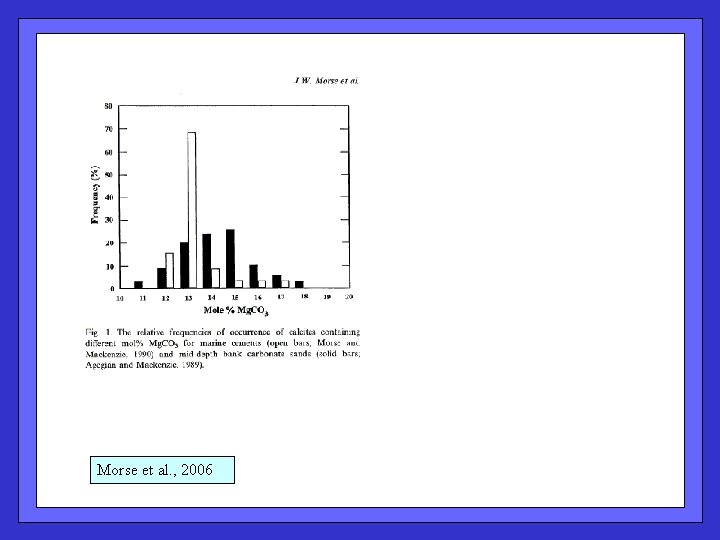
Histogram of mol % Mg. CO 3 for cements (open) and carbonate bank sands (filled). ( contrast w. “normal” biogenic calcite, < 1 mol % Mg. CO 3 ) Morse et al. , 2006
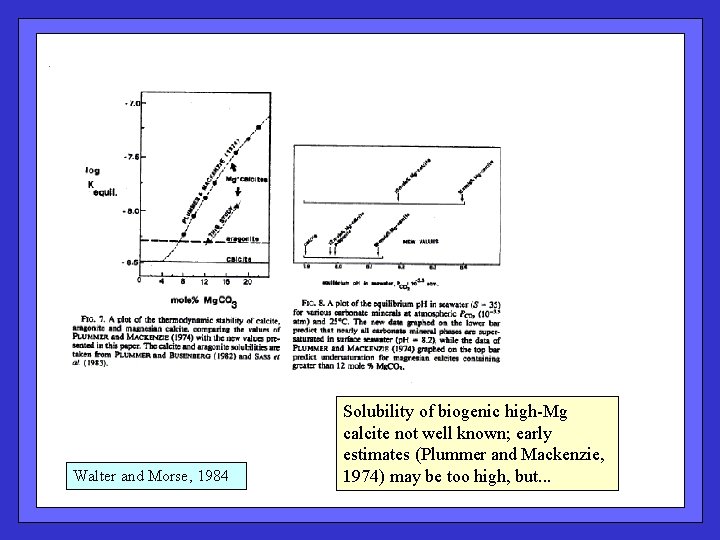
Walter and Morse, 1984 Solubility of biogenic high-Mg calcite not well known; early estimates (Plummer and Mackenzie, 1974) may be too high, but. . .
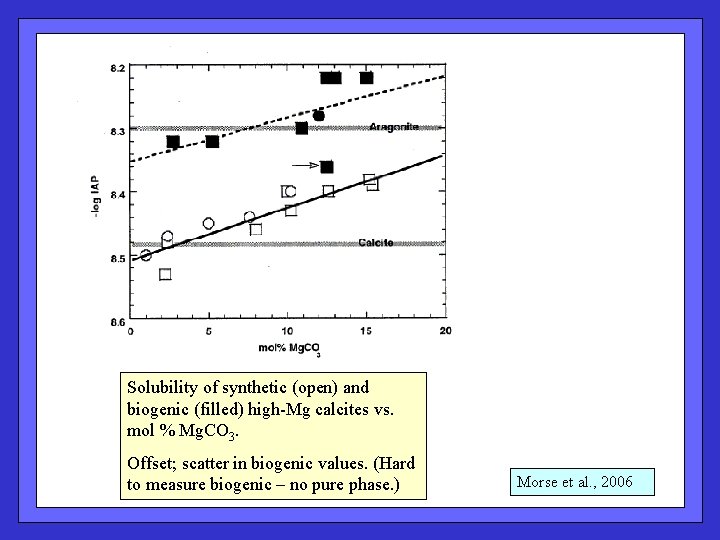
Solubility of synthetic (open) and biogenic (filled) high-Mg calcites vs. mol % Mg. CO 3. Offset; scatter in biogenic values. (Hard to measure biogenic – no pure phase. ) Morse et al. , 2006
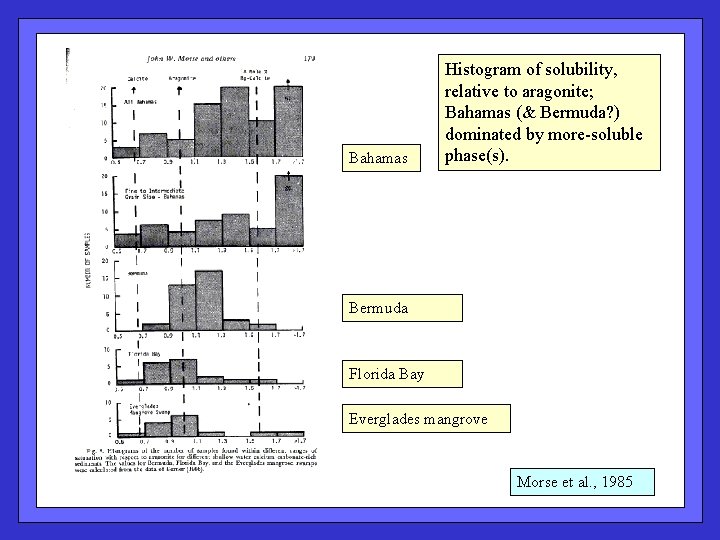
