Sewage or Waste water Characteristics Wastewater consists of


























- Slides: 31

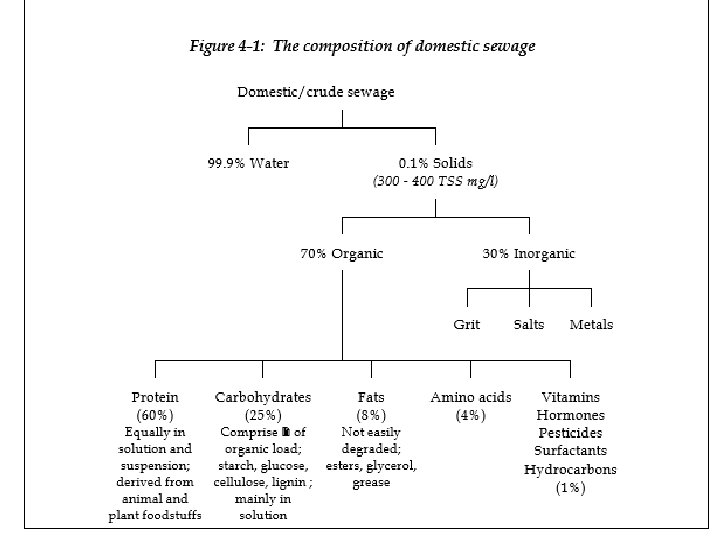
Sewage or Waste water Characteristics • Wastewater consists of human excreta, waste from domestic processes, trade effluent from industrial processes, infiltration and washing from streets and roofs in raining times. It include every thing else that find its way from consumer’s house to sewer. • Domestic sewage having almost uniform quality, but quantity will be different. Industrial effluents having different quality as well as quantity. Domestic Wastewater characteristics are usually as. In organic part of sewage carbohydrates ( 25 -50 %) Proteins (40 - 60%) and oil and fats (10%) are present. In inorganic part of sewage salt, grit and metals are present in various composition. The knowledge of wastewater characteristics is essential for design Operation and management of collection treatment and disposal. These characteristics are physical, chemical and biological.


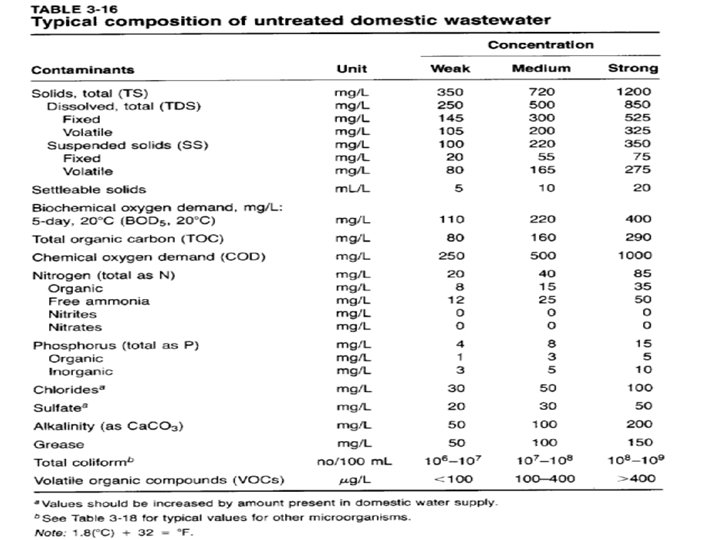
Physical Characteristics of wastewater Sewerage characteristics can be divided into three broad categories: 1. Physical (Temperature, colour, smell, Solids (2) Chemical (BOD, COD, Nutrients and Dissolved solids; and (3). Bacteriological. Physical Characteristics. The physical characteristics are its total solids content, odor, temperature, density, color and turbidity. Fresh sewage is usually gray, light brown, yellowish and having no unpleasant odor. Foul and unpleasant odor may develop as a result of septic or stale condition. Its color changes to black or dark brown when sewage becomes stale. Its odor changes within 2 -4 hours due to exhaustion of oxygen and presence of H 2 S gas in sewage. Temperature affect chemical reaction and biological activities. Solid content in wastewater are important factor for wastewater treatment processes. Solids like total suspended solids (TSS), volatile suspended solids (VSS) and settle able solids affect the operation and sizing of treatment units.

