Seven Year FollowUp of a Phase 2 Clinical












- Slides: 16

Seven Year Follow-Up of a Phase 2 Clinical Trial to Induce Tolerance in Mismatched Living Donor Renal Transplant Recipients 26 th International Congress of The Transplantation Society, Sunday, August 21, 2016

Why is the pursuit of tolerance so compelling? • Better control of the immune system: potential for “one organ transplant for life”… • Financial Costs • Compliance • Elimination of IS drug related morbidity/mortality • No improvement in long-term patient and graft survival in 20 years!

Key Issues for Tolerance Induction Protocols for Clinical Application • Safety • Durability • Broad Applicability • Economics • Long term outcomes superior to that achieved with SOC approaches to SOT

Major Hurdles in Applying mismatched HSC to Solid Organ Transplant v Conditioning v GVHD v Engraftment v Donor/Recipient HLA Disparity

www. Science. Translational. Medicine. org 7 March 2012 Vol 4 Issue 124 ra 28

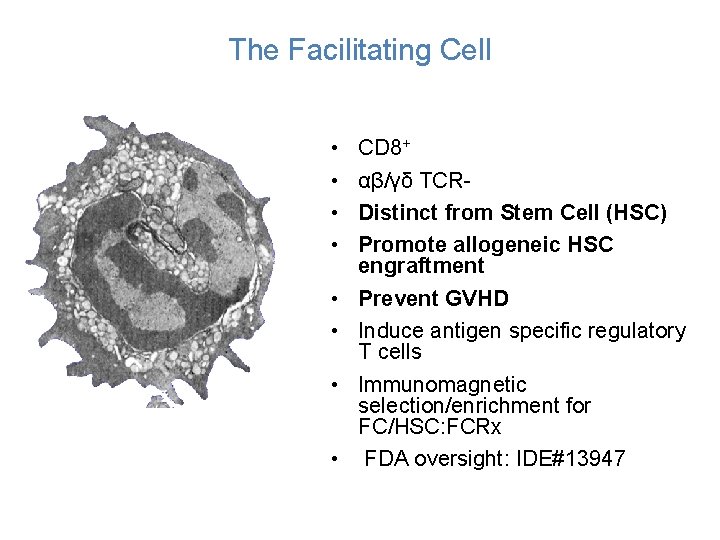
The Facilitating Cell • • CD 8+ αβ/γδ TCRDistinct from Stem Cell (HSC) Promote allogeneic HSC engraftment Prevent GVHD Induce antigen specific regulatory T cells Immunomagnetic selection/enrichment for FC/HSC: FCRx FDA oversight: IDE#13947

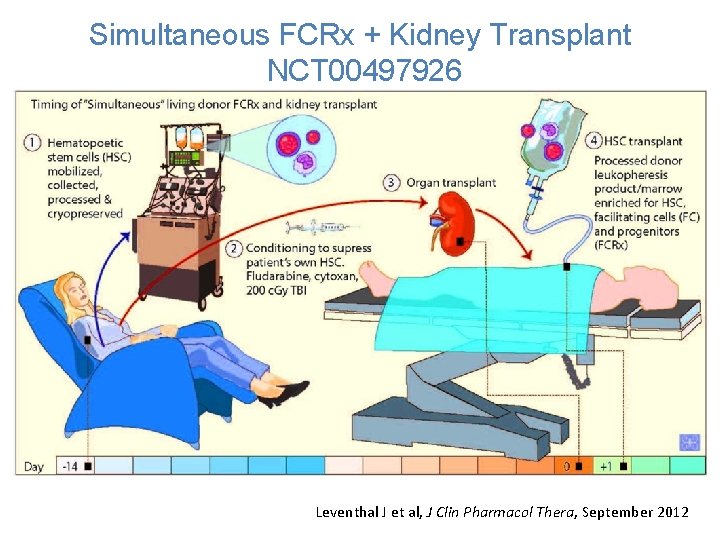
Simultaneous FCRx + Kidney Transplant NCT 00497926 • 41 patients enrolled into Phase 2 trial initiated in 2009 • 31 subjects transplanted • First 28 subjects have reached > 12 months of follow up (range 15 – 87 months)