Bahamas Histogram of solubility, relative to aragonite; Bahamas (& Bermuda? ) dominated by more-soluble phase(s). Bermuda Florida Bay Everglades mangrove Morse et al. , 1985
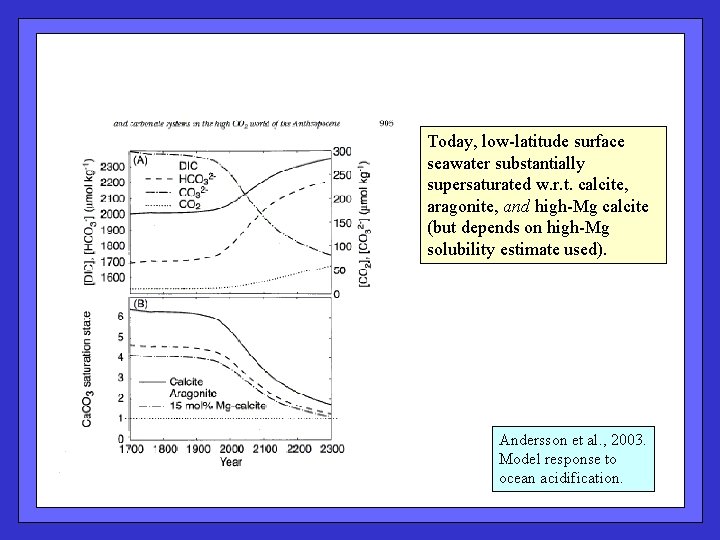
Today, low-latitude surface seawater substantially supersaturated w. r. t. calcite, aragonite, and high-Mg calcite (but depends on high-Mg solubility estimate used). Andersson et al. , 2003. Model response to ocean acidification.

Stoichiometry of OM oxidation by oxygen, nitrate, and sulfate Predicted aragonite saturation state during oxygen respiration? Small alk decrease; larger CO 2 increase. Expected change in [CO 3=] for O 2 respiration?