Physical Characteristics of wastewater Solids: - Solids comprise matter suspended or dissolved in water and wastewater. Solids are divided into several different fractions and their Solids like total suspended solids (TSS), volatile suspended solids (VSS) and settle able solids affect the operation and sizing of treatment units. Solids: - Solids comprise matter suspended or dissolved in water and wastewater. Solids are divided into several different fractions and their concentration provide useful information for characterization of wastewater and control of treatment processes. Total solids ; -Total solids (TS) are the sum of total suspended solids and total dissolved solids (TDS). Each of these groups can be further divided into volatile and fixed fractions. Total solids is the material left in the evaporation dish after it has dried at 103 -105 0 C. Total solids can be expressed in mg/L. Total suspended solids: - Total suspended solids (TSS) are referred to as non-filterable residue. It is determined by filtering a well mixed sample through 0. 45μm to 2 μm pore sized membrane. The residue retained on the filter is dried in an oven at a temperature of 103 -1050 C for at least 1 hour. TSS is in mg/L

Physical Characteristics of sewage Total dissolved solids: - Dissolved solid are also known as filterable residues. To determine total dissolved solids a well mixed sample is filtered through a standard glass fiber filter of 2 μm or less pore size, and the filtrate is evaporated for at least one hour in an oven at 180 C. The increase in dish weight represents the TDS. It is expressed in mg/L. Fixed and Volatile Solids: -The residue for total solids, total suspended solids or total dissolved solids tests is ignited to constant weight at 500 o C ± 50. The weight lost on ignition is called volatile solids, whereas the remaining solids represent the fixed total suspended or dissolved solids. The determination of volatile portion of solids is useful in controlling waster water treatment plant operations because it gives a rough estimation of the amount of organic matter present in the solid fraction of waster water, activated sludge and industrial waste. Settleable Solid: -Settleable solids is the term applied to the material settling out of suspension within a defined time. It may include floating material. Its units are mg/L.

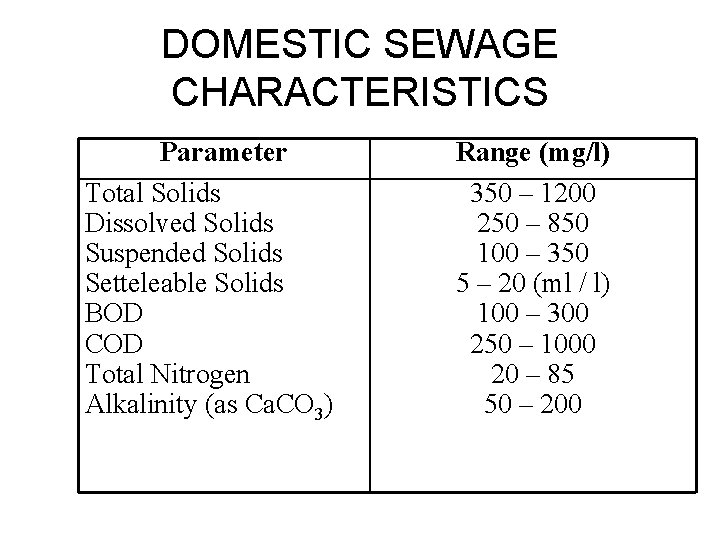
DOMESTIC SEWAGE CHARACTERISTICS Parameter Total Solids Dissolved Solids Suspended Solids Setteleable Solids BOD COD Total Nitrogen Alkalinity (as Ca. CO 3) Range (mg/l) 350 – 1200 250 – 850 100 – 350 5 – 20 (ml / l) 100 – 300 250 – 1000 20 – 85 50 – 200