Simultaneous FCRx + Kidney Transplant NCT 00497926 Conditioning to suppress Patient’s own Immune system. Fludarabine, Cytoxan, 200 c. Gy TBI Leventhal J et al, J Clin Pharmacol Thera, September 2012

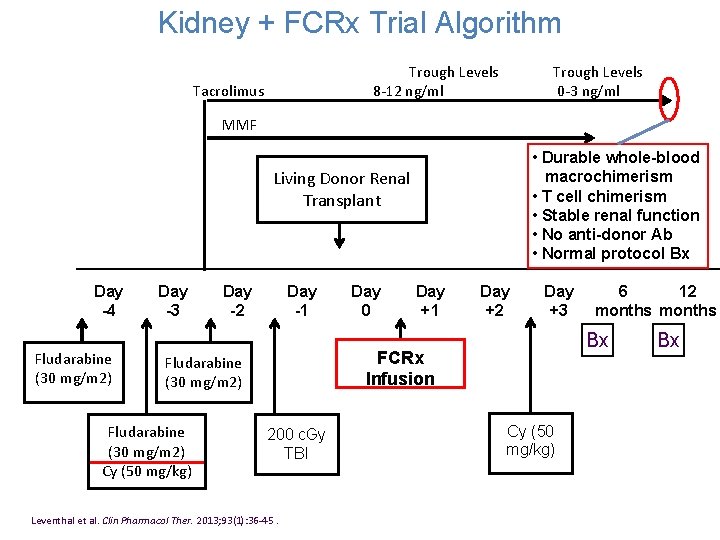
Kidney + FCRx Trial Algorithm Trough Levels Tacrolimus 8 -12 ng/ml MMF Day -4 Fludarabine (30 mg/m 2) Day -3 Trough Levels 0 -3 ng/ml • Durable whole-blood macrochimerism Living Donor Renal • T cell chimerism Transplant • Stable renal function • No anti-donor Ab • Normal protocol Bx Day -2 Day -1 Day 0 Day +1 Day +2 FCRx Fludarabine Infusion (30 mg/m 2) Day +3 6 12 months Bx Bx Cy (50 Fludarabine 200 c. Gy mg/kg) (30 mg/m 2) TBI Cy (50 mg/kg) Leventhal et al. Clin Pharmacol Ther. 2013; 93(1): 36 -45.

Patient characteristics (n=31) • Male/Female 25/6 • Age (Mean yrs) 39. 2 (range 18 -64) • LURD 13 • LRD 18 • Re –Tx 1 • ESRD cause PKD – 7; Ig. AN – 7; Reflux – 4; DM – 3; HTN -2; Membranous – 2; Chronic GN-2; Alports-1; FSGS – 1; Unknown - 2

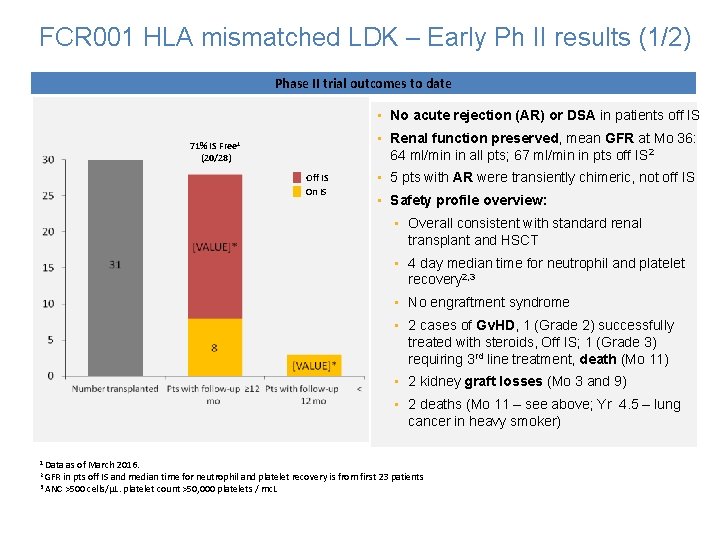
FCR 001 HLA mismatched LDK – Early Ph II results (1/2) Phase II trial outcomes to date • No acute rejection (AR) or DSA in patients off IS • Renal function preserved, mean GFR at Mo 36: 64 ml/min in all pts; 67 ml/min in pts off IS 2 71% IS Free 1 (20/28) Off IS On IS • 5 pts with AR were transiently chimeric, not off IS • Safety profile overview: • Overall consistent with standard renal transplant and HSCT • 4 day median time for neutrophil and platelet recovery 2, 3 • No engraftment syndrome • 2 cases of Gv. HD, 1 (Grade 2) successfully treated with steroids, Off IS; 1 (Grade 3) requiring 3 rd line treatment, death (Mo 11) • 2 kidney graft losses (Mo 3 and 9) • 2 deaths (Mo 11 – see above; Yr 4. 5 – lung cancer in heavy smoker) 1 Data as of March 2016. 2 GFR in pts off IS and median time for neutrophil and platelet recovery is from first 23 patients 3 ANC >500 cells/µL. platelet count >50, 000 platelets / mc. L