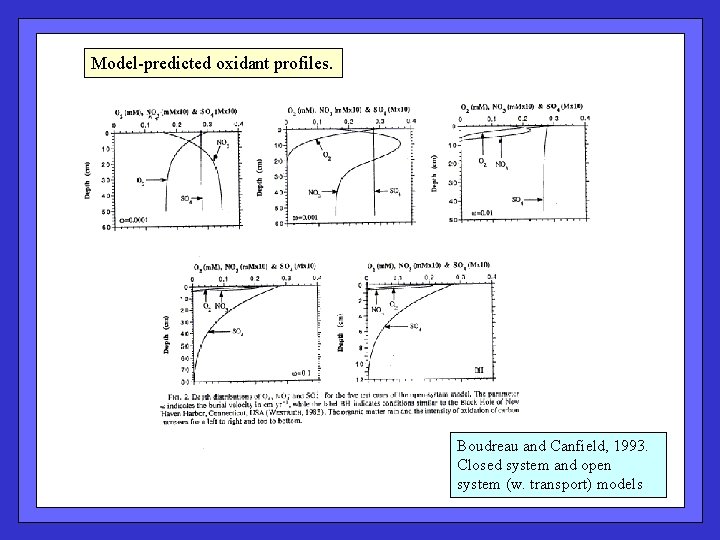
Model-predicted oxidant profiles. Boudreau and Canfield, 1993. Closed system and open system (w. transport) models
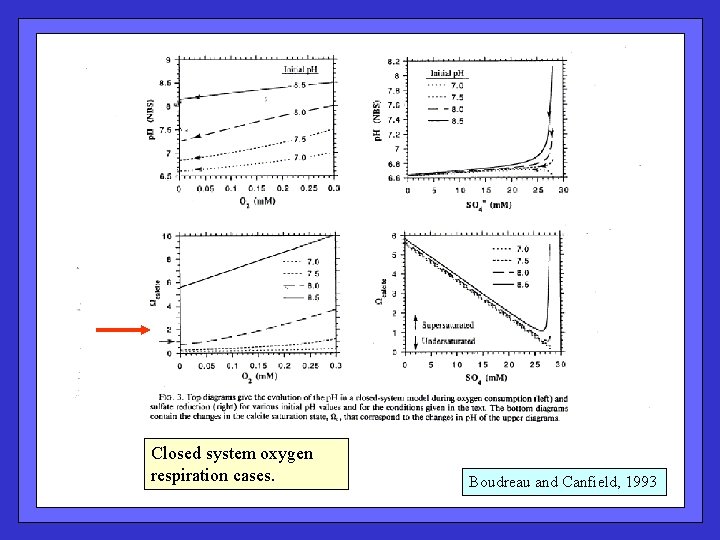
Closed system oxygen respiration cases. Boudreau and Canfield, 1993
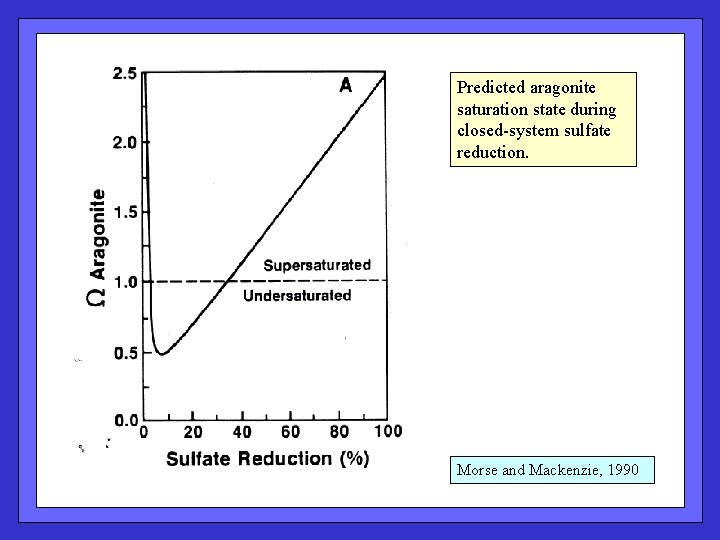
Predicted aragonite saturation state during closed-system sulfate reduction. Morse and Mackenzie, 1990
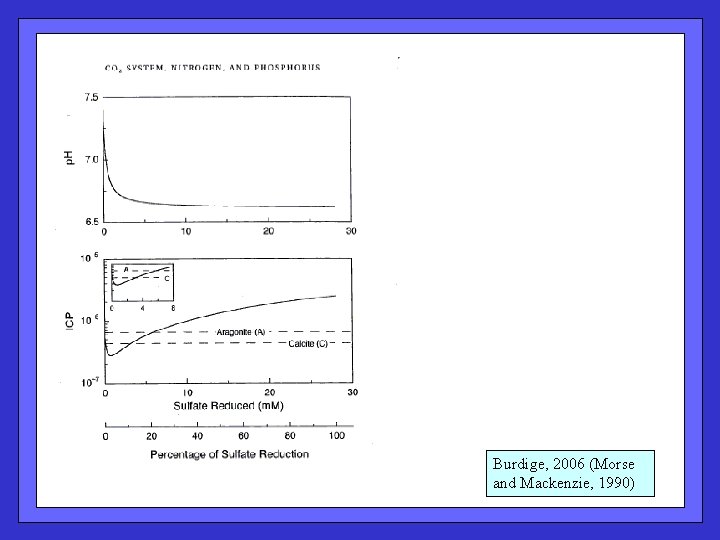
Burdige, 2006 (Morse and Mackenzie, 1990)

Stoichiometry of OM oxidation by oxygen, nitrate, and sulfate Predicted aragonite saturation state during closed-system sulfate reduction: Remember simplified versions of alkalinity (~ HCO 3 - + 2 x CO 3=) and DIC (~ HCO 3 - + CO 3=). Expression for [CO 3=] ? What is delta [CO 3=] for SR? Boudreau and Canfield, 1993 Why should saturation state drop?