Example • Solid analysis were conducted of wastewater. Based on test data determine the concentration of total solids, total volatile solids, fixed solids ; suspended solids, and volatile suspended solids. The data is weight of empty dish is 52. 842 g; weight of dish + sample 110. 0 g; weight of dish + dry solids 52. 902 g; weight of dish + ignited solids is 52. 859 g; weight of filter 0. 1160 g; sample volume 48 ml; weight of filter + dry solids is 0. 1270 ; weight of filter + ignited solids is 0. 1170 grams. Assume density of is 1 gram per ml. • Solution: Volume of sample = weight of sample+ Dish – weight of Dish = 110. 0 g – 52. 842 g= 57. 158 g As 1 gram = 1 ml so volume is 57. 158 ml. • Total dry solids = weight of Dish + dry solids - weight of Empty dish = 52. 902 - 52. 842 = 0. 06 gram • concentration in mg/liter = weight / volume = 0. 06 g/ 57. 158 ml = 0. 00105 g/ml = 1049. 7 mg/l. • Total Volatile solids = Weight of Dish + dry solid – Dish + ignited solids = 52. 902 - 52. 859 = 0. 043 gram • Concentration in mg/liter = 0. 043 g/57. 158 ml = 0. 00075 g/ml = 752. 30 mg/l. • Total Fixed solids = Total solids – Volatile solids = 1049. 7 – 752. 30 = 297. 40 mg/L • Suspended Solids • Suspended dry solids = Weight of filter + dry solids – weight of filter = 0. 1270 – 0. 1160 = 0. 011 gram • Concentration = 0. 0110 g / 48 ml = 0. 000229 g/ ml = 229. 2 mg/ l • Suspended volatile solids = weight of filter + dry solids - filter + ignited solids = 0. 1270 - 0. 1170 = 0. 0110 g • Concentration = 0. 0110/ 48 = 229. 2 mg/L

Example • Calculate the percentage of total solids and percentage of volatile solids in the sample and percentage of total solids that are volatile and fixed from the following laboratory analysis of a wastewater. • weight of dish on simple balance = 50. 2 g; weight of dish plus sludge sample= 90. 80 g ; weight of dish plus dry sludge solids = 52. 783 and weight of dish plus ignited solids = 50. 969 g; • Solution: Weight of sample = weight of dish + sludge sample – weight of empty dish = 90. 80 - 50. 2 = 40. 6 grams • Weight of dry sludge solids = Weight of dish + dry solids – weight of empty dish = 52. 783 - 50. 20 = 2. 583 gram; • Percentage of dry solids = (2. 583/40. 6) *100 = 6. 36 % • Weight of volatile solids = weight of dish + dry solids – weight of dish + volatile solids = 52. 783 – 50. 969 = 1. 814 grams • Percentage of volatile solids = (1. 814/40. 6) * 100 = 4. 47 % • Percentage of dry solids that are volatile = (1. 81/2. 583)* 100 = 70. 07 % • Percentage of fixed solids 100 - 70. 07 = 29. 93%

Chemical Characteristics • Chemical Characteristics: In sanitary sewage about 75 % of suspended solids and 40% of filterable solids are organic. These solids are derived from both animals and plant along with human. Organic compounds are usually consists of C; H; O; N along with S; P and Iron. The organic substances found in sewage are Protein (40 -60%); Carbohydrates (25 -50%) and 10% as fats and oils. Along with these organic compounds small amount of synthetic organic compounds like VOCs, Pesticides, insecticides and Organic Pollutants are also presents in sewage. Sewage also contain inorganic substances. All the test representing these organic and inorganic constituents come under the heading of chemical characteristics. Test like BOD, COD, Nitrogen, phosphorus, alkalinity etc give the chemical characteristics of sewage.

Chemical Characteristics • BOD (Biochemical Oxygen Demand): When biodegradable Organic Matters is released into a water body, microorganisms feed on the wastes, breaking them into simpler organic and inorganic substances. When this decomposition occurs in aerobic environment the process produces nonobjectionable, stable end products like CO 2, SO 4, PO 4 and NO 3. A simplified • form of Aerobic decomposition is O. M + O 2 +Microorganisms CO 2 + H 2 O + C 5 H 7 NO 2 (New Cells ) + stable Products like NO 3; PO 4; NO 3…. ) When insufficient O 2 is available Anaerobic decomposition occurs by different microorganisms. They produce end products that can be highly objectionable, including H 2 S; NH 3 and CH 4. The reaction is O. M +Microorganisms CO 2 + H 2 O + C 5 H 7 NO 2 (New Cells) + Unstable Products (NH 3; H 2 S; CH 4…. ). CH 4 is a stable gas, while other products are usually unstable. Bacteria placed in contact with organic matter will utilize it as food source.

