Session III Perspectives from EWG Members Carole Lgar

Session III: Perspectives from EWG Members Carole Légaré Health Canada Deborah Driscoll Ph. RMA Carla Brichesi ANVISA Celia Witten FDA






ICH E 6 R 3: Perspectives from Sponsors Deborah Driscoll 4 June 2020 International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use

Ensuring the intent & spirit of GCP during this time of rapid acceleration and change Patient Safety & Quality 8

Build a Foundation for Modern Clinical Research Innovative & Adaptive Trial Design Flexibility & Agility in Study Conduct Enabled with Modern Technology Leveraging New Data Sources 9

Stakeholder Engagement on ICH E 6(R 3) Good Clinical Practices Perspectives from EWG Members: ANVISA June 4 th, 2020 Carla Brichesi Regulation and Health Surveillance Specialist ANVISA - Brazilian Health Surveillance Agency
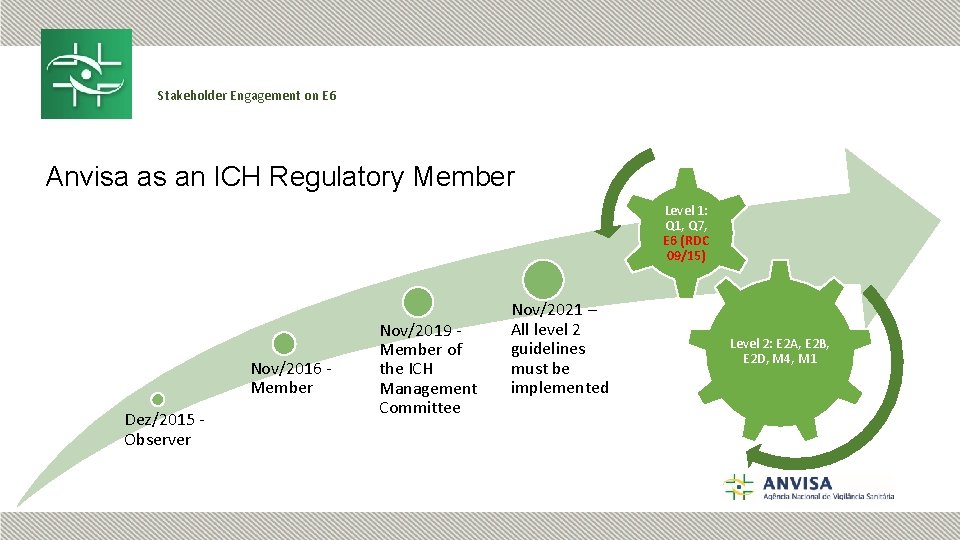
Stakeholder Engagement on E 6 Anvisa as an ICH Regulatory Member Level 1: Q 1, Q 7, E 6 (RDC 09/15) Nov/2016 Member Dez/2015 Observer Nov/2019 Member of the ICH Management Committee Nov/2021 – All level 2 guidelines must be implemented Level 2: E 2 A, E 2 B, E 2 D, M 4, M 1

Stakeholder Engagement on E 6 Anvisa’s main considerations for E 6 due to the Covid-19 pandemic v IMP sent directly to participants’ home • Good distribution practices • Keep adequate records of drug transportation and distribution v Remote Source Data Verification • Efficacy endpoints and safety data • Confidentiality of study participant • Upload of documents in a cloud, video or telephone review of medical records, remote access to EHR

Stakeholder Engagement on E 6 Main points to be reviewed in E 6 based on GCP inspections at Brazilian sites: v Protocol: This document must be clear, concise and avoid unnecessary procedures (unnecessary burden for participants). v Training/Qualification: GCP training may not be necessary for professionals performing their daily/routine activities. v Medical Care of Trial Subjects: New types of technology used in medical decision (machine learning / artificial intelligence).

Stakeholder Engagement on E 6 v ICF: New forms of consent: electronic consent, electronic signature, consent process done remotely Utilization of different medias (like videos) to support the comprehension of the study Informed Assent for minors v Trial Design: Engagement of patient representatives when designing a protocol v Investigator Selection: Investigator should be a qualified medical doctor in the field of the trial

Thank you!

Good Clinical Practice ICH-E 6 GCP Perspectives from the Expert Working Group Celia M. Witten, Ph. D. , M. D. Deputy Center Director, CBER/FDA June 4, 2020 Stakeholder Engagement on ICH E 6 Guideline for Good Clinical Practice Web Conference Organized by FDA in Collaboration with the Clinical Trials Transformation Initiative (CTTI)

ICH-E 6 an Important Global Standard • Clinical trials as global enterprises • Clinical trials are increasingly multinational (whether for large diseases or rare diseases) • International standard for GCP would help facilitate clinical development and ensure the integrity of the results • Why revise ICH E 6(R 2) • Research landscape is evolving • Trial designs are increasingly innovative – E. g. , decentralized trials • Technology is evolving on all fronts • Need to maximize efficiencies to facilitate innovations and clinical trials

Why revise ICH E 6(R 2) Evolution of evidence generation platforms • Digitization of research and healthcare • Real-world data sources • • Use of Electronic Health Records Patient-generated data Data gathered from digital health tools Telemedicine/remote assessments of patients for safety & physician reported outcomes Examples of current work streams • The use of EHR to generate evidence • Utility of digital health tools • Decentralized clinical trials • Clinical trials in healthcare settings • Data standards

ICH-E 6 an Important Global Standard Lessons and examples from public health emergencies • • Safety of trial participants is paramount Remote assessment Remote monitoring Leveraging existing infrastructure Identifying critical processes and prioritize them Employing all available technological tools and innovations to ensure trial integrity Effective communication

Contact Information Celia Witten, Ph. D. , M. D. Deputy Director, FDA Center For Biologics Evaluation And Research 10903 New Hampshire Ave Silver Spring, MD 20993 240 -402 -8351 celia. witten@fda. hhs. gov
- Slides: 20