Species that yield protons upon dissociation H 2 S is produced during SR Some H 2 S dissociates into HS- and H+ Some H+ reacts with CO 3= to yield HCO 3[CO 3=] drops; can drive dissolution Boudreau and Canfield, 1993 With continued SR, alk production overshadows H 2 S dissociation.

Re-oxidation of reduced metabolites by pore water oxygen releases acidity close to the sediment-water interface. This lowers pore water p. H and drives dissolution (though some of this acidity can be neutralized by bottom water alkalinity). S burial as Fe. S lowers the H 2 S concentration, and thus reduces the proton release due to H 2 S dissociation; this minimizes initial dissolution due to SR Boudreau and Canfield, 1993; Ku et al. , 1999
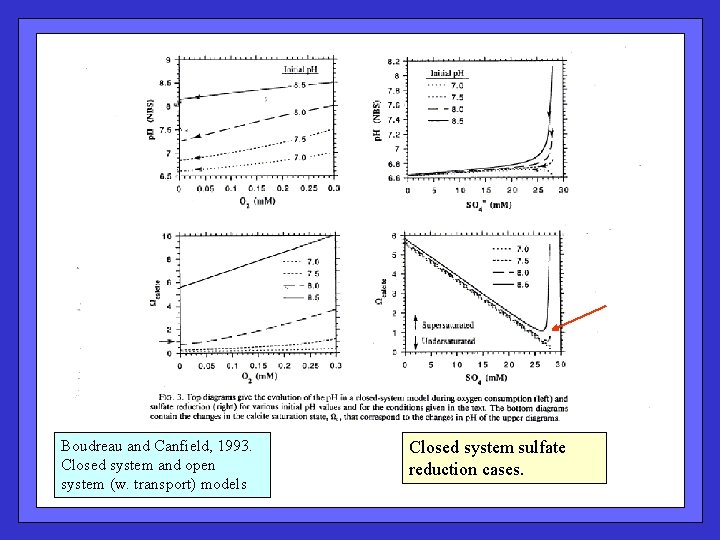
Boudreau and Canfield, 1993. Closed system and open system (w. transport) models Closed system sulfate reduction cases.