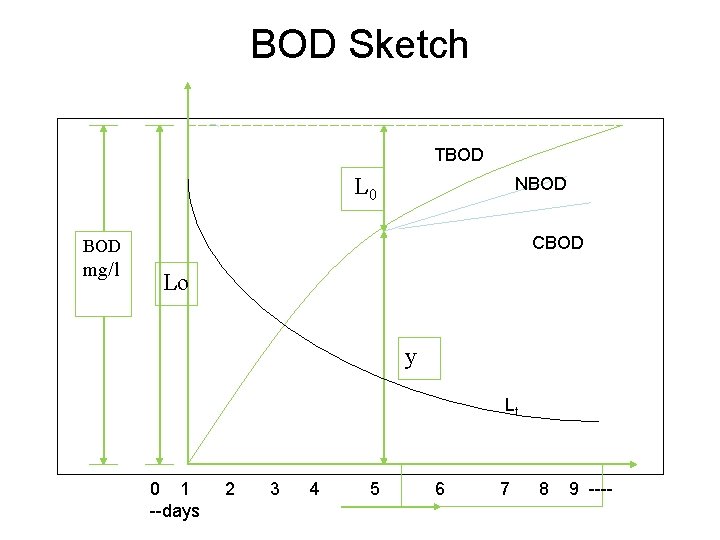
Biochemical Oxygen Demand (BOD) The amount of oxygen required by the bacteria to oxidize the organic matter present in sewage to stable end products is known as biochemical oxygen demand. Significance: Used in design of waste water treatment plants. Used to measure efficiently of waste water treatment plant. DERIVATION OF BOD EQUATION Biological oxidation of organic matter by bacteria is considered to be a first order reaction for all practical purposes. In a first order reaction, the rate of reaction is proportional to the concentration of the reactant present. So, we can say that in case of biological oxidation of organic matter by bacteria, the rate of oxidation is proportional to the organic matter REMAINING.

BOD EQUATION •

BOD EQUATION Let ‘y’ be the concentration of organic matter (BOD) consumed up to time ‘t’ y = Lo – Lt y i. e = Lo – Lo e –Kt => y = Lo ( 1 - e –Kt) BOD consumed = ultimate BOD (1 – e –Kt) in ‘t’ days Typical value of K = 0. 23 per day for domestic sewage at 20 o. C. Value of ‘K’ is temperature dependent. KT = K 20 (θ) T-20 where θ = 1. 047 K T = K 20 (1. 047) T-20 KT = Value of K at temperature T in 0 C Limitation in BOD test: The limitation of the BOD test are as i. A high concentration of active microorganisms required ii. Pre-treatment is required if toxic materials are present in wastewater iii. Only biodegradable organics are measured iv. Long time is required to calculate BOD

BOD Sketch TBOD L 0 NBOD CBOD mg/l Lo y Lt 0 1 2 3 4 5 6 7 8 9 -----days

Example Calculate the ultimate BOD for a sewage whose 5 day BOD at 20 o. C is 250 mg/l. Assume K = 0. 23 per day what will be BOD after 2 days. Solution: y = Lo (1 – e Kt) 250 = Lo (1 – 5 x 0. 23) Lo = 365. 83 mg/l y 2 = 365. 83 (1 – e – 0. 23 x 2) y 2 = 134. 89 mg/l = 135 mg/l Answer Example: The BOD 5 of waste water is determined to be 150 mg/L at 200 C. The k Value is known to be 0. 23 per day. What would be the BOD 8 if test were run at 150 C. Solution: Yt = L 0 (1 - e –kt) => Y 5 = L 0( 1 -e -0. 23* 5) 150 = L 0 (1 -e -1. 15) => 150/0. 683 = 219. 50 mg/L Now KT = K 20Θ(T-20) = K 15 = K 20(1. 047)-5 = 0. 23 * 1. 047 -5 = 0. 183 Now Y 8 = L 0 (1 -e-k. T) = 219. 5 (1 -e -0. 183*8) = 168. 73 mg/L Answer