Patient Safety • Low intensity conditioning well tolerated: integration of HD eliminates potential ESRD-related drug toxicities • Post-transplant nadir period is brief ( < 2 weeks) and easily managed on an outpatient basis ; limited need for blood product support • Clinical interface with subjects is more robust than for SOC KTx • No engraftment syndrome • Patients regain immunocompetence to respond to mitogen (PHA) and MHCdisparate third party alloantigens over the first post-transplant year. Long-term chimeric subjects have not demonstrated clinical evidence of immune defect (no late opportunistic infections). TCR repertoire reconstitution is robust. Persistent pre-transplant vaccine-based immunity, respond to new vaccines • Chimerism has been stable following IS withdrawal; no DSA, no allograft rejection. Peripheral blood chimerism represents a good noninvasive biomarker of tolerance

Gv. HD Experience in FCRx Trial • • Gv. HD (Grade 2) D 95, flare D 106 (Skin and GI) – 48 yo M ESRD Ig. A Nephropathy 5/6 HLA mm 43 yo unrelated female donor – Occurred immediately after subject switched from TAC to SRL for CNI-induced toxicity; CMV viremia was noted (donor CMV+. recipient CMV -), CMV colitis diagnosed, treated with Ganciclovir and Foscarnet – resolved. – GVHD resolved following treatment with corticosteroids – Currently off IS Gv. HD (Grade 3) ~ D 134 (GI) – 45 yo M ESRD PCKD 4/6 HLA mm 37 yo unrelated female donor – 2 Mo postop course complicated by pneumonia (treated at local hospital) and CMV reactivation – Presented with severe diarrhea (>1 L/d) occurring around 5 days prior to presenting to local hospital; colon bx revealed Gv. HD; started on steroids and transferred to NW 6 days later with persistent diarrhea – Repeat biopsy at NW revealed combined Gv. HD/CMV colitis – Despite being treated for CMV and 2 nd/3 rd line Gv. HD, as well as bowel rest and parenteral nutrition, diarrhea persisted with continued diagnosis of Gv. HD and intermittent CMV colitis – At ~11 mo post-transplant, condition deteriorated with pulmonary process of undetermined etiology, ultimately developing septic shock which progressed to multiorgan failure and death

Optimization of FCRx Protocol: 2009 -2016 • Strict Adherence to Conditioning Regimen • Exclusion of Highly Sensitized Subjects • Enhanced Subject Follow-up • Exclusion of Female > Male Gender MM in unrelated D/R pairs • US LDK statistics @ 5 yrs: PS 84. 8%, GS 82. 9%

FCRx trial to induce chimerism and tolerance: Summary • 30/31 transplanted subjects receiving FCRx demonstrated macrochimerism post-transplant, ranging from 6% to 100% at 1 month; • Durable chimerism established in 23 of 31 subjects; the majority developed “full” (>95%) whole blood / T cell chimerism. • 21 subjects fully weaned off of immunosuppression (3 - 70 months); 2 additional subjects in final stages of weaning • 2 graft losses • 2 cases of GVHD • 2 deaths (steroid resistant GVHD/CMV (mo 11), lung cancer (yr 4. 5)

Regenerex, LLC Conflict of Interest Statement: Dr. Ildstad is the Chief Scientific Officer and Founding Scientist of Regenerex, LLC, a biotechnology company that was formed to develop and commercialize a bone marrow product to treat numerous diseases.