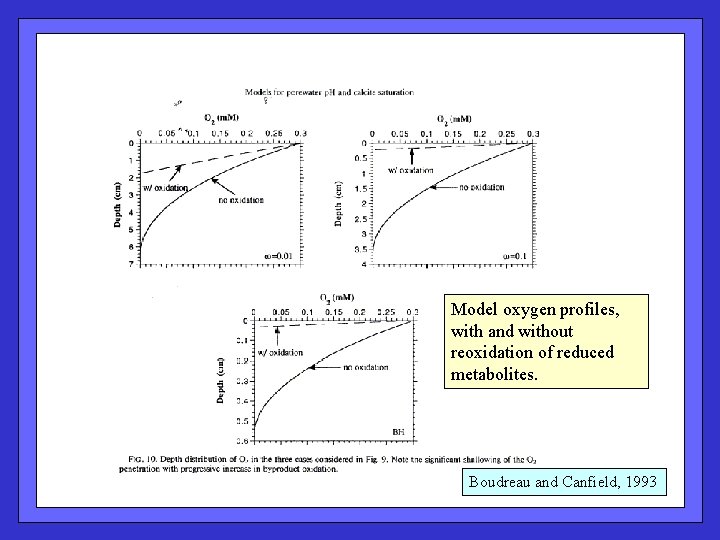
Model oxygen profiles, with and without reoxidation of reduced metabolites. Boudreau and Canfield, 1993
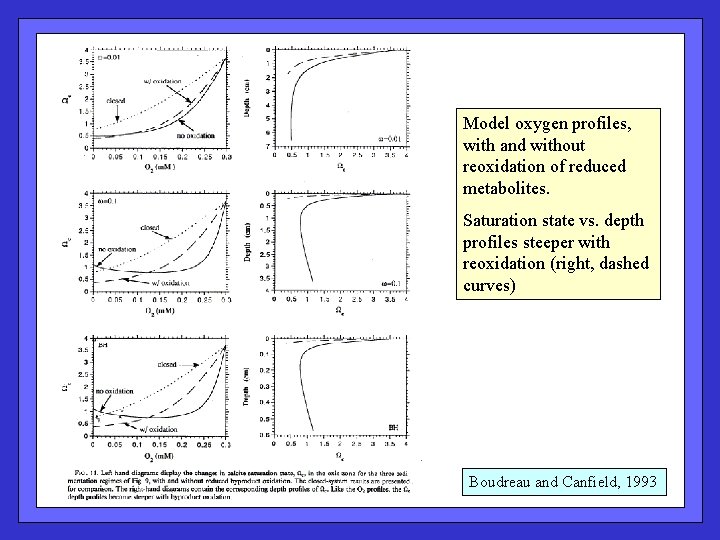
Model oxygen profiles, with and without reoxidation of reduced metabolites. Saturation state vs. depth profiles steeper with reoxidation (right, dashed curves) Boudreau and Canfield, 1993
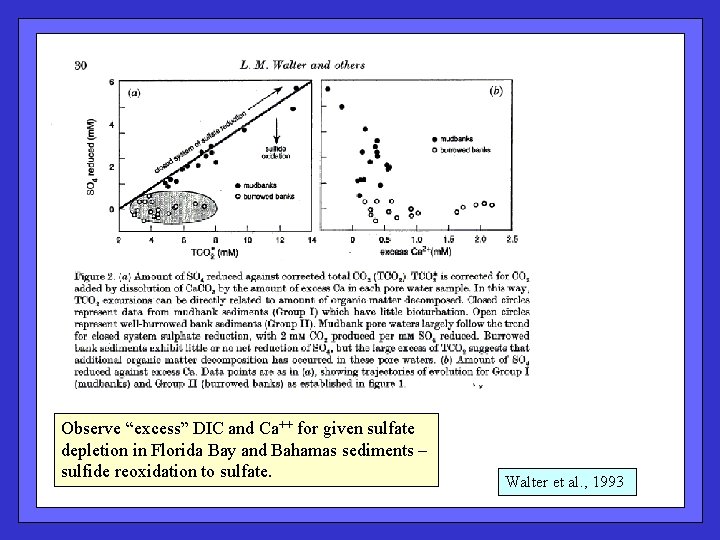
Observe “excess” DIC and Ca++ for given sulfate depletion in Florida Bay and Bahamas sediments – sulfide reoxidation to sulfate. Walter et al. , 1993

Shallow-water metabolic dissolution (+ S and O stable isotopes) in Florida Bay; Sulfate reduction / reoxidation cycle equivalent to enhanced oxic respiration in terms of saturation state. Expected changes due to reaction stoichiometry – DIC: Ca = 2: 1 DIC: Alk = 1: 1 No net change in sulfate Ku et al. , 1999