Example The BOD remaining in a sewage sample after 4 and 8 days was 160 and 60 mg/l respectively at 20 o. C calculate the 5 day BOD of the sewage at 25 o. C. SOLUTION: BOD exerted / consumed = y BOD remaining = L L = Lo e-Kt 160 = Lo = 60 = 0. 375 = ln (0. 375) = K = Lo At 25 o. C Lo e-K x 4 Lo e-K x 8 160 / e – 4 K [160 / e – 4 K ] e–K 8 160 e – 8 K +4 K e – 4 K -4 k 0. 245 per day = 160/e – 4 x 0. 245 K 25 Y 5 Y 5 = = = =426. 3 mg/l K 20 (1. 047) T-20 0. 245 (1. 047) 25 -20 0. 308 per day Lo (1 - e –Kt) 426. 3 (1 – e – 0. 308 x 5) 335 mg/l

DERIVATION OF BOD EQUATION for Nitrogenous O. M • As Carbonaceous BOD is easily decomposed into various compounds, the Nitrogen which is a critical element for protein synthesis is bound up in complex organic molecules is released as ammonia nitrogen. Similarly the living organisms when died, nitrogen is converted into ammonia. The oxidation of ammonia required oxygen. Also it is an important nutrient for algal growth. • Increased nitrogen control utilizes aerobic bacteria to convert ammonia(NH 3 ) to Nitrite (NO 2 -). This type of bacteria is known as Nitrosomonas. The Nitrate bacteria (Nitrobactor) convert nitrite to Nitrate ((NO 3 -). This process is termed as nitrification. The Nitrogenous Oxygen demand consists of two steps. NH 3 + 3 O 2 Nitrosomonas → 2 NO 2 -+ 2 H + + 2 H 2 O • 2 NO 2 - + O 2 → Nitrobactor 2 NO 3 • The overall reaction is NH 3 +2 O 2 → NO 3 - + H+ +H 2 O

DERIVATION OF BOD EQUATION for Nitrogenous O. M As Oxygen is consumed in this reaction. This type of oxygen demand is termed as Nitrogenous Oxygen Demand. The total concentration of Organic and ammonia nitrogen in sewage is known as Kjeldahl Nitrogen (TKN). Theoretical NBOD = grams of Oxygen used / Grams of Nitrogen Oxidized =(4*16)/ 14 = 4. 57 gram of O 2/ gram of Nitrogen. For Untreated waste water Ultimate CBOD = 250 -350 mg/L; Ultimate NBOD = 70 -230 mg/L Also Ultimate NBOD = 4. 57 * TKN

Example of NBOD • A domestic sewage has 30 mg/L of nitrogen in the form of ammonia. Calculate (a) the ultimate nitrogenous oxygen demand (b) the ratio of the ultimate NBOD to the concentration of nitrogen in the waste. • Solution: the overall nitrification reaction is • NH 3 +2 O 2 → NO 3 - + H+ +H 2 O • The molecular weight of ammonia is 17, while of oxygen is 32. It means one gram mole of ammonia required two gram mole of oxygen(2*32=64). Since ammonia contains 14 gram of Nitrogen, and the concentration of nitrogen is 30 grams • So NBOD = 30 mg/L* (17 g NH 3 /14 gram of N)*(64 gram O 2/17 gram. NH 3)=137. 142 grams. • The oxygen demand to nitrogen is (137. 142 mg O 2 /L)/(30 mg N/L) = 4. 57 mg of O 2 / mg of N

Chemical Oxygen demand (COD) • In addition to CBOD and NBOD measured, there are two other indicators to describe the oxygen demands of wastewater. They are Chemical oxygen demand theoretical oxygen demand. • Chemical Oxygen Demand (COD): The biodegradable organic matters are degraded completely by microorganisms either of CBOD or NBOD. There are some organic matters like cellulose, phenols, benzene and tannic acid which are resists to biodegradation. Similarly other organic matters like pesticides, insecticides and various industrial chemicals are non biodegradable and they are toxic to microorganisms. The COD is a measured quantity that does not depend on microorganisms. To calculate the concentration of oxygen for non biodegradable materials a strong oxidizing agent known as potassium dichromate will be used. The reaction is Organic matter (Ca. Hb. Oc) +Cr 2 O 7 -2+ H 2 O - Cr +3 + CO 2 + H 2 O