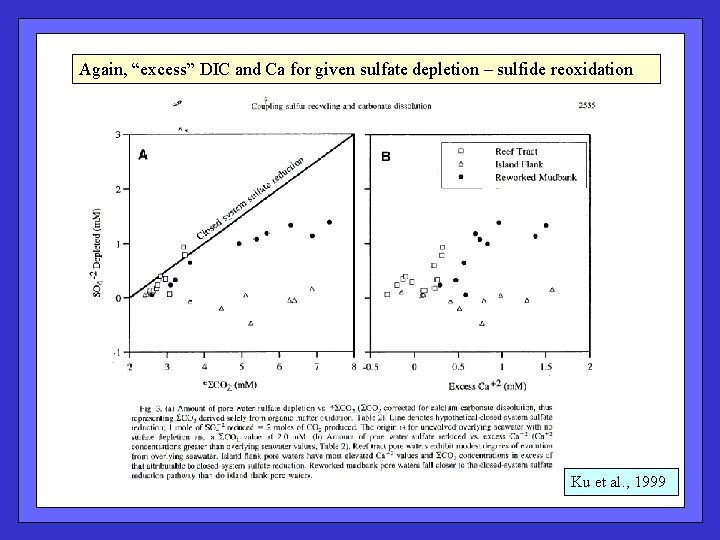
Again, “excess” DIC and Ca for given sulfate depletion – sulfide reoxidation Ku et al. , 1999
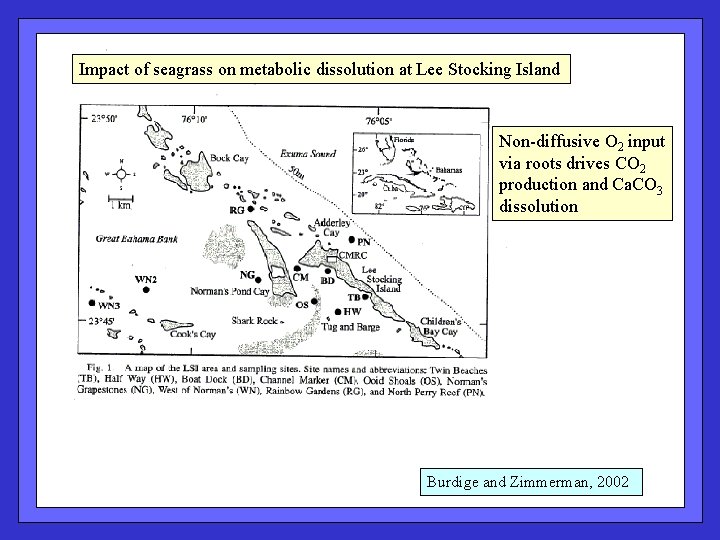
Impact of seagrass on metabolic dissolution at Lee Stocking Island Non-diffusive O 2 input via roots drives CO 2 production and Ca. CO 3 dissolution Burdige and Zimmerman, 2002
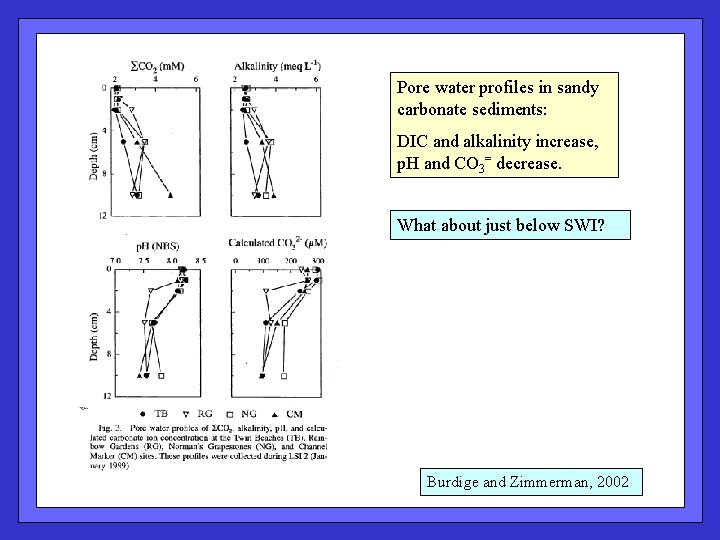
Pore water profiles in sandy carbonate sediments: DIC and alkalinity increase, p. H and CO 3= decrease. What about just below SWI? Burdige and Zimmerman, 2002
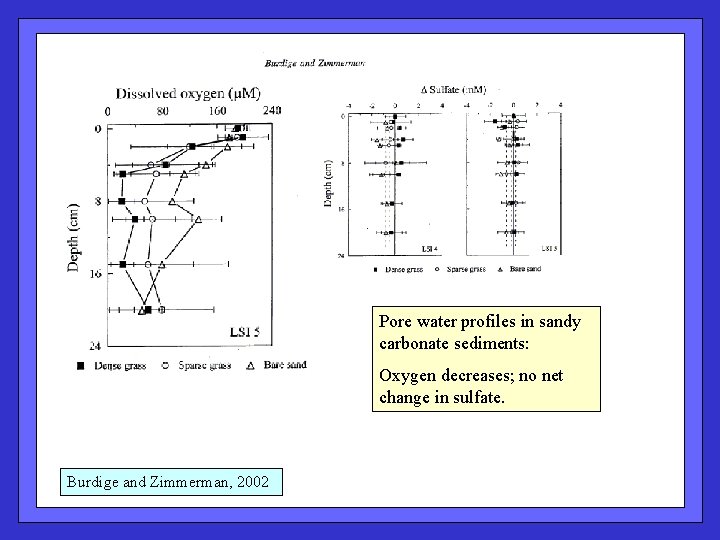
Pore water profiles in sandy carbonate sediments: Oxygen decreases; no net change in sulfate. Burdige and Zimmerman, 2002
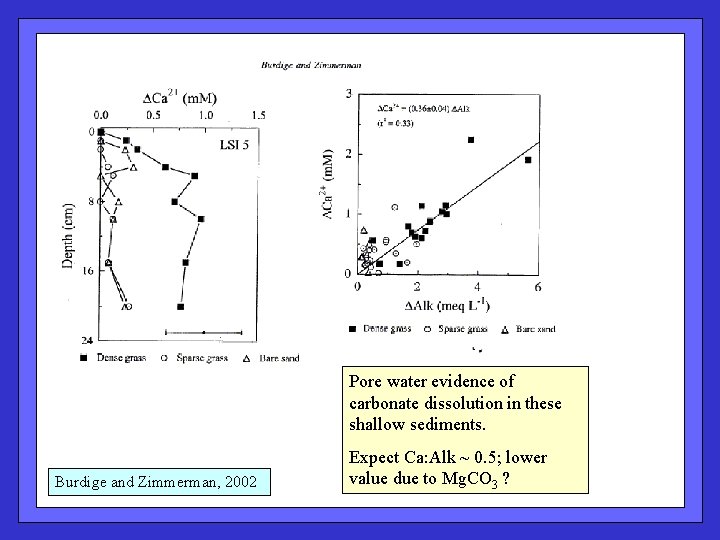
Pore water evidence of carbonate dissolution in these shallow sediments. Burdige and Zimmerman, 2002 Expect Ca: Alk ~ 0. 5; lower value due to Mg. CO 3 ?
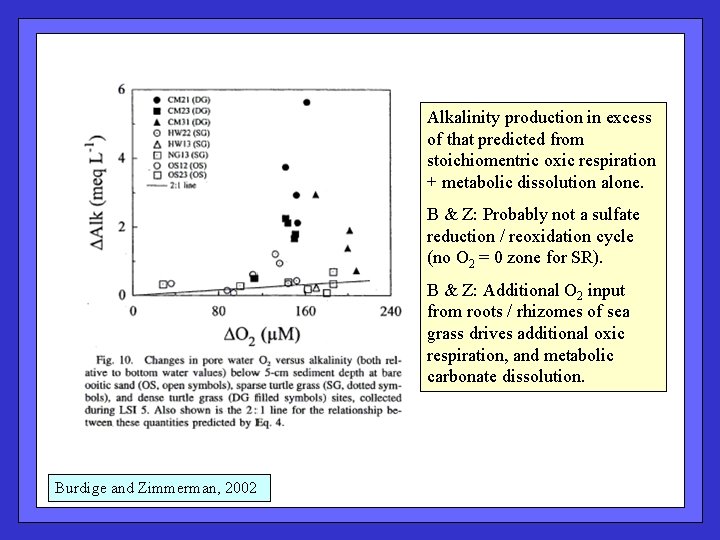
Alkalinity production in excess of that predicted from stoichiomentric oxic respiration + metabolic dissolution alone. B & Z: Probably not a sulfate reduction / reoxidation cycle (no O 2 = 0 zone for SR). B & Z: Additional O 2 input from roots / rhizomes of sea grass drives additional oxic respiration, and metabolic carbonate dissolution. Burdige and Zimmerman, 2002

Can dissolution of shallow carbonate-rich sediments (including high-solubility high-Mg carbonates) offset the ocean acidification impact of fossil fuel CO 2 release? Probably not (e. g. , Andersson, 2005; 1 m reactive depth, 50% porosity, 95: 5 at 15%: 80% Ca. CO 3, observed mineralogy…) but let’s look.
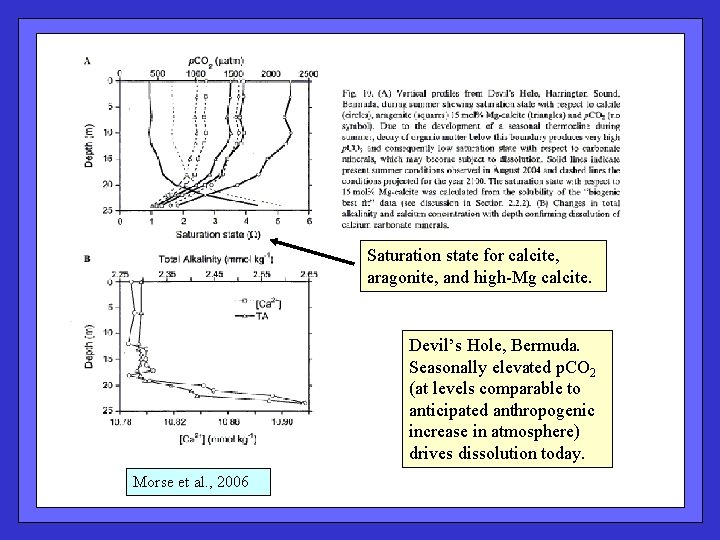
Saturation state for calcite, aragonite, and high-Mg calcite. Devil’s Hole, Bermuda. Seasonally elevated p. CO 2 (at levels comparable to anticipated anthropogenic increase in atmosphere) drives dissolution today. Morse et al. , 2006
Morse et al. , 2006
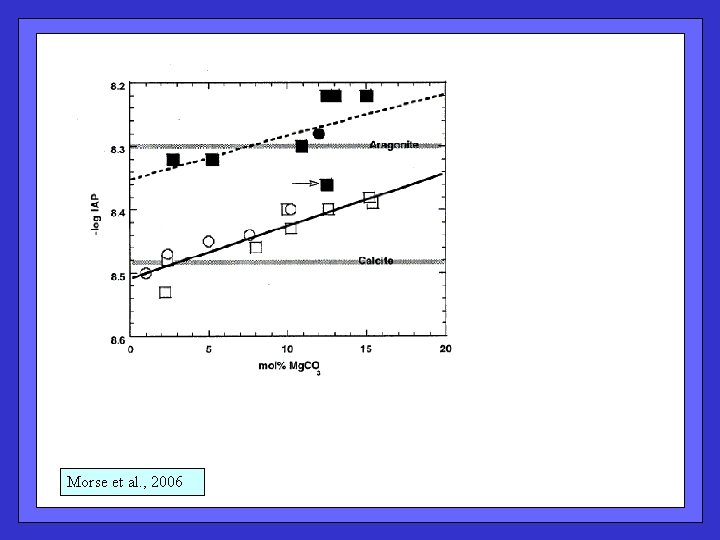
Morse et al. , 2006

Morse et al. , 2006 Multi-component shallow water dissolution model. Sequential dissolution of most-soluble phase; does not include metabolic dissolution (Andersson does); dissolution is a function of water residence time on banks.