COD and Theo. D • The COD test is much quicker than BOD test, but it does not distinguish between the biodegradable and non biodegradable organic matter. The measured COD is usually more than BOD if there is non biodegradable impurity present. If all are the biodegradable organic matter then COD remains the same as that of BOD. Roughly the BOD/COD is 0. 4 to 0. 8. • Theoretical Oxygen Demand (Theo. D): Organic matter of animal or vegetable origin in wastewater is generally a combination of carbon, hydrogen, oxygen, nitrogen and other elements. If the chemical composition of an organic matter is known the amount of oxygen required to oxidize it to carbon dioxide and water can be calculated using stoichiometry. This amount of oxygen is known as Theoretical Oxygen Demand. If that oxidation is carried out by bacteria then it is BOD, if by chemical process then it is COD. If a combination of both then it is Theo. D

Theo. D Example • The following data have been obtained from a waste water characterization. BOD 5 = 400 mg/L; k = 0. 29 d-1; NH 3 = 80 mg/L. Estimate the total quantity of oxygen in mg/L that must be furnished to completely stabilize this waste water. What is the COD and Theo. D for this waste. • Solution: The Carbonaceous BOD is • BOD 5 = Y 5 = Lo (1 -e(-5 k) ) => 500 = Lo (1 -e (-5*0. 29) => Lo = 500 / (1 - e (-5*0. 29 ) )= 523. 23 mg/L. • The Nitrogenous BOD is NH 3 +2 O 2 → HNO 3 +H 2 O. • (80 mg/L of NH 3 / 17 g/mol of NH 3)* (2 mol. O 2 /mol NH 3)*32 g/ mol. O 2 )= 301. 17 mg/L. • Total amount of Oxygen = 523. 23 + 301. 17 =824. 40 mg/L. • The BOD/COD varies from 0. 4 to 0. 8. let it may be 0. 6 then COD = 400/0. 6 = 666. 67 mg/L • The theoretical oxygen demand is • Theo. D = COD +NBOD = 666. 67 + 301. 17 = 967. 84 mg /L • as COD already having CBOD.

Biological/Micro bacteriological Characteristics of sewage • In sewage a lot of Suspended, colloidal or dissolved degradable organic material, present in certain quantities and ratios which depends on the nature of the wastewater. Most biological waste and wastewater treatment processes employ bacteria as primary microorganisms; certain other microorganisms may also play an important role. Bacteria is a single cell organisms. Their mode of reproduction is usually binary fission. The three categories of bacteria are spherical (0. 5 to 1. 0 μm in dia. ), cylindrical (0. 5 to 1. 0 μm in width by 1. 5 to 3. 0 μm in length) or helical (0. 5 to 5. 0 μm in width by 6. 0 to 15. 0 μm in length). Bacteria are usually consists of 80% water and 20% dry material, of which 90% is organic and 10% in organics. An approximate formula for organics fraction is C 5 H 7 O 2 N, when phosphorus is considered then the formula is C 60 H 87 O 23 N 12 P. 24

Biological/Micro bacteriological Characteristics of sewage • The inorganic portion is P 2 O 5, SO 3, Na 2 O, Ca. O, Mg O, K 2 O and Fe 2 O 3. As all of these elements and compounds are derived from environment, a shortage of any of these substances would limit and in some cases, change the growth. Population dynamics of bacteria in biological treatment depends on environmental factors which include: p. H; temperature; type and concentration of the substrate; essential nutrient concentration and its availability; (nitrogen, phosphorous, sulfur, etc. ); essential minerals; media toxicity; byproducts; and degree of mixing. 25

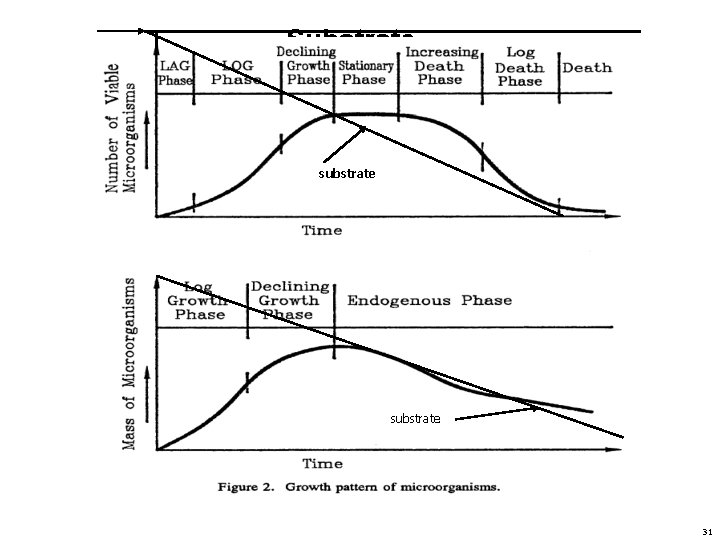
Bacteria and its growth phases • Bacteria is a microscopic organisms that normally multiply by binary fission. In binary fission the parent cell divides and breaks into two daughter cells. The division time is known as fission time or generation time and is varies with species and environmental conditions. It may be days or less, but usually about 20 minutes in favorable environmental condition is provided. Bacterial cells may have their food in soluble form and are capable of movement or incapable of movement according to the species. The formula for bacterial cell formation is 2 N. Where N =natural number. Like if generation time is 20 minutes then in 6 hours the number of microorganisms will be 262144. This is a hypothetical value, as bacteria would not continue to divide indefinitely due to various environmental limitations as food availability, nutrients concentration, p. H, temperature etc. 26

Bacteria and its growth phases • Growth Phases: When a single species of bacteria is come in contact to a medium containing substrate and all substances required for growth. The growth will occurs is based on the number of viable cells N. It follows a number of phases discussed as • Lag Phase: In the beginning of this phase microbes are becoming adjusted to their new environment. Microbes are recovering from transplant injuries and are absorbing water and substrate and becoming healthier but no new division occurs. The duration of lag phase is generally dependent on the age of added microbes and its concentration. If the parent culture is young and biologically active, the lag phase will be extremely short. 27

Bacteria and its growth phases • Log growth Phase: During the Log growth Phase, microbes starts its division. One bacterium is divided into two, two into four and so on. The rate of fission is the maximum possible the cell wall and membranes are thinnest during this phase and the metabolic activities are at the maximum rate. The cells are physiologically young, biologically active and more vulnerable to deleterious influence than mature, less active, cells. It is a phase of exponential growth and the log of the number of viable cells versus time is a straight line relationship. • The germination time depends on species and the nutrients and environmental condition and vary from few minutes to as much as several days. Usually however from 10 to 60 minutes. In this phase there is no deficiency of substrate availability and no death of microbes occurs. 28

Bacteria and its growth phases • At the end of log growth phase the cell begins to encounter difficulties such as depletion of the substrate or an essential nutrient and the accumulation of toxic end products, which may have reached an inhibitory level, which is the beginning of the declining growth phase. • Declining growth phase: The phase in which the rate of fission begins to decline and the microorganisms die in increasing number so that the increase in number of viable cells is at slower rate. Usually the declining growth phase is due to depletion of substrate and not due to an accumulation of toxic end products. The end of decline growth phase is the beginning of new phase known as stationary phase. Stationary phase: The phase in which the population of viable cells is at a relatively constant value. The time required to reach this phase depends primarily on the species, the concentration of the microbes, the composition of medium, and the temperature. 29

Bacteria and its growth phases As the environment becomes more and more adverse to microbial growth, it is the end of stationary phase and the beginning of new phase known as increasing death phase. Increasing death phase: The phase in which the cells die exceeds the growth rate. Finally the increasing death phase progresses into the log death phase. If the cell growth is measured as the total mass of viable cells produced instead of the number of viable cells, the cycle of microbes are divided into three phases. They are log phase; declining growth phase and endogenous phase. Endogenous phase is that phase in which microbes used their store food or start to eat each other due to want of food. This is an important stage in wastewater treatment 30

Substrate substrate 31