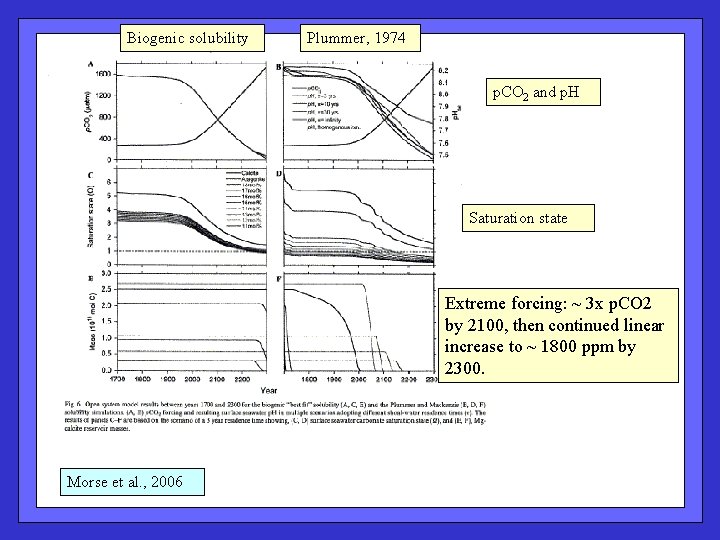
Biogenic solubility Plummer, 1974 p. CO 2 and p. H Saturation state Extreme forcing: ~ 3 x p. CO 2 by 2100, then continued linear increase to ~ 1800 ppm by 2300. Morse et al. , 2006
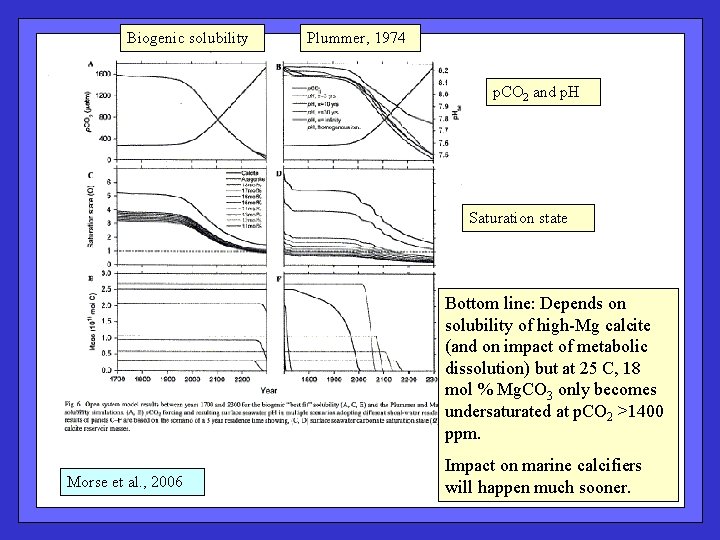
Biogenic solubility Plummer, 1974 p. CO 2 and p. H Saturation state Bottom line: Depends on solubility of high-Mg calcite (and on impact of metabolic dissolution) but at 25 C, 18 mol % Mg. CO 3 only becomes undersaturated at p. CO 2 >1400 ppm. Morse et al. , 2006 Impact on marine calcifiers will happen much sooner.
- Slides